Fluorescein and Indocyanine Green Angiography
Steven R. Sanislo
Mark S. Blumenkranz
Thierry C. Verstraeten
In many instances, angiography can serve as a useful guide to the clinician in the diagnosis and management of infectious and inflammatory diseases of the retina and choroid. Sodium fluorescein is the most common angiographic dye used for this purpose, but indocyanine green (ICG) dye is also useful in certain cases. Because angiography yields a high-contrast image for the permanent medical record, the potential exists for overuse of this modality, when more simple and inexpensive methods will suffice. In instances where clinical history and ophthalmoscopy alone are not sufficient to either establish a working diagnosis or develop and monitor a treatment plan, fluorescein or ICG angiography in conjunction with other serologic studies may play an important role. Because of restrictions on the length of this chapter, as well as the availability of excellent reviews on specific diseases, both in other portions of this series as well as elsewhere,1,2,3,4 an encyclopedic listing of the angiographic features of all major inflammatory and infectious conditions is not provided. Our intent is to establish basic guidelines for the indications and interpretation of fluorescein and ICG angiography in this broad category of diseases.
PRINCIPLES OF FLUORESCEIN
ANGIOGRAPHY
The rationale for the use of sodium fluorescein for diagnostic imaging of the fundus is covered more completely in other chapters. By absorbing photic energy in the wavelength range of 465 to 490 nm and emitting at a different wavelength range of 520 to 530 nm, sodium fluorescein yields a dynamic high-contrast image of the retinal structures, emphasizing the discrete intravascular compartments silhouetted against their adjacent structures, when photographed with appropriate barrier and excitation filters in place. In some instances, the presence of the monochromatic filters alone is sufficient to decrease light scattering and consequently improve resolution of fine detail in eyes with hazy intraocular media. In most instances, however, the luminescence provided by the excited fluorescein molecule is responsible for the apparent image enhancement available by this technique. With the recent availability of more sophisticated video and digital signal processing techniques, the capacity for image enhancement has been further increased. Because of its relatively low molecular weight of 376 Daltons, sodium fluorescein freely traverses the fenestrated capillaries and larger vessels comprising the choroid. However, the tight junctions between adjacent retinal pigment epithelial cells and healthy mature retinal capillary endothelial cells effectively block the ingress of fluorescein from the choroid to the subretinal space, or from the retinal vessels into the neurosensory retina or vitreous, respectively, under normal conditions. The loss of this selective barrier effect as a result of disease underlies the value of fluorescein angiography as an imaging modality. In addition to ischemia, mechanical or thermal trauma and inflammation result in breakdown of the barrier effect of tight junctions and allow passage of fluorescein, generally in proportion to the degree of the inflammation.1,2,3,4 Because the inflammation may be either generalized or localized to a specific region of the chorioretinal complex, depending on the nature of the disease process, the fluorescein angiogram may yield valuable information not only with regard to severity of the breakdown of the barrier such as fluorophotometry but also with regard to localization of the site of inflammation. In instances in which inflammation coexists with ischemia or neovascularization, fluorescein angiography can be particularly valuable in distinguishing between these elements. Additionally, fluorescein angiography can show alterations to the retinal pigment epithelium during acute inflammatory phases and also show chronic changes in the pigment epithelium that often remain after the inflammation has subsided.
PRINCIPLES OF INDOCYANINE GREEN (ICG)
ANGIOGRAPHY
Indocyanine green angiography differs from fluorescein angiography in several ways. Indocyanine green dye is more highly bound to protein than sodium fluorescein and has peak absorption and emission wavelengths in the infrared spectrum, outside of the visible range. Because the fluorescence efficiency of the ICG molecule is only 4% of that of sodium fluorescein, photographic film is too insensitive to record this weak fluorescence. To detect ICG fluorescence, charge coupled device (CCD) cameras with a digital acquisition process are necessary. Indocyanine green angiography can image the choroidal vasculature better than fluorescein angiography because the larger and more highly protein bound ICG molecule is unable to diffuse out of the fenestrated choroidal vessels. In addition, the infrared emission spectrum of ICG dye is not blocked by the overlying retinal pigment epithelium. Thus ICG angiography is an excellent technique for evaluating choroidal hypoperfusion or choroidal leakage.
Indocyanine green angiography can be helpful in the diagnosis and management of various infectious and inflammatory diseases. ICG patterns of hypofluorescence and hyperfluorescence have been described for many inflammatory diseases including: multiple evanescent white dot syndrome (MEWDS),5,6,7 acute multifocal placoid pigment epitheliopathy (AMPPE),8 multi-focal choroiditis,9 Vogt-Koyanagi-Harada disease (VKH),10,11,12 ocular sarcoidosis,13 and Behcet disease.14,15 ICG angiography often shows discreet or patchy areas of hypofluorescence in the active inflammatory phases of these diseases indicative of choroidal alterations, often much more extensive than is clinically apparent with ophthalmoscopy or fluorescein angiography. In some of these disorders such as MEWDS the ICG, abnormalities resolve completely with resolution of the patient’s symptoms, whereas in others ICG abnormalities may persist. Hyperfluorescence may also be seen with ICG angiography, as in VKH disease where diffuse choroidal leakage can be prominent in late phases of the study. By better-visualizing the condition of the choroidal circulation, ICG angiography is helping us to better-understand the pathophysiology and possible mechanisms involved in infectious and inflammatory diseases of the retina and choroid. Currently, the clinical usefulness of ICG angiography for infectious and inflammatory diseases remains somewhat limited. However, with increasing familiarity of the angiographic findings in these diseases, ICG angiography is likely to play a greater role in their diagnosis and management.
METHOD FOR ASSESSING MECHANISM OF VISUAL LOSS AND RESPONSE TO TREATMENT
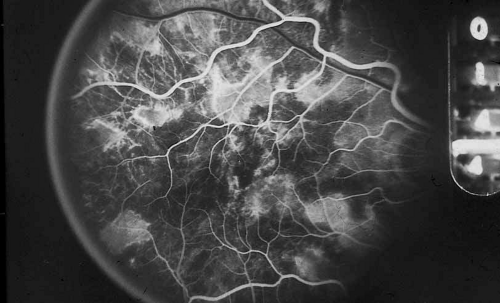
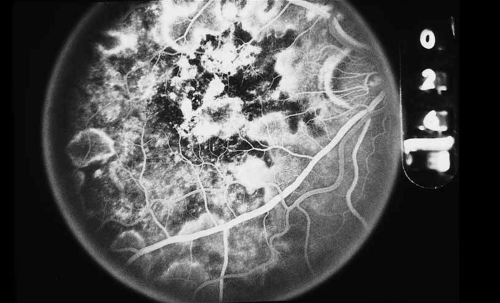
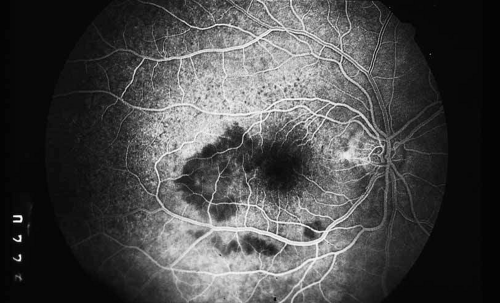
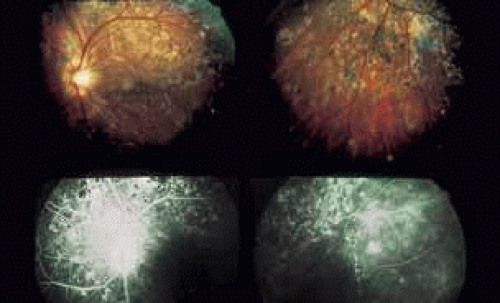
As indicated earlier, in the large majority of cases, the combination of the clinical history and ocular examination including binocular slit lamp biomicroscopy are generally sufficient to establish a working diagnosis and formulate a treatment plan. In selected instances, including the peculiar leopard spot appearance of luetic neuroretinitis16,17,18,19,20,21 or the classic brush-fire appearance of serpiginous choroiditis (see Figs. 1, 2, and 3),22,23,24,25,26,27,28,29,30 the diagnosis may be established on the basis of the angiographic findings alone. More often, the angiography is used to answer specific questions about the individual case, relating either to the mechanism of visual loss or to the measurement of the response to treatment. The following is a partial list of complications of inflammations and infections of the posterior segment that result in visual loss that can be better defined by angiography:
Optic neuropathy
Cystoid macular edema
Retinal vascular occlusion
Retinal (macular) capillary nonperfusion
Retinal pigment epithelial detachment
Retinal pigment epithelial dysfunction
Retinal neurosensory detachment
Choroidal neovascularization
Choroidal vascular occlusion
Neovascular glaucoma
Vitreous hemorrhage
As an example, patients with posterior segment involvement by the spirochete Treponema pallidum may manifest visual loss on the basis of a large number of different mechanisms.16,17,18,19,20,21 These include perioptic neuritis, retinal periphlebitis or periarteritis, diffuse neuroretinitis, cystoid macular edema, or choroidal neovascularization. Fluorescein angiography may be helpful not only in establishing the causative agent but also in determining the predominant mechanism of visual loss (see Figs. 4 and 5).
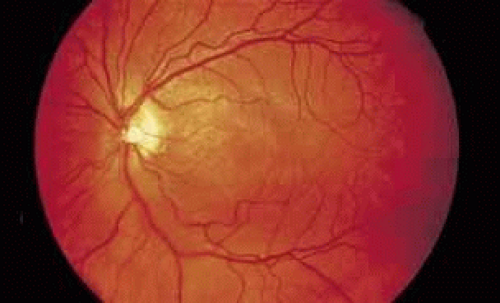 Fig. 4 Luetic neuroretinitis. Color photograph showing placoid opacity at the level of the retinal pigment epithelium. |
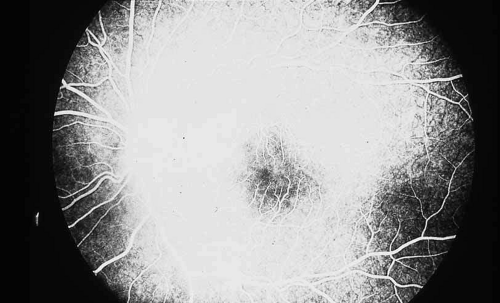 Fig. 5 Luetic neuroretinitis. A late frame of the angiogram in Figure 4 shows extensive staining of the entire area corresponding to leakage into the outer retina and staining of the pigment epithelium. The patient was successfully treated with intravenous penicillin therapy and subsequently developed diffuse “leopard spot” changes in the pigment epithelium characteristic of resolved luetic neuroretinitis. |
QUANTITATION OF DISEASE SEVERITY
MACULAR INVOLVEMENT
One of the most useful applications of fluorescein angiography in infections and inflammations of the posterior segment is the delineation and quantization of cystoid macular edema. Although it is generally possible to ascertain the presence or absence of gross cystoid macular edema in eyes with clear media, this is often not the case in eyes with intraocular inflammation. The presence of intraocular inflammation, such as the vitreous cells accompanying pars planitis31,32,33 or sarcoidosis,34,35,36,37,38,39,40 may prevent clear viewing of the macula. The tell-tale hyperfluorescence seen in the normally hypofluorescent macula after dye injection is an important indicator. The presence of cystoid macular edema by angiography may lead the clinician to take a more aggressive posture in the treatment of these diseases, especially in younger patients in whom some uncertainty exists regarding the relative contribution of the media opacity versus the macular function to the reduced vision (see Figs. 6 and 7).
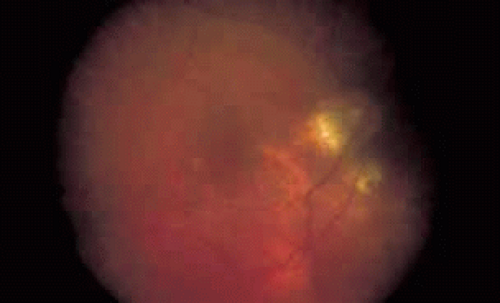 Fig. 6 Presumed ocular histoplasmosis. This patient developed decreased vision with mild vitritis. Color photograph shows a hazy view of the central macula. |
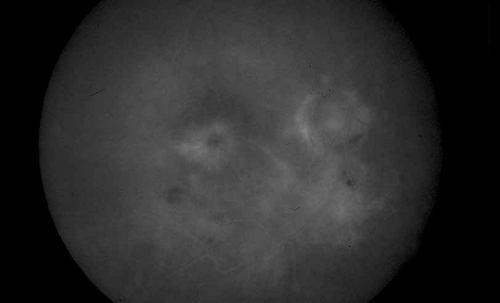 Fig. 7 Presumed ocular histoplasmosis. Fluorescein angiogram of Figure 6 demonstrates cystoid macular edema as the cause of this patient’s decreased vision. |
Once a treatment regimen has been instituted, the follow-up fluorescein angiogram can serve as a marker for treatment response in addition to other clinical variables, including visual acuity and severity of intraocular inflammation (see Fig. 8).
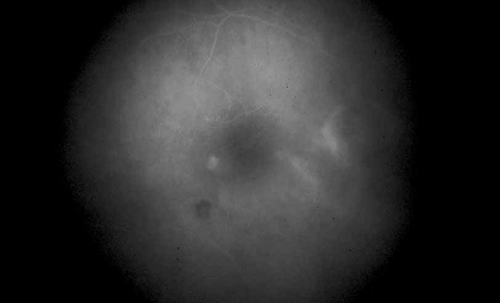 Fig. 8 Presumed ocular histoplasmosis. Repeat fluorescein angiogram from Figure 7 shows complete resolution of cystoid macular edema after intravitreal injection of triamcinolone–acetamide. |
OPTIC NERVE DISEASE
The fluorescein angiogram can provide useful information with regard to inflammatory involvement of the optic nerve in association with retinal inflammation either in eyes with clear or opaque media. In eyes with clear media, excessive staining of the optic nerve in the late frames of the angiogram may signify sub clinical inflammation, even in the absence of any obvious disc swelling or engorgement. Patients with Vogt-Koyanagi-Harada disease,41 sympathetic ophthalmia42,43,44,45,46 and luetic neuroretinitis frequently have occult inflammation of the optic nerve, which may signify the clinical stage of the intraocular inflammation more accurately than the retinal findings, particularly in long-standing cases with extensive atrophic retinal abnormalities in which it is difficult to monitor change (see Fig. 9).
Another circumstance in which fluorescein angiography is helpful in defining the cause and extent of inflammatory disease is in the context of a patient who has a coexisting disease process. Patients with diabetes mellitus who are found to have cystoid macular edema after cataract extraction represent a difficult management problem. Although diffuse leakage may occur from the perifoveal capillary net, in response to hyperglycemia or low-grade intraocular inflammation such as that which follows cataract surgery, the presence of late staining of the optic nerve in a fluorescein angiogram in such a patient suggests that at least a portion of the edema may be related to postsurgical inflammation.
A third instance in which fluorescein angiography concentrating on the optic nerve may be helpful is in eyes with vascular engorgement of the optic nerve, especially with media opacities. Fluorescein angiography may help the clinician distinguish abnormally leaking capillaries from neovascularization. In acute multifocal hemorrhagic retinal vasculitis, optic nerve involvement is common in the early stages of the disease,47 with optic nerve disc neovascularization seen later (see Figs. 10, 11, 12, and 13). Another fluorescein angiographic clue to the distinction between optic disc vessel engorgement and true neovascularization is the presence of associated large zones of retinal capillary nonperfusion such as those seen in sarcoidosis (see Figs. 14 and 15),34,35,36,37,38,39,40 acute multifocal hemorrhagic retinal vasculitis (see Figs. 10 and 11),47 or Eale disease (see Figs. 16 and 17).48,49,50,51,52
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree