Fluorescein Angiography
Gisela Barcat Angelelli
Eric P. Jablon
Richard B. Rosen
D. Virgil Alfaro III
INTRODUCTION
In 1871, the sodium fluorescein molecule was synthesized by Baeyer and used as an early ophthalmic research tool (1). Ninety years later, Novotny and Alvis introduced the concept of serial fundus photography after intravenous injection of sodium fluorescein dye to study the retinal and choroidal circulation. This was the first human fluorescein angiogram, what they called “a method of photographing fluorescein circulating in blood vessels (2).”
Their initial observations were of diabetic and hypertensive patients, but it was not long before this novel technique was used to study age-related macular degeneration (AMD). Currently, fluorescein angiography (FA) is the gold standard for the differential diagnosis of neovascular AMD and determination of lesion characteristics (3).
This chapter focuses on the application of the powerful tool of FA for AMD, the classification system that has developed as a result of detailed observations, and the findings of clinical trials, which have used FA in directing treatment and retreatment of choroidal neovascularization (CNV).
FA PRINCIPLES AND TECHNIQUES
FA represents an application of the physical phenomenon of luminescence. Luminescence is the emission of light in the visible spectrum from any source that has been stimulated by electromagnetic radiation. It occurs when energy in the form of electromagnetic radiation is absorbed at a shorter wavelength, shifted, and reemitted at a longer visible wavelength. The process involves decay from a wavelength of higher energy to a wavelength at a lower energy state, causing free electrons in an excited state to emit energy in the form of light when returning to a lower state.
Fluorescence is the luminescence maintained in response to continuous excitation; therefore, fluorescence is present only if the excitation is present. Thus, retinal angiography is possible because of these chemical and physical properties of sodium fluorescein.
Sodium fluorescein (C20H12O5Na) is a water-soluble hydrocarbon that emits green-yellow light having a wavelength 520 to 530 nm after excitation with blue light of wavelength 465 to 490 nm. In other words, if a blue light is projected as an exciting light from a camera into the eye, the sodium fluorescein circulating inside the eye will absorb and discharge a green-yellow reemission, or responder light, that will come toward the camera. Because these frequencies are within the visible spectrum of light, conventional photographic devices and techniques are able to capture angiographic images. This small molecule with a weight of 376.27 Da—compared to 775 Da for indocyanine green (ICG)—readily diffuses through the fenestrated vessels of the choriocapillaris but does not cross the intercellular tight junctions between the retinal pigment epithelium (RPE) and retinal vascular endothelium (RVE). Consequently, any condition that compromises the intact blood-retinal barrier, obstructs blood flow, or changes the normal pigmentation of the retina or pigment epithelium can cause abnormalities on angiography allowing the physiology of the fundus to be studied (4).
Maximal fluorescence of sodium fluorescein occurs at a pH of 7.4, ideal for human blood (5). It is relatively inert, making intravenous injection safe with minimal adverse reactions (6). When mixed with human blood, fluorescein
becomes 80% bound to plasma proteins (compared to near-total binding of ICG to serum globulins), while 20% is unbound or free. Only this 20% of sodium fluorescein not bound to protein will fluoresce.
becomes 80% bound to plasma proteins (compared to near-total binding of ICG to serum globulins), while 20% is unbound or free. Only this 20% of sodium fluorescein not bound to protein will fluoresce.
Before the angiogram, the patient should be informed of the nature and side effects of the study. After signing a consent form, the patient is seated in front of the camera with one forearm extended.
First, stereo color and red-free photographs are taken of the macula. Then, 5 to 10 mL of fluorescein dye is injected intravenously.
There are a few immediate side effects associated with fluorescein injection, particularly nausea and vomiting.
After fluorescein injection, angiophotography commences in 10 to 12 seconds. Immediately after the injection of dye, six rapid-sequence photos are taken of the primary macula during the filling phase (7). This is followed by a stereo pair of the primary macula at approximately 30, 40, 60, 90, and 180 seconds postinjection. Late stereophotos are taken between 5 and 10 minutes postinjection (8).
The first fluorescein angiograms in the late 1960s were made with the classic black body Zeiss fundus camera, equipped with a Contax camera, which advanced the film with a circular turning knob and recycled the photographic flash every 1 to 3 seconds, depending on its intensity (9,10). Novotny and Alvis (2) then modified the Zeiss fundus camera about which they noted in their original article that the main limitation of this prototype was the flash apparatus causing a 12-second delay between photographs. Advances in the original angiography system included modifications of the power pack and flash unit, addition of dual camera backs for ease in performing color fundus photography and FA, use of a motor drive for rapid and motionless film advancement, and a stereo separator for stereophotography (7).
Since their introduction in the mid-1980s, digital angiographic systems have gained widespread use in the ophthalmic community and have undergone technical improvements that have advanced their ability to provide high-resolution images (9,10).
The advent of digital imaging allows the physician to determine appropriate patient diagnosis and treatment options immediately in comparison to the patient’s previous imaging studies. This is accomplished using digital overlay techniques that allow for precise evaluation of changes in fundus abnormalities detected on the angiographic studies (9,10).
Technicians are able to make adjustments in gain, focus, and flash intensity on a real time; assess the quality of the study as it is ongoing; and allow for immediate feedback and training with the physician.
Another advantage to digital imaging is in the area of patient education. Digital imaging allows the physician to manipulate and display the angiographic study while explaining diagnostic and treatment options to the patients on a real-time basis (10). Advantages of such systems also include immediate image processing compared to the development time of film and ease in file storage, file transfer, and file incorporation into electronic medical record systems.
A generation of newly created and defined medical retinal specialists was dedicated to acquire, interpret, and archive these images as a guide for treatment, lasers, vitreoretinal surgery, and pharmacotherapeutic advances (10).
In the Macular Photocoagulation Study (MPS), the photographs used for treatment were obtained 72 to 96 hours prior to treatment (8). Based on findings of the Treatment of AMD with Photodynamic Therapy (TAP) study, treatment for subfoveal CNV with verteporfin photodynamic therapy (PDT) should be performed within a week. In grading the FA, the distance of the neovascular lesion to the center of the fovea, the membrane size, and the leakage characteristics are noted using a stereo viewer. For film angiography, a ration of reticule measurement to the camera magnification factor is used to measure the lesion dimensions. Many digital angiography systems contain measurement software for exact measurements and automatic conversion for different camera settings, yet an enlarged early- or mid-frame FA image is invaluable during thermal laser treatment to ensure adequate coverage of the CNV. Utilization of microfilm reader, angiogram projector, or a digital display to guide laser treatment suite offers the best usage of the angiogram study.
The patients should be properly informed of later effects of FA like temporary yellow skin color, fluorescent urine, and photosensitivity for 24 hours, and they should be informed of the rare but potentially serious risks like hives, asthmatic symptoms, and laryngeal edema (6,7,8,9,10,11).
These can usually be managed by intravenous administration of cortisone. Any facility that performs FA should be equipped with a proper resuscitation kit and have in place a care plan and adequately trained personnel in the event of an adverse reaction (7).
FA INTERPRETATION
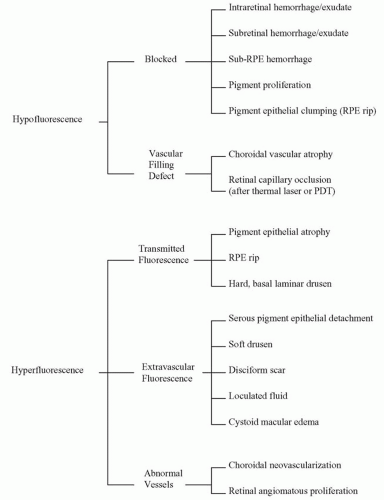
AMD is a retinal disease in which all of the various abnormal FA patterns can be observed (Fig. 10.1). These angiographic abnormalities are broadly classified as those leading to decreased fluorescence (hypofluorescence) or abnormally increased fluorescence (hyperfluorescence) (12). Hypofluorescence represents either blocked fluorescence or a vascular filling defect. Pertaining to AMD, blocked fluorescence is typically due to intraretinal/sub-retinal/subpigment epithelial hemorrhage or pigment proliferation/clumping. The depth of a lesion can be determined by relating the level of the blocked fluorescence to details of the retinal circulation. In advanced non-neovascular AMD, hypofluorescence can develop from choroidal vascular atrophy, and retinal vascular occlusion can occur after thermal laser or verteporfin PDT.
Hyperfluorescence in AMD can be the result of loss of the normal barrier to background choroidal fluorescence known as transmitted fluorescence. Examples include hard drusen, nongeographic atrophy (non-GA) of the RPE in which abnormal background fluorescence fades during the course of the study (window defect), and geographic atrophy (GA) of the RPE in which atrophy of the choriocapillaris reveals the staining of the underlying sclera (13). Leakage of dye into a confined space is characterized by progressive, uniform hyperfluorescence known as pooling, which can be seen with soft drusen and serous RPE detachment. Abnormal blood vessels are noteworthy for their lack of intercellular tight junctions allowing permeability to fluorescein. CNV and intraretinal neovascularization (IRN) in AMD lead to early and progressive hyperfluorescence with late leakage.
INDICATIONS FOR FA IN AMD
While FA is an integral part in the care process of AMD, it does not replace history and careful ophthalmic examination in assessing a patient, and it is not required for all patients or at each visit (9). FA is indicated for any patient with AMD and vision loss, metamorphopsia, or new scotoma in which CNV is suspected (14,15). Because not all patients with neovascular AMD are symptomatic, those at high risk for developing CNV should be carefully examined for signs of such changes (16). While stereoscopic slit lamp ophthalmoscopy can usually detect evidence of CNV (including subretinal fluid, hard exudate, blood, pigment epithelial elevation, or a gray-green membrane), angiography is needed to detect the size, exact location, and leakage
characteristics of the lesion (17). CNV may be undetectable by clinical examination alone. In patients with clinical signs of advanced nonneovascular AMD, FA may be helpful in assessing progression of pigment epithelial atrophy, particularly if vision changes are reported.
characteristics of the lesion (17). CNV may be undetectable by clinical examination alone. In patients with clinical signs of advanced nonneovascular AMD, FA may be helpful in assessing progression of pigment epithelial atrophy, particularly if vision changes are reported.
Because of the increasing incidence of AMD with age, elderly patients with media opacity that may limit careful macular examination, such as cataracts or keratopathy, may benefit from FA (13). Angiography may reveal neovascular or nonneovascular changes that might alter the treatment recommendations or preoperative counseling regarding cataract extraction or corneal transplantation.
FA is a crucial part of the postoperative assessment in patients with CNV who have undergone thermal laser photocoagulation or verteporfin PDT because of the greater sensitivity in detecting CNV, revealing a significant percentage of recurrent lesions not suspected on clinical examination (18). Based on the MPS, for treatment of classic CNV including well-demarcated boundaries with thermal laser, the initial postoperative FA is indicated between 2 and 4 weeks to confirm that the entire CNV lesion has been treated and is obliterated. If adequate treatment is present, repeat FA should be performed in 4 to 6 weeks followed by intervals at the discretion of the treating physician (9). According to the TAP and Verteporfin in Photodynamic Therapy (VIP) studies following verteporfin PDT, the evidence suggests repeat FA should be performed at 3-month intervals with retreatment as indicated (19). Like the MPS, TAP, and VIP, similar angiography protocols should be used, including early-, mid-, and latephase 30-degree film stereophotos centered on the macula after rapid (less than 6 seconds) injection of 5 mL of 10% of sodium fluorescein solution (10).
Over the past two decades, a great deal of effort and expense has gone into the study of new treatments for neovascular AMD. These studies could not be carried out without the ability of FA to objectively document treatment response. Ongoing trials with injected antiangiogenic drugs, non-verteporfin PDT, prophylactic laser to high-risk nonneovascular AMD, transpupillary thermotherapy, radiation therapy, selective feeder vessel laser therapy, serum apheresis, submacular surgery, and macular translocation all include FA in their pretreatment and posttreatment protocol. Currently, there are insufficient data to direct the use of FA after these unproven treatments (9).
ANGIOGRAPHIC PATTERNS IN AMD
Nonneovascular AMD
The majority of patients with AMD have the nonneovascular form, which consists of drusen and RPE abnormalities. Several types of drusen exist that differ histopathologically and angiographically. Hard drusen are small (63 µm), round discrete deposits on ophthalmoscopy that correspond to lipidized RPE or accumulation of hyaline material in the inner and outer collagenous zones of the Bruch’s membrane (20). In FA, hard drusen typically appear as transmission defects due to overlying RPE thinning or depigmentation (21). Angiography often reveals a greater number of hard drusen than can be seen clinically (22).
Soft drusen are larger (greater than 63 µm) with irregular, poorly defined borders and the propensity to coalesce and become confluent. FA of soft drusen shows progressive hyperfluorescence and dye pooling without leakage beyond its margin (Fig. 10.2). Histopathologically, soft drusen are localized detachments between the RPE and (a) basal laminar deposit in an eye with diffuse basal laminar deposit, (b) basal linear deposit in an eye with diffuse basal linear deposit, or (c) localized accumulation of basal linear deposit in an eye without diffuse basal linear deposit (20




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree