Chapter 1 Fluorescein Angiography
Basic Principles and Interpretation
![]() For additional online content visit http://www.expertconsult.com
For additional online content visit http://www.expertconsult.com
For nearly 50 years, fundus photography and fluorescein angiography have been valuable in expanding our knowledge of the anatomy, pathology, and pathophysiology of the retina and choroid.1 Initially, fluorescein angiography was used primarily as a laboratory and clinical research tool; only later was it used for the diagnosis of fundus diseases.1–5 An understanding of fluorescein angiography and the ability to interpret fluorescein angiograms are essential to accurately evaluate, diagnose, and treat patients with retinal vascular and macular disease.
Basic principles
Fluorescence
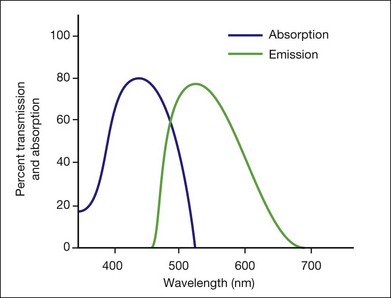
This is a fundamental principle of fluorescein angiography. In the procedure, the patient, whose eyes have been dilated, is seated behind the fundus camera, on which a blue filter has been placed in front of the flash. Fluorescein is then injected intravenously. Eighty percent of the fluorescein becomes bound to protein and is not available for fluorescence, but 20% remains free in the bloodstream and is available for fluorescence. The blue flash of the fundus camera excites the unbound fluorescein within the blood vessels or the fluorescein that has leaked out of the blood vessels. The blue filter shields out (reflects or absorbs) all other light and allows through only the blue excitation light. The blue light then changes those structures in the eye containing fluorescein to green-yellow light at 520–530 nm. In addition, blue light is reflected off the fundus structures that do not contain fluorescein. The blue reflected light and the green-yellow fluorescent light are directed back toward the film of the fundus camera. Just in front of the film a filter is placed that allows the green-yellow fluorescent light through but keeps out the blue reflected light. Therefore the only light that penetrates the filter is true fluorescent light (Fig. 1.1).
Pseudofluorescence
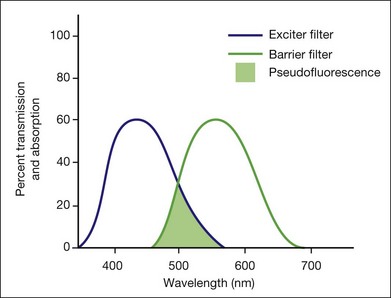
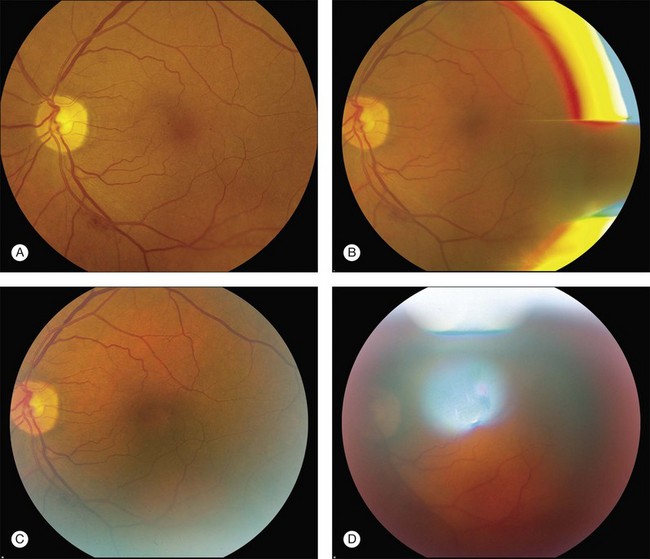
Pseudofluorescence occurs when nonfluorescent light passes through the entire filter system. If green-yellow light penetrates the original blue filter, it will pass through the entire system. If blue light reflected from nonfluorescent fundus structures penetrates the green-yellow filter, pseudofluorescence occurs (Fig. 1.2). Pseudofluorescence (i.e., fake fluorescence) causes nonfluorescent structures to appear fluorescent. It can confuse the physician interpreting the fluorescein angiogram and lead him or her to think that certain fundus structures or materials are fluorescing when they are not. Pseudofluorescence also causes decreased contrast, as well as decreased resolution. Because fluorescein angiography uses black-and-white film, the nonfluorescent or pseudofluorescent light appears as a background illumination. The background illumination from pseudofluorescence is especially heightened if there are white areas of the fundus, such as highly reflective, hard exudates. Pseudofluorescence must be avoided. Therefore the excitation (blue) and barrier (green-yellow) filters should be carefully matched so that the overlap of light between them is minimal.
Equipment
Film-based versus digital fluorescein angiography – historical perspectives
Fluorescein angiography finds its origins in the late 1960s with the publication of an original article describing its use as well as subsequent atlases and textbooks for a medical retinal specialty in its infancy.1,6 The landmark text Atlas of Macular Diseases by Dr J. Donald Gass set a new standard for the use of stereoscopic fluorescein angiography in fundus diagnosis.7
As digital photography has evolved with improved resolution, the convenience of digital-based fluorescein angiography has gained wider acceptance. Though film-based images offer the highest amounts of resolution and 35-mm negatives are often easier to view for stereo, images are relatively difficult to manipulate, and training and effort are required to process and duplicate film. Transmitting or sharing film-based images is also time-consuming compared with digital images (Box 1.1).8
Box 1.1
Equipment and materials needed for angiography
Fundus camera and auxiliary equipment
Matched fluorescein filters (barrier and exciter)
Digital photoprocessing unit (computer-based) and software user interface
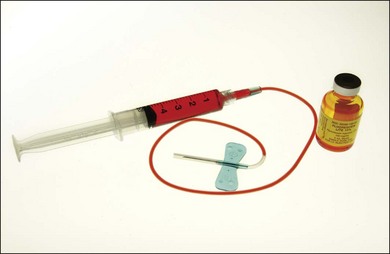
5 mL of l0% fluorescein solution
20-gauge, 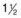 -inch needle to draw the dye
-inch needle to draw the dye
Armrest for fluorescein injection
Camera and auxiliary equipment
Cameras differ in the degree of fundus area included in the photographs. Fundus cameras may range from 35° to 200° wide-field camera systems such as Optos.9,10 In clinical retinal practice, cameras ranging from 35° to 50° are routinely used (Fig. 1.3). Regardless of range, a camera with the ability to yield high resolutions of the posterior pole is essential for most macular problems, especially when laser treatment is to be done, as with background diabetic retinopathy, branch-vein occlusion, or choroidal neovascularization.
Wide-angle angiography has the benefit of capturing a single image of the retina in high resolution well beyond the equator. The potential for clinical efficiency and sensitivity in detecting neovascularization in the far periphery as well as acquiring an excellent clinical picture of the degree of capillary retinal nonperfusion is an exciting development in fluorescein angiography (Fig. 1.4).
Fluorescein solution
Various side-effects and complications can occur with fluorescein injection (Box 1.2).11–15
Box 1.2
Side-effects and complications of fluorescein injection
Extravasation and local-tissue necrosis13
Inadvertent arterial injection
Vasovagal reaction (circulatory shock, myocardial infarction)12
Neurologic problems (tonic-clonic seizures)
Vomiting occurs infrequently, affecting only 0.3–0.4% of patients.11,13 When it does occur, it usually begins 40–50 seconds after injection. By this time most of the initial-transit photographs of the angiogram will have been taken. A receptacle and tissues should be available in case vomiting does occur. When patients experience nausea or vomiting, they must be reassured that the unpleasant and uncomfortable feeling will subside rapidly. Photographs can be taken after the vomiting episode has passed. A slower, more gradual injection may help to prevent vomiting.
Technique
Aligning camera and photographing
The photographer moves the camera from side to side to ascertain the width of the pupil and the focusing peculiarities of the particular cornea and lens. The photographer studies the eye through the camera lens, moving the camera back and forth and up and down, looking for fundus details (e.g., retinal blood vessels). The photographer then determines the single best position from which to photograph (Figs 1.6 and 1.7).
Any abnormalities, such as an unusual light reflex or a poorly resolved image the photographer sees through the camera system, will appear on the photograph. If the ophthalmoscopic view seen through the camera is not optimal, the photograph will not be optimal (Fig. 1.8). If the view is optimal, well aligned, in focus, and without reflexes, the photograph can be optimal. A helpful concept for the photographer is “what you see is what you get (or worse – never better).”
Focusing
The photographer first turns the eyepiece counterclockwise (toward the plus, or hyperopic, range) to relax his or her own accommodation; this causes the crosshairs to blur. The photographer then turns the eyepiece slowly clockwise to bring the crosshairs into sharp focus. The eyepiece is focused properly when the crosshairs appear sharp and clear (Fig. 1.9, online). They must remain perfectly clear while the photographer focuses on the fundus with the camera’s focusing detail. With experience, the photographer becomes expert in adjusting the eyepiece and in keeping the crosshairs in focus throughout the entire photographic sequence.
Digital angiography
In theory, film-based photography has advantages over digital imaging: image resolution and stereoscopic viewing.8 Film-based images contain 10 000 lines of resolution, in contrast to digital imaging, which may have as little as 1000 lines of resolution.8 However, some argue that, despite higher resolution in film, the greater ability to magnify digital images makes the disadvantage of digital photography less clinically relevant.8 Digital angiography offers advantages, including the instantaneous availability of the angiogram, and the avoidance of the equipment and time necessary to develop film. With instantaneous images, digital angiography facilitates education and discussion concerning the patient’s condition and treatment options. Also, digital angiography facilitates training of ophthalmic personnel. We have found that it is useful to stay in the room during the initial frames of the angiogram to ensure that the desired pathology is photographed. Any changes can be promptly made, and the photographer can also learn from this prompt feedback. Digital angiography, however, necessitates an ongoing investment of money both in software updates and storage of digital electronic files. Also, excessive image manipulation with image-editing software may result in artifacts. Specifically, some areas may appear overly hyperfluorescent due to limited dynamic range in images and software manipulation. Care should be taken in avoiding misinterpretation of hyperfluorecence and hypofluorescence in digital images.
Using stereophotography
Stereophotography separates, photographically, the tissues of the eye for the observer. Stereo fluorescein angiography facilitates interpretation by separating in depth the retinal and choroidal circulation.16,17 Stereo angiography is considered absolutely essential in certain situations.18 The photographic protocol for the Macular Photocoagulation Study required stereo fluorescein angiography. Without well-resolved stereo images, interpretation of angiograms with, for instance, choroidal neovascularization associated with age-related macular degeneration, can be extremely difficult, if not impossible (Fig. 1.10, online). On the other hand, stereophotography, although extremely helpful in cases that are difficult to interpret, is not always absolutely necessary because other fundus features and characteristics usually indicate the level at which abnormal fluorescence is located.
Photographing the periphery
Photographing the peripheral retina with a standard 50° fundus camera demands precision and skills acquired only after many hours of practice. Problems with patient position and camera alignment and focus are compounded by marginal corneal astigmatism, unsteadiness of patient fixation, light reflexes, and awkward camera placement. All steps necessary for taking posterior photographs, such as alignment and focusing, must be employed to achieve good peripheral fundus photography. The Zeiss camera comes with an astigmatic dial to help neutralize the induced astigmatism. A tilt mechanism, now standard on most cameras, helps position the camera for extreme superior and inferior peripheral photography (Fig. 1.11).
Positioning the patient
The patient is positioned at the camera with the chin in the chinrest and the forehead against the head bar. Because the most common cause of poor fluorescein photographs is involuntary movement of the patient’s head, the photographer should prepare and make adjustment for this before the fluorescein is injected. The photographer should aim and focus the camera on the specific area of primary interest, at the same time noting the patient’s responses. If the photographer finds that the camera must continually be moved closer to the patient while aligning it or taking preliminary photographs, or if reflexes suddenly appear in the view even though the camera is steady, then the patient’s head has moved away from the chinrest. If so, the photographer can make some adjustment before injecting the fluorescein dye. Sometimes having an assistant hold the patient’s head in the chinrest is helpful (Fig. 1.12, online). The photographer either may lower the entire camera and chinrest or raise the patient’s chair. This causes the patient to lean forward in the chinrest and against the forehead bar, making it more difficult for the patient to pull back.
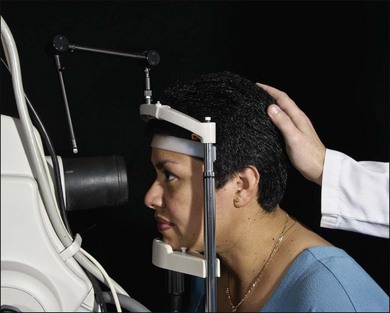
Fig. 1.12, online An assistant holds the patient’s head as a reminder to the patient to keep the chin in the chinrest and forehead against the bar.
Injecting the fluorescein
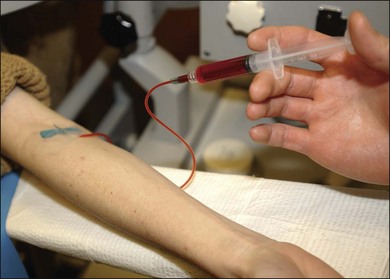
The color stereoscopic fundus photographs are taken first, before the fluorescein is injected. For injection, we recommend a syringe with a 23-gauge scalp-vein needle (Fig. 1.13, online). The scalp-vein needle has several advantages: it is small enough to enter most visible veins, and an intravenous opening is then available in the event of an emergency. Once in the vein, it requires no further attention, and although it can be taped in place, this usually is unnecessary. Whenever an antecubital vein is not visible or accessible, the vein in the back of the hand or radial (thumb) side of the wrist can usually be used for injection. Injecting the fluorescein into a hand or wrist vein increases the circulation time by a few seconds, but this seldom makes any difference.
Developing a photographic plan
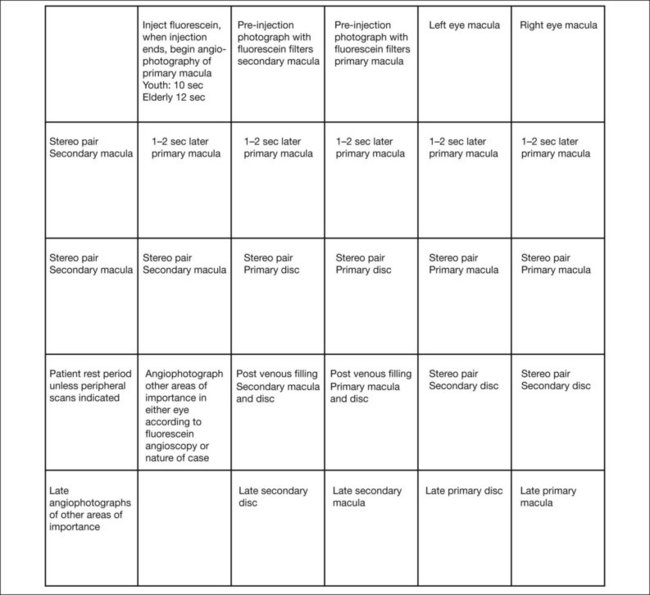
To photograph and print the fluorescein angiogram, we suggest the following comprehensive plan, designed to yield maximal angiographic information from each fundus and to facilitate a thorough and complete interpretation (Fig. 1.14). In contrast to film-based angiograms which required multiple duplicate attempts to assure at least one image would be optimal, fewer photographs are necessary in digital angiography. Although most angiograms will be complete by following this procedure, there will be exceptions. This plan must be modified if abnormalities occur in areas other than the macula and disc.
It is both essential and extremely cost-effective for the physician to indicate specifically what areas to photograph. The photographer should be directed as to where to start the angiogram and the issues important for each specific angiogram. It is most efficient to use a photographic instruction slip that indicates the specific number of color photographs to take of each area, where to start the angiogram, what the diagnosis is, and any other information about the patient or fundus that is pertinent to the photographic process (Fig. 1.15, online). Although digital color and angiograms avoid the issue of wasted film and developing costs, unnecessary computer storage of images and patient inconvenience can be avoided with good technique and a repeatable, accurate algorithm for angiography.
At this point the fluorescein injection is begun. The needle is inserted in a vein in the patient’s arm (Fig. 1.16, online). The photographer waits for confirmation of successful venous access and awaits verbal confirmation that infusion is about to begin. Once the injecting clinician starts the infusion of fluorescein, the photographer begins the initial “injection” image. When the injecting clinician has completed infusion, he or she announces “injection complete” and the photographer takes the “end-of-injection” image. Because it is important to observe the site of the needle tip for extravasation of fluorescein, the lights are turned off only at the end of the injection. An alternative method is to turn the lights off after the needle has been inserted in the vein. The person injecting can hold a hand light to observe the fluorescein flow into the vein to be sure extravasation is not occurring. With the lights off, the photographer can become dark-adapted, which allows him or her to be better able to see the flow of fluorescein into the fundus as it occurs.
Box 1.3 provides a checklist of important steps in the fluorescein angiography procedure.
Box 1.3
Checklist for fluorescein angiography
• Inform patient about fluorescein angiography. Obtain written informed consent
• Dilate patient’s pupil if necessary
• Prepare fluorescein solution, scalp-vein needle, and syringe
• Input patient identification and demographic data in computer database
• Position patient for alignment, focus, and comfort
• Take red-free photos if indicated
• Simultaneously start timer from zero and inject fluorescein dye
• Take preinjection photograph: these serve as controls in detecting auto- and pseudofluorescence
• Shoot at exact start of injection as timer is turned on and shoot second shot at exact finish of injection (length of time of injection is automatically recorded)
• Start fluorescein photograph 8 seconds after the start of injection in young patients and 12 seconds in older patients
• Follow fluorescein angiography plan
• When photography is done, reassure patient that urine will be discolored for a day or so
• At the discretion of the physician, have patient wait for observation for possible reactions to fluorescein
Interpretation
Fundus anatomy and histology
1. preretinal area, where contraction from an epiretinal membrane may influence the retinal circulation and where hemorrhage may be located
2. vascular layers of the sensory retina, both superficial and deep
3. avascular portion of the sensory retina, particularly the outer plexiform layer, the principal site of intraretinal edema and exudate
4. retinal pigment epithelium, which has the potential for many manifestations, including proliferation, depigmentation, hyperpigmentation, and detachment
5. choroidal circulation, including the choriocapillaris and the large choroidal vessels
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree