Follicular Conjunctivitis
Uday Bhatt
Ruby Lagnado
Harminder Singh Dua
DEFINITIONS
Follicles
A follicle is a nonspecific conjunctival response to chronic irritation: mechanical, chemical, or microbial. Conjunctival follicles do not appear in the neonatal period because the lymphoid system is immature, and they are less apparent in children under the age of 2. After the first few weeks of life, lymphoid tissue becomes evident in the human conjunctiva, with a diffuse and indiscriminate distribution. Under the influence of chronic irritation or stimulation, the lymphocytes increase and aggregate into follicles resembling, in structure and function, the lymph nodes or the solitary lymphatic patches of the intestine.1, 2
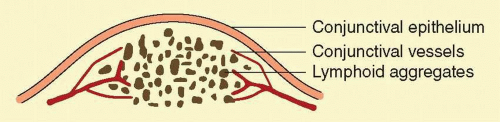
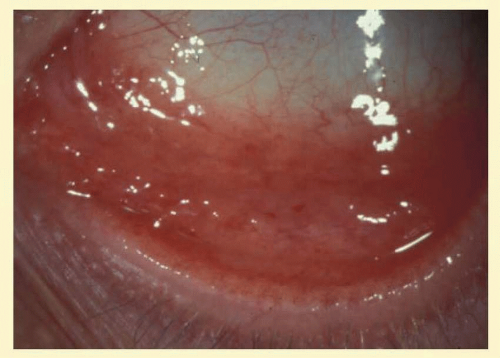
Clinically, follicles are yellowish to grayish white, discrete, round elevations of the conjunctiva. The central part of the follicle is avascular, but dilated blood vessels may surround the base and sweep up from the base over the convexity (Fig. 7.1). Follicles are 0.2 to 2 mm in size, although larger follicles may be seen, particularly in chlamydial disease.3 They can be seen with the naked eye, but with the slit lamp their apex looks a bit pale as the conjunctival vessels are blanched due to the pressure of lymphoid accumulation in a small space.
Follicles on the superior tarsal conjunctiva are well defined as a result of the close adherence of the tarsus to the epithelial mucosal tissues. They are also seen in the conjunctival fornices, where they are less regularly spaced and tend to be larger (Figs. 7.2 and 7.3). Histologically, follicles consist of lymphoid germinal centers with fibroblasts in the center. T cells occupy the periphery, and B cells the center of the infiltrates.4
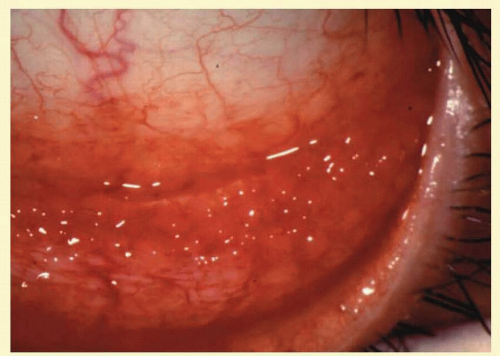
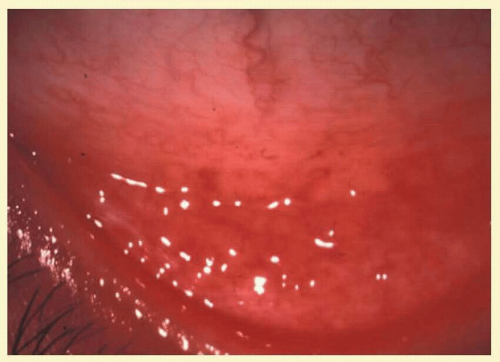 FIG. 7.2 Nonspecific follicular conjunctivitis demonstrating the classical appearance of follicles in the inferior fornix. (Courtesy of Peter R. Laibson, MD.) |
Papillae
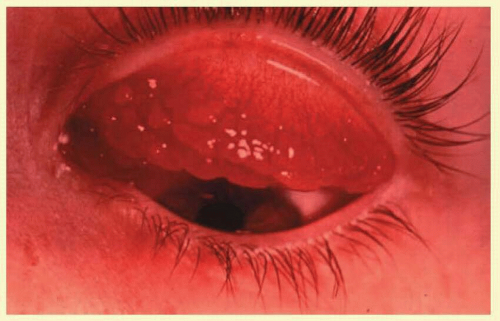
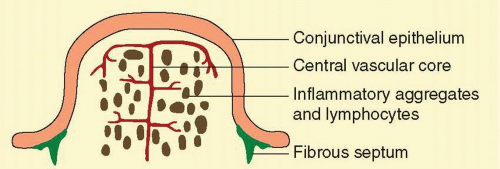
Follicles are often clinically confused with conjunctival papillae (Fig. 7.4). Papillae are vascular changes most prominent on the palpebral conjunctiva, where fibrous septa anchor the conjunctiva to the underlying tarsus. In contrast to follicles, they are small flat projections of the conjunctiva with a dilated vascular core (with side branches along the way) surrounded by edema and a mixed inflammatory infiltrate producing raised elevations of the conjunctival epithelium. The presence of the central core of blood vessel (cf. follicle with blanched apex) and tabloid shape distinguish it from the follicles (Fig. 7.5). They are nonspecific signs of conjunctival inflammation, most commonly associated with allergic or bacterial conjunctivitis.
Follicular Conjunctivitis
Follicular conjunctivitis is not a specific disease entity, but a connotation for a large number of conditions of varying etiologies that have follicles as a predominant pathologic and clinical manifestation.
MICROANATOMY OF THE FOLLICLE
Regardless of the inciting agent, histologically the structures of follicles are very similar, with presence of immature cells centrally and mature cells peripherally. The characteristic feature is a dense, localized infiltration of the subepithelial tissues by large mononuclear lymphocytes. The larger central mass is surrounded by smaller lymphocytes that frequently form secondary nodules. Other cells, such as plasma cells and polymorphonuclear cells, may appear in certain conditions. The cells of the follicle are supported by a loose and ill-defined reticulum that blends with the surrounding connective tissue. There is no true capsule, but in certain conditions, especially trachoma, a dense, surrounding sheet of connective tissue appears with increasing cicatrization.1, 2 Initially, there are prominent lymphatics packed with mononuclear cells and a network of blood vessels surrounding a follicle. Later, endothelial lined spaces may develop and extend toward the center of the follicle.
FOLLICULOSIS
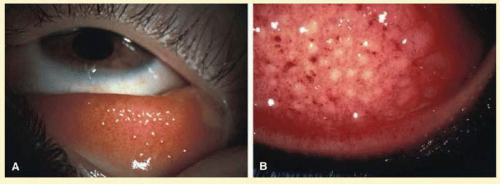
Folliculosis of childhood is not pathologic, but is a physiologic change of childhood and adolescence in which follicles are prominent in the fornix and decrease toward the lid margin (Figs. 7.6 and 7.7). There are no associated signs of inflammation, and the condition is analogous to the lymphoid hyperplasia of tonsils in children. Clinically insignificant follicles are seen frequently on the upper border of the tarsus, whereas follicles present in the central portion of the upper tarsal plate tend to suggest an underlying pathologic process.
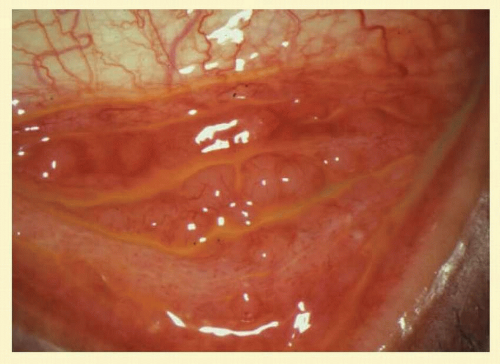 FIG. 7.6 Nonspecific follicular hypertrophy in an asymptomatic young adult. (Courtesy of Peter R. Laibson, MD.) |
ADENOVIRUSES
Pathophysiology and Taxonomy
Adenoviruses are double-stranded DNA viruses surrounded by a nonenveloped polypeptide icosahedral capsid with glycoprotein projections. They were first detected in 1953 as a latent virus in surgically removed adenoidal tissue from asymptomatic children.5 There are at least 52 serotypes of adenovirus, and they have been classified into seven species, A to G, on the basis of their hemagglutination properties and biophysical and biochemical criteria.6 In addition, recently two more serotypes, Ad 53 and 54, have been identified to cause Epidemic keratoconjunctivitis (EKC) in Japan.7 Most of the recently described serotypes (42 to 49, 51) belong to species D and were first isolated from human immunodeficiency virus (HIV)-infected patients. Adenoviruses are a common cause of a variety of human illnesses, accounting for approximately 10% of all febrile illnesses in infants and 5% of all such illnesses in children.8
Ocular Adenoviral Infection
Ocular adenoviral infections have a variety of clinical presentations; the two most common forms are EKC and pharyngoconjunctival fever (PCF). Both are characterized by a marked follicular response. Two less common manifestations are nonspecific sporadic follicular conjunctivitis and chronic papillary conjunctivitis.
The different syndromes tend to be caused by distinct serotypes, although the features do overlap. Most of the keratoconjunctivitis and all of the epidemic keratoconjunctivitis types are caused by species D; however, serotype 4, the only included species E, is also known to cause conjunctivitis.6 Ad 3 and Ad 4 are the most common pathogens that cause sporadic keratoconjunctivitis and PCF. Ad 4, 8, 19, and 37 have been implicated in sporadic cases as well as outbreaks of severe EKC.9, 10, 11 and 12 Although an epidemic due to both types 8 and 19 has been reported, most disease is due to a single serotype.
Epidemic Keratoconjunctivitis
At least 21 documented serotypes cause EKC. The most commonly associated serotypes include adenovirus 8, 19, and 37, and, less frequently, serotypes 2-5, 7, 9-11, 14, 16, 21, 29, 53, and 54.6, 7 The clinical features of adenoviral keratoconjunctivitis were first described by Fuchs in 1889,13 but it was not until 1955 that adenovirus was identified as the cause of the disease.14 The natural level of immunity against adenoviruses is low in the general population, and therefore every individual is susceptible.
Adenovirus is highly contagious, and measures to limit the spread of infection should be taken. Spread is by person-to-person contact, contaminated ophthalmic instruments, or swimming water. Transmission in ophthalmology clinics has been well reported, most frequently as a result of applanation tonometry; however, slit lamp examination and application of eye drops may also result in spread, emphasizing the need for adherence to strict hand-washing protocols.10, 15, 16 In Asia, however, EKC is endemic and does not appear to be transmitted through medical intervention.
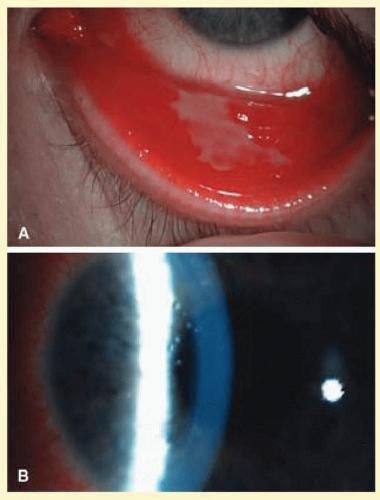
EKC has a wide spectrum of duration and intensity of symptoms. After an incubation period of 2 to 14 days, symptoms begin in one eye; symptoms develop in the second eye 2 to 4 days later. Signs include conjunctival hyperemia and chemosis, intense tearing, and swelling of the conjunctival plica. Ipsilateral preauricular lymphadenopathy is found in many patients. The corneal involvement helps distinguish it from other causes of viral conjunctivitis (Figs. 7.8 and 7.9).
Corneal epithelial involvement is noted 2-4 days after the onset of symptoms and takes the form of diffuse fine epithelial keratitis and may stain with fluorescein and rosebengal. The epithelial keratitis may persist for 2 to 3 weeks. About a week after the onset of symptoms, larger nummular lesions develop. These have irregular, raised borders and a central ulceration that stains with both fluorescein. At 2 weeks, the subepithelial infiltrates (SEIs) begin to appear underneath the focal epithelial lesions. These infiltrates consist of lymphocytes, histiocytes, and fibroblasts accompanied by a disruption of the collagen fiber of Bowman layer.17 The nummular opacities are thought to represent persisting viral replication in subepithelial keratocytes, triggering a host reaction. The immune basis is supported by the clinical observation that the opacities usually resolve with topical steroids, but recur when the steroids are discontinued.18 Rarely, these nummular opacities or infiltrates may impair visual function and may persist for months or years. Usually, the conjunctival inflammation and epithelial disease resolve in 10 to 16 days, but may last up to a month. The follicles and lymphadenopathy resolve more gradually in 2 to 4 weeks; however, the SEIs may persist for many months. EKC is also the commonest cause of viral membranous or pseudomembranous conjunctivitis (Fig. 7.10).
Management
Currently, there is no specific antiviral therapy available to treat adenoviral keratoconjunctivitis. Cidofovir, or HPMPC (3-hydroxy-2-phosphonylmethoxypropyl cytosine), is a broad-spectrum antiviral agent19 shown to have significant antiviral activity, reducing viral shedding in rabbits with adenoviral conjunctivitis.20 In humans, cidofovir 1% was shown to lower the frequency of severe corneal opacities, but its clinical use 4 to 10 times daily is limited by local toxicity.21 The mainstay of treatment is symptomatic in mild cases. In more severe cases, topical corticosteroids are useful where there are SEIs in the visual axis. Topical steroids should otherwise be used with caution in follicular conjunctivitis, because herpetic conjunctivitis can occur without corneal involvement. If low-dose steroids are used, they may need to be gradually tapered over months to prevent recurrence.
Adenoviruses are highly stable and resistant to solvents; however, they are deactivated by hypochlorite solution. The live virus has been found to survive as long as 35 days on plastic surfaces.11 Patients should avoid touching their eyelids and wash their hands frequently. It has been reported that nearly 50% of patients with adenoviral conjunctivitis carry it on their hands.16 Although most patients are culture negative 7 to 10 days after the onset of symptoms,22 positive cultures have been reported for up to 12 months after infection.23 Infected children should be advised to stay at home for 1 week. In the setting of hospital outbreaks, it is essential to implement strict procedures to reduce transmission, and infected personnel should remain off work for 2 weeks.
Conjunctival adenovirus infection is highly contagious and occurs worldwide sporadically and epidemically. Although not permanently blinding, adenoviral ocular infection remains the most common ocular viral infection worldwide. The social and economic cost of community epidemics is high.24
Pharyngoconjunctival Fever
Adenoviruses also produce the classic triad of fever, pharyngitis, and acute follicular conjunctivitis in PCF. PCF occurs more commonly in children, and is spread by respiratory secretions. It is caused by serotypes 3 and 7, but serotypes 2, 4, and 14 have also been documented as etiological agents. In addition, sporadic outbreaks caused by serotypes 1, 5, 6, 8, 11, and 19 have been reported.25, 26 and 27 The symptoms are similar to EKC, with tearing and foreign body sensation that is initially unilateral. The follicular conjunctivitis produces a watery discharge with associated preauricular or submandibular lymphadenopathy, and occasional petechial hemorrhages. Although corneal involvement is not a key feature, there is occasionally a punctate keratitis; however, SEIs are rare. PCF is a selflimiting condition that varies in severity and may last from 4 days to 2 weeks. The virus remains in the conjunctiva for 2 weeks, but fecal excretion can last a month.27 Treatment is symptomatic, and topical steroids are rarely needed.
Nonspecific Follicular Conjunctivitis
It is actually a very mild form of conjunctivitis and consequently usually seen in the physician’s office. It can occur in any of the serotypes of adenoviruses that can cause EKC or PCF. It resolves spontaneously over 7 to 10 days without any sequelae. It is endemic and actually serves as a reservoir for more severe outbreaks.28
RNA VIRUSES
Paramyxoviruses
Measles—Rubeola
The measles (rubeola) virus causes photophobia, an acute catarrhal papillary conjunctivitis, superficial punctate keratitis, superficial pannus, and occasionally Koplick’s spots on the conjunctiva and caruncle.29 Rarely, an immune interstitial keratitis may occur. In a healthy adult, four types of corneal lesions were identified: small, punctate epithelial lesions staining only with rose-bengal; small, round, or larger and irregular (when they coalesced) epithelial defects; large or tiny filaments; and target lesions when stained with both fluorescein and rose-bengal (alternating concentric rings stained with fluorescein and rose-bengal, outline stained with rose-bengal).30 The disease usually resolves with no ocular sequelae. However, in debilitated individuals, particularly those who are vitamin A deficient, it can progress to severe keratitis and keratomalacia.31, 32 Herpes simplex virus (HSV) infection also may affect children with measles, and may be responsible for onefifth of their corneal ulcers.32
Mumps
Conjunctivitis is a common manifestation of mumps characterized by dilatation of the conjunctival and episcleral vessels without marked secretion, and sometimes by chemosis of the bulbar conjunctiva.33 It is self-limiting and resolves spontaneously. A follicular conjunctivitis is not typical, but was reported once in the literature.34 The other common ocular features are dacryoadenitis and punctate keratitis followed by interstitial keratitis. A case of keratouveitis has also been described.35
Newcastle Disease
Newcastle disease is of worldwide distribution in fowls, but in humans, it was first reported as a case of conjunctivitis in a laboratory worker by accidental inoculation of infectious material.36 Subsequently many other outbreaks have been reported.37, 38 The virus and the vaccine cause a limited infection in humans. It is unique in causing a unilateral follicular and papillary conjunctivitis with hyperemia, edema, and chemosis, mainly in the lower fornix associated with mild tearing and preauricular adenopathy.39 Rarely, there is a transient, fine, superficial keratitis in the first week.40 The disease is self-limiting and does not require therapy.
Togaviridae
Childhood rubella is often accompanied by a catarrhal conjunctivitis and sometimes a mild follicular conjunctivitis. In around 2% of cases, approximately 1 week after the appearance of rash, there may be a mild superficial punctate keratitis in the central cornea associated with photophobia and tearing.41 The acute disease usually resolves spontaneously without leaving any sequelae.42
Picornoviridae
Acute Hemorrhagic Conjunctivitis
Acute hemorrhagic conjunctivitis (AHC), also known as Apollo 11 disease, has affected millions of people since it first appeared in Ghana in 1969. Large epidemics have occurred in Africa and Asia, and it was first reported in the Western Hemisphere in 1981 in Brazil.43
The major causative agents are enterovirus 70 (EV-70) and coxsackievirus type 24 (CA-24).44 Adenovirus types 8, 19, and 37 have also recently been recognized as causes of hemorrhagic conjunctivitis, and this may herald a new stage in the evolution of these viruses.45 The clinical features of AHC caused by EV-70 have, in more recent epidemics, been reported to be milder, with a maintained high transmission rate that may be due to biological transformation of EV-70.46 Apart from conjunctival secretions, respiratory and oral transmission of coxsackievirus CA-24 variant would explain the rapid and extensive spread of AHC during an outbreak.47
The disease is characterized by a short incubation period of 1 to 2 days and sudden onset of lacrimation, lid swelling, itching, foreign body sensation, and periorbital pain, which rapidly become bilateral. The common signs are papillary conjunctivitis with a minor follicular response; hyperemic conjunctival chemosis, characteristic subconjunctival petechial, or sheetlike hemorrhages appearing as concentric ridges encircling the corneal limbus, which are more pronounced temporally; a fine punctate epithelial keratitis; and preauricular adenopathy. The hemorrhages are petechial at first, but rapidly become confluent. Subepithelial opacities and erosions are rare, and the conjunctivitis resolves within 3 to 5 days, although the hemorrhages may take longer. Respiratory and gastrointestinal involvement may occur.48 More rarely, neurologic complications, including Guillain-Barré syndrome and Bell’s palsy, may develop.49 Corneal superinfection may occur after AHC, and use of topical steroids may predispose an already compromised cornea to develop microbial keratitis. Folk remedies, such as the use of human urine on the eye, have also resulted in gonococcal ophthalmia.50
Orthomyxoviridae
The orthomyxoviruses include the influenza virus, which can cause an acute follicular conjunctivitis with mucopurulent discharge.
DNA VIRUSES
Poxviridae
Parapoxvirus
Orf virus is a member of the paravaccinia group of DNA viruses that cause a contagious pustular dermatitis, predominantly in sheep and goats.51 Transmission of Orf virus to humans occurs after contact with infected or recently vaccinated animals and/or fomites in conjunction with skin trauma. It occasionally affects humans, commonly causing lesions of the hand. Ocular infection is unusual; however, it was reported in a farmwife who developed a raised ulcerated lesion at the inner canthus, follicular conjunctivitis, and lymphadenopathy after handling sheep.52 This may have been as a result of direct finger inoculation occurring while she carried an infected lamb. Treatment in this case was with idoxuridine 0.5% for the eye, with resolution in 3 weeks. Blindness has been reported in human orf infection, possibly because of secondary bacterial infection.53
Herpes Simplex
Primary herpes simplex infection is frequently associated with blepharitis and a vesicular lesion on the lid margin, a clinical feature that aids diagnosis. It typically occurs in children and may have associated upper respiratory symptoms and stomatitis. Half of all primary HSV infections involving the lid margin will develop corneal epithelial manifestations ranging from fine punctate epithelial staining to dendritic ulceration.54, 55 However, frank geographical keratitis has also been reported in primary infection.56 In primary herpetic eye disease, the ulceration is more commonly fine microdendrites involving the cornea and conjunctiva, unlike the prominent dendrites in recurrent disease.
The incidence of HSV ocular infection presenting as acute follicular conjunctivitis in the absence of corneal or lid signs in cases of clinically diagnosed epidemic keratoconjunctivitis is 1.3% to 7%.57, 58 The clinical features of primary and recurrent HSV conjunctivitis are quite similar.59 The corneal changes are rare, and it is bilateral in 13% of cases.59 It has a mean duration of 8 days. The low rate of bilateral disease and the short duration may help discriminate HSV from adenoviral conjunctivitis, as clinically differentiating the two disease entities is extremely difficult, particularly in the early stages.
Most herpetic infections of the eye are caused by human herpesvirus type 1 (oral); however, type 2 (genital) may occur in the newborn and adults after oral-genital inoculation. Neonates who develop herpes simplex primary conjunctivitis should receive intravenous treatment with acyclovir in addition to topical treatment.
The Herpetic Eye Disease Study (HEDS) assessed whether long-term prophylactic treatment with acyclovir would prevent recurrence of ocular herpetic disease.60 This was a prospective randomized, multicenter trial, and although it did not primarily address herpetic conjunctivitis, its findings did suggest that the use of 800 mg (or 400 mg b.i.d.) oral acyclovir daily over a year reduced the recurrence of ocular herpetic disease.
Cytomegalovirus
Epstein-Barr
Epstein-Barr virus (EBV), a ubiquitous DNA herpesvirus, is the most common causative agent of infectious mononucleosis syndrome. Ocular manifestations include a follicular or membranous conjunctivitis with or without subconjunctival hemorrhages.63, 64 Discrete granular subepithelial infiltrates may be seen, with overlying punctate epithelial keratitis.65 The treatment of EBV-associated ocular disease is not defined because of the small number of documented cases. Other ocular complications include uveitis, choroiditis, retinitis, papillitis, and ophthalmoplegia.63
ADULT INCLUSION CONJUNCTIVITIS (PARATRACHOMA)
Chlamydia trachomatis is an obligate intracellular bacterium that can cause several different conjunctivitis syndromes, varying in nature according to which serotype is involved. Serotypes A to C are responsible for trachoma, and are discussed in the section on chronic conjunctivitis.
Serotypes D to K of C. trachomatis cause an acute follicular conjunctivitis with a mucopurulent discharge. Historically, the diagnosis was made after visualization of chlamydial inclusion bodies on the conjunctival smears, thus giving the disease its name. These trachomata are transmitted sexually, may be asymptomatic, and may involve multiple sites simultaneously. Transmission is through direct contact of the eye with infected genital or urinary secretions.66, 67 and 68 Indirect transmission can occur in poorly chlorinated swimming pools.69 Eye-to-eye transmission is probably more common than previously thought.70
Adult inclusion conjunctivitis (AIC) is more common in developed countries with the reported prevalences between 0% and 28%.71 It occurs more commonly in the young and sexually active; within 2 to 3 months after the patient has a new sexual partner. The serious nonocular complications of chlamydial infection, such as epididymitis and salpingitis, make it an important diagnosis to establish and treat. Chlamydia has been reported to be the cause of a follicular conjunctivitis in adults in 5% to 18% of conjunctivitis.72
Approximately 40% to 50% of men with nongonococcal urethritis and 70% of their partners are infected with C. trachomatis, although 25% of the females are asymptomatic.73
Serotypes D to K produce a syndrome clinically distinct from trachoma and do not cause permanent visual loss. It is not clear why AIC is more benign, but reasons may include a lack of re-exposure to the agent, the patient’s age at initial infection, and a lower innate pathogenicity of the organism.
Clinical Features
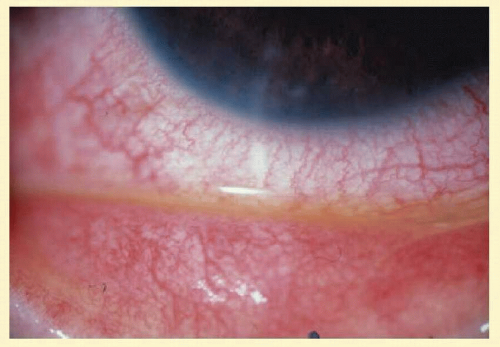
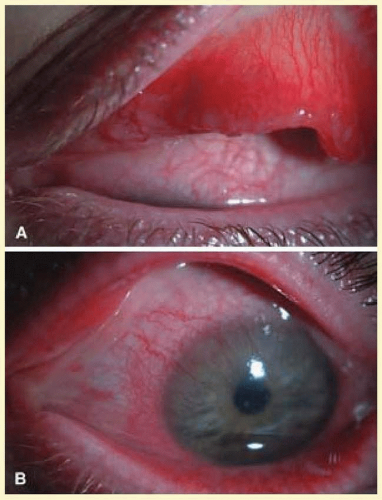
The disease affects initially one eye, 1 to 2 weeks after exposure to the organism. The symptoms may be acute or subacute, with redness, irritation, and a mucopurulent discharge. A palpable, nontender preauricular node may develop, but there are no systemic symptoms. Most of the conjunctival findings are located inferiorly, with a predominantly follicular reaction also associated with some degree of papillary reaction (Fig. 7.11). Limbal swelling is also often present.
Pseudomembranes do not occur, and conjunctival scarring, if it does occur, is minimal. In the second week, the cornea may have a mild or moderate superficial punctate keratitis that may be fine or coarse. Later in the infection, peripheral epithelial or subepithelial infiltrates may occur, which have an intact epithelium. Occasionally, iritis will occur in the later stages and, rarely, Reiter’s syndrome. A superficial micropannus may develop in those who had subepithelial infiltrates, but there is no frank corneal scarring.
Diagnosis
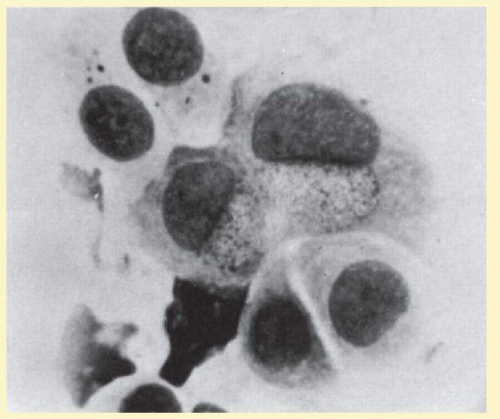
After a detailed history and clinical examination a fluorescent monoclonal antibody test is the most sensitive method of rapid diagnosis.74 Giemsa-stained conjunctival smears looking for perinuclear inclusion bodies have a high false-negative rate75 (Fig. 7.12). In recent years, polymerase chain reaction (PCR) assay has become more widely used as an effective diagnostic tool to accurately detect the presence of chlamydial material in ocular specimens (see section on “Laboratory Diagnosis”).
Treatment
Patients and their partners should be screened for nonocular chlamydial infection and for other venereal diseases. Systemic treatment is mandatory. The recommended treatment for chlamydial infection is azithromycin 1 g as a single daily dose for 7 days, or doxycycline 100 mg twice daily for 7 days. Alternative regimes are erythromycin 500 mg four times a day for 7 days, or ofloxacin 300 mg twice a day for 7 days.76
NEONATAL INCLUSION CONJUNCTIVITIS
Neonatal inclusion conjunctivitis (NIC) is caused by Chlamydia serovars D to K and is the most common cause of infectious neonatal conjunctivitis worldwide.75 It is transmitted through infected genital secretions during passage through the birth canal. The frequency of genital chlamydial infection in women from developed countries may be 2% to 8%,77 increasing with lower socioeconomic status. Thirty percent to 40% of untreated women will have babies affected with NIC, and 10% to 20% will develop chlamydial pneumonitis.78
Acute conjunctivitis with purulent discharge develops 5 to 12 days after birth. This may manifest earlier if there is premature rupture of membranes. The infection may initially be unilateral, with a papillary reaction, as the immune system is too immature to cause a follicular reaction, and there is no preauricular lymphadenopathy. Pseudomembranes occur in more severe cases as does corneal scarring and neovascularization. Diagnosis is based on laboratory tests. Giemsastained inclusion bodies may often be seen, unlike in AIC. Monoclonal fluorescent antibody staining should also be done to assist in the rapid diagnosis. Although McCoy cell culture may be used as a diagnostic tool, the advent of PCR techniques has enabled rapid and accurate diagnosis.79 PCR is a better diagnostic tool in mild disease.80
About half of neonates with chlamydial pneumonitis have had NIC.81 Treatment is oral erythromycin syrup 50 mg/kg/day in two to four divided doses for 14 days. The mother also requires systemic treatment. Tetracycline is contraindicated for the baby or for the mother if she is breast-feeding. Neonatal ocular prophylaxis with silver nitrate or antibiotic ointments does not prevent perinatal transmission of C. trachomatis from mother to infant; however, these agents do prevent gonococcal ophthalmia.
CHRONIC FOLLICULAR CONJUNCTIVITIS
Follicular conjunctivitis that lasts for more than 16 days is considered chronic and may persist for months to years. The causes of chronic follicular conjunctivitis are listed in Table 7.1.
Chlamydia Trachomatis
Trachoma is an ancient disease and remains the most common cause of chronic follicular conjunctivitis and the foremost cause of blindness worldwide after cataract.82 It is the most common cause of preventable blindness. Trachoma was described in the Egyptian Ebers papyrus in 1900 BC and remains endemic to that country. It was endemic in Europe into the 20th century. Would-be immigrants to the United States were screened for trachoma on arrival at Ellis Island, and those with the characteristic follicles were turned away.
Trachoma has disappeared from Western Europe and North America as a result of improved living conditions. However, trachoma continues to affect 150 million people in 48 countries, of which 6 million are blind.82 In areas where trachoma is constantly present at high prevalence, active disease is found in more than 50% of preschool children. As many as 75% of women and 50% of men over the age of 45 may show signs of scarring disease.83 Infected children are the major reservoir for trachomatous infection in a community.84, 85 The prevalence of active trachoma decreases with increasing age, with less than 5% of adults showing signs of active disease. Although similar rates of active disease are observed in male and female children, the later sequelae of trichiasis, entropion, and corneal opacification are more common in women than men.86 This is probably related to the close contact of women with children who are suffering from the active disease.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree