FIGURE 43.1 The arborizing VII cranial nerve acts as the “electrical wiring” of the face.
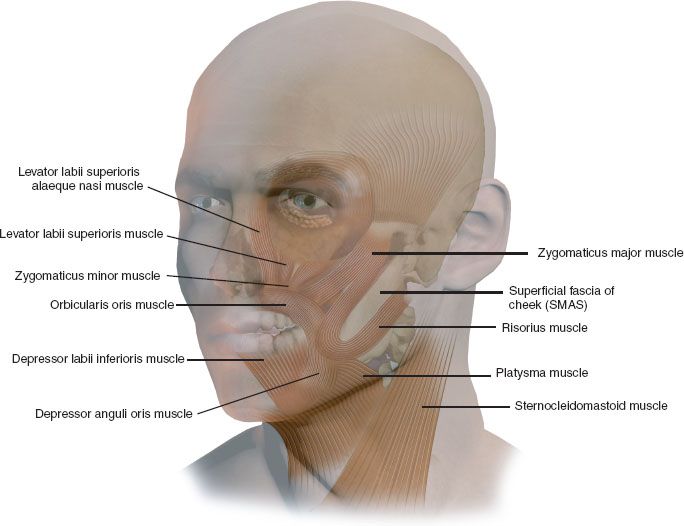
FIGURE 43.2 A facial nerve deficit creates generalized facial laxity related to atonicity of the deep foundation of the face, upper lip immobility, related to weakness of the lip elevators, zygomaticus major and minor muscles, and an inadequate smile “spread” related to weakness of the risorius muscle. Additionally, oral incompetence (pursing, blowing, whistling, intraoral seal) related to weakness of the orbicularis oris sphincter, lower lip flaccidity related to paralysis of the depressor labii muscle, and inspiratory collapse of nasal vestibule related to weakness of the levator labii superioris alaeque nasi muscle can occur. The upper face suffers from paralytic ectropion of the eyelids related to weakness of orbicularis oculi muscle and ptosis of the brow related to paralysis of the frontalis muscle.
1. Generalized facial laxity related to atonicity of the deep foundation of the face (mimetic muscles and superficial aponeurotic system)—VII nerve trunk
2. Upper lip immobility (asymmetric smile with “shadowing” of the upper teeth) related to weakness of the lip elevators and zygomaticus major and minor muscles—buccal branch of the facial nerve
3. Inadequate smile “spread” (asymmetric smile) related to weakness of the risorius muscle—buccal branch of facial nerve
4. Oral incompetence (pursing, blowing, whistling, intraoral seal) related to weakness of the orbicularis oris sphincter—buccal branch of facial nerve
5. Lower lip flaccidity (drooling, speech impairment, chewing) related to paralysis of the depressor labii muscle—marginal mandibular branch of facial nerve
6. Inspiratory collapse of nasal vestibule (decreased nasal air flow, congestion) related to weakness of the levator labii superioris alaeque nasi muscle—buccal branch of the facial nerve
7. Paralytic ectropion of the eyelids (tearing, corneal exposure, lagophthalmos) related to weakness of orbicularis oculi muscle—zygomatic (orbital) branch of facial nerve
8. Ptosis of the brow (impaired upward gaze) related to paralysis of the frontalis muscle—frontalis branch of facial nerve
In view of the myriad of functional and structural deficits, successful reanimation of the paralyzed face is a lofty challenge to the reconstructive facial surgeon. The two defining objectives of any successful reanimation surgery are to achieve (1) static facial balance at rest and (2) symmetric, dynamic movement (preferably voluntary and spontaneous) by supplementation or substitution of the paralyzed muscles.
I have delineated methods of static rebalancing in my description of multiple applications of autogenous fascia lata slings in providing deep structural support of the facial foundation and correction of droopiness of the lips, eyelids, and nostrils.
Given the “symphony of motion” of the multiple mimetic muscles of facial expression, no single procedure can supplant the intricacies of motion/vectors needed for “natural” facial movement. The ideal situation, of course, is direct and/or interpositional nerve grafting of a recent facial nerve injury. Frequently, however, those repairs are neither feasible nor temporally suitable, and secondary reconstructive options need to be considered. In cases of partial paralysis or in older adults, regional muscle transfers (temporalis, masseter, platysma, frontalis) are the procedures of choice. These procedures, although very effective, require lengthy neuromuscular retraining and may lack involuntary spontaneity. In younger patients, however, the preference is for facial nerve “specificity” by reinnervation from the contralateral facial nerve (via cross-facial nerve graft) into the distal branches of the injured facial nerve. Or, in long-standing facial paralyses where the target muscles have undergone “denervation atrophy,” a substitute muscle is necessary to reanimate the paralyzed face (free gracilis muscle transfer). The focus of this chapter is on the use of cross-facial nerve grafts and microvascular free gracilis muscle flaps in achieving such dynamic motion.
The contemporary era of dynamic correction of facial palsy emerged with the introduction of the microvascular free transfer of the gracilis muscle by Harii in 1976 based on Tamai’s earlier experimental (1970) and clinical work in the transfer of muscle as a vascularized unit. The initial gracilis transfer was neurotized by the ipsilateral V cranial nerve but, of course, required “triggering” by smiling or clenching the teeth (that concept is now being “recycled” by Manktelow and Klebuc’s work using the branch to the masseter as the motor nerve). Usage of the contralateral normal facial nerve as a source of innervation was conceived by Scaramella (1970) and further refined by Smith and Anderl (1979). Gordon and Buncke showed in rhesus monkeys that heterotopic muscles could function adequately when motored by long interpositional nerve grafts. O’Brien in 1980 synthesized these concepts into an algorithm defining the clinical indications for cross-facial nerve grafting and free microvascular gracilis muscle transfers in long-standing facial paralysis. Other muscles, such as pectoralis minor, serratus anterior, extensor brevis, latissimus dorsi, and palmaris longus, have also been used as recipient muscles in facial reanimation. The gracilis muscle, however, is the most commonly used and, in my opinion, the easiest to work with and most predictable in outcome. The advantage of this approach is the ability to obtain synchronous and spontaneous bilateral facial expression and a more “natural” smile.
HISTORY
A careful history is elicited from the patient (or parents) confirming mode of onset, duration, and improvement (if any) since onset. In infants or toddlers with congenital facial paralysis, parents are questioned regarding problems during the pregnancy (infection, drug exposure, smoking, and eclampsia), positioning of the fetus, family history, and birth trauma (forceps delivery, small pelvic canal). If a hereditary etiology (i.e., CHARGE syndrome) is suspected, a geneticist should evaluate the family or siblings. A description of the timing of the onset (immediate or delayed), early treatment, and neurophysiologic testing should be obtained from the family.
In teens or adult patients, the etiology is usually more clear (intracranial or facial nerve trauma, developmental, acoustic neuroma, brain stem tumor, parotid tumor, Bell palsy, lymphangioma or hemangioma, or iatrogenic). Timing of the onset, early treatment, response to physical therapy or acupuncture, staging of return of facial function (timing of initiation of return and “plateau” of facial movement), prior testing (EMG/NCS or CAT scan), and neurologic, ophthalmologic, and speech evaluations should be elicited from the patient. Prior photos from family events are very helpful in assessing smile and facial expression. Work history and impact on social and professional interactions are helpful in understanding self-esteem issues and coping skills related to the functional disabilities. The final question in the assessment should be “Are you motivated to have your facial paralysis corrected?”
Functional problems associated with an established facial paralysis are fairly predictable (see clinical characteristics above). History related to periocular problems should include queries regarding visual impairment, corneal exposure, immobile brow, excessive tearing, impaired upward gaze, incomplete eye closure, and blepharospasm (synkinesis). Does the patient require taping, eye drops, or night time lubrication? Midface subjective findings include facial atonicity and flaccidity (while puffing cheeks); biting of the inside of the mouth, tongue, and lips; droopiness of the nostrils, cheek, and corner of the lip; difficulty with bolus management (oftentimes needing tongue to sweep food to healthy side); impaired chewing; inadequate oral seal while whistling, blowing, or sucking; and dribbling from the corner of the mouth while cup feeding or drinking through straw. Speech patterns affected are primarily the bilabial sounds—plosives (p’s and b’s) and phonemes (f’s, w’s, v’s, and m’s). Patients may complain of late-day lack of articulation, lower volume, or speech fatigability.
PHYSICAL EXAMINATION
Specific findings on physical examination are dependent on the level and extent of the lesion. In total or near total paralyses, the perioral, midface, and periocular regions are most affected. In milder cases, the patient may appear normal at rest; in more severe cases, droopiness and loss of muscle tone may be apparent (flatness of the nasolabial fold, loss of the nasolabial crease, elongation of the upper lip, sagging of the brow and corner of the eye, “open” eye). With animation, however, the discrepancies are much more visible. While smiling, there is asymmetry between the void and active side with an overpull by the healthy muscles. “Shadowing” of the upper teeth is noted (tooth exposure on the palsied vs. the healthy side is an easy measure of upper lip mobility). “Smile spread” is diminished on the affected side (measured by the distance between the midline tubercle and lateral commissure). The paralyzed lower lip is invaginated/inverted causing “shadowing” of the lower teeth. On inspiration, there is downward displacement of the affected nostril with snarling causing obstruction of the vestibular airway. Puckering and pursing of the lips are impaired, compromising the ability to create an intraoral seal. With attempted lip approximation or puckering, there is usually a gap between the upper and lower lip allowing escape of air or fluids.
In the periocular region, the affected eyebrow is “flattened” having lost is arch and paralleling the supraorbital rim. With upward gaze, there is a 1.0 to 1.5 cm discrepancy between the affected and healthy side. In residual Bell palsy or neuropraxic injuries within the intratemporal canal, intensive blepharospasm (synkinesis) may be “triggered” by the yawning, chewing, or clenching teeth. Extensive tearing (epiphora) may be noted in the affected “tear trough” (particularly when tilting the head) due to inadequate seal of the lower lid to the eyeball. In severe cases, lagophthalmos (gap) is noted between upper and lower lip with voluntary closure. The cornea is usually protected by automatic upward deviation of the globe with incomplete closure (Bell phenomenon). The blink reflex is often absent in lesions at the brain stem level.
INDICATIONS
Facial reanimation by cross-facial nerve grafting and gracilis muscle transfer is primarily an operation for younger patients. Axonal regeneration across an interpositional nerve graft tends to diminish with age and may be inadequate to neurotize the transferred muscle from a distant source. In my opinion, age 35 to 40 is the “cutoff” for cross-facial nerve grafting. Very excellent regional muscle transfers (temporalis and platysma) offer a “one-stage” facial reanimation with evidence of facial movement at 4 to 6 weeks postoperatively (albeit extensive neuromuscular retraining is necessary to “trigger” the transferred muscle). Most adults are not willing to wait the 1½ to 2 years necessary to experience spontaneous facial movement after microsurgical reconstruction utilizing the two-stage procedure. Regional muscle transfers (temporalis and platysma) are also the procedures of choice in the partial paralyses following Bell palsy, acoustic neuroma resection and trauma. In these patients, the risk of jeopardizing existing motion by splicing into intact facial nerve branches on the palsied side does not justify the more extensive microsurgical reconstruction.
CONTRAINDICATIONS
Elderly patients, particularly those with medical comorbidities, may not tolerate the lengthy operative times of the respective two stages and should be considered as candidates for static and/or regional muscle transfers.
PREOPERATIVE PLANNING
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


