Extraocular Muscles
Joseph L. Demer
The extraocular muscles (EOMs) play a vital role in the visual system, providing both static adjustment of binocular alignment necessary to enable stereopsis and also the precise dynamic movements necessary to acquire and maintain visual targets on the fovea despite ubiquitous motion of the head and body. Ocular saccades, rapid refixational movements of the eyes, are the fastest movements made by any part of the body, and maintained foveal fixations are arguably the most precise movements. Every aspect of the structure of the EOMs and their associated connective tissues is specialized for the unique functional requirements of the oculomotor system, and these structural specializations are of clinical significance for ocular motility. The EOMs differ from all other striated muscles in many important respects.
Description of ocular kinematics requires definition of a coordinate system of rotations. A convenient coordinate system is that of Fick, specifying the order of rotations in the following order: (i) horizontal, about a fixed vertical axis; (ii) vertical, about a horizontal axis rotated with the eye; (iii) torsional, about the line of sight that has in turn made the preceding two rotations with the eye. An abducting (external) horizontal rotation moves the line of sight temporally, whereas an adducting (internal) horizontal rotation moves the line of sight nasally. An elevating (sursumduction) vertical rotation moves the line of sight toward the vertex of the head, whereas a depressing (deorsumduction) vertical rotation moves the line of sight toward the feet. An intorting rotation about the line of sight (incycloduction) rotates the superior pole of the eye nasally, whereas an extorting rotation about the line of sight (excycloduction) rotates the superior pole of the eye temporally. It should be noted that rotational kinematics of any three-dimensional (3-D) object such as the eye are not commutative in the mathematical sense.1 The order of rotations of an object is important in determining the resulting orientation, although for most clinical purposes the rotations of normal eyes can be regarded as effectively commutative.
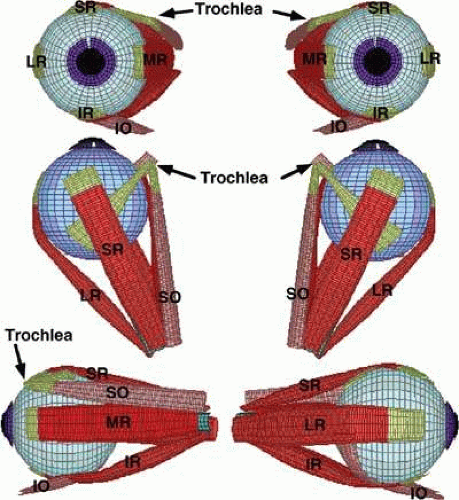
In humans there are six oculorotary muscles, functioning as antagonist pairs. The medial rectus (MR) and lateral rectus (LR) muscles accomplish horizontal eye movements, with the MR accomplishing adduction and the LR accomplishing abduction. The superior rectus (SR) and inferior rectus (IR) muscles act as a vertical antagonist pair, with the SR supraducting and the IR infraducting the globe. However, the vertical rectus EOMs have additional actions that are not strictly antagonistic, as will be detailed below. The superior oblique (SO) and inferior oblique (IO) muscles act as an antagonist pair essential for torsion of the globe around the line of sight (cycloduction). The SO accomplishes incycloduction, whereas the IO accomplishes excycloduction. The SO also acts as an infraductor, mainly in adduction, while its incycloducting action is mainly in abduction. The IO also acts as a supraductor but has horizontal action that depends upon horizontal eye position. Vertical and horizontal actions of the oblique EOMs are thus not strictly antagonistic, as will be detailed below.
Embryology of the Extraocular Muscles
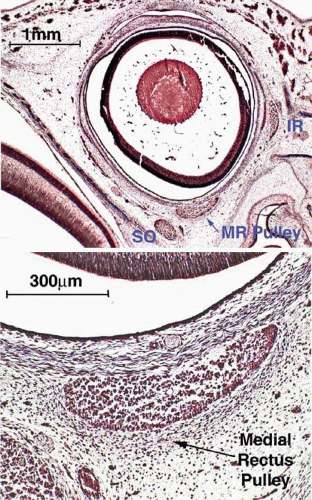
The fibers of the striated EOMs originate from condensations of cranial mesoderm, whereas their associated connective tissues and the orbital smooth muscles originate from the neural crest.2,3 Myogenesis of the striated EOMs occurs in two waves, primary and secondary.4 Even as early as the primary wave of myogenesis at 11 weeks of gestation (Figs. 1.1A and B), the perimuscular connective tissues, such as the trochlea and the soft pulleys of the rectus EOMs, have begun to be associated with the condensations of myoblasts that will form the EOMs (Fig. 1.1A). The periocular connective tissues develop further by the secondary wave of myogenesis. The developing EOMs attract innervation from their corresponding cranial nerves.
Internal Structure of the Striated Extraocular Muscles
The oculorotary EOMs, although not the levator palpebrae superioris (LPS), consist of two distinct layers (5) (Fig. 1.2). The global layer, in humans containing a maximum of approximately 10,000 to 15,000 fibers in the midlength of the EOM, is located adjacent to the globe in rectus EOMs5,6 and in the central core of the oblique EOMs.7 In the rectus EOMs and the SO, the global layer anteriorly becomes contiguous with the principal tendon and inserts on the sclera.6,8,9,10 Rare muscle fibers have been reported to extend within the tendon even to the sclera,11 but these are of dubious functional significance. In the IO, the global layer inserts directly on the sclera without a tendon.13
The orbital layer of each rectus EOM contains 40% to 60% of the total number of fibers in the EOM, but the number varies according to the specific EOM, being greatest for the MR and least for the SR.5 The orbital layer of the rectus and IO EOMs does not insert on the globe at all, but instead inserts on the corresponding connective tissue pulley.6,8,9,10 Orbital layer insertions on pulley tissues are evident in rabbit, cow, dog, and horse.12 Histologic study in rat shows insertion of the IR orbital layer on a pulley.13 Ruskell et al.10 have proposed that orbital layer insertion into connective tissue sleeves may be a general mammalian feature. In all mammals, the orbital layer is located on the orbital surface of all rectus EOMs, sometimes forming a C-shaped configuration, and constitutes the concentric outer layer of the oblique EOMs.7 The separation between orbital and global layers is particularly evident for the inferior rectus (IR) muscle. In animals such as cow, the orbital and global layers of the IR constitute grossly distinct and separable laminae, whereas in humans the less prominent border between the layers is nevertheless typically evident as a bright demarcating line on T1-weighted magnetic resonance imaging (MRI) in the plane parallel to the IR muscle. An intermediate transitional zone between the orbital and global layers of rectus EOMs is sometimes described.
For the global layer of rectus EOMs, the maximum number of fibers is in midorbit, with the number of fibers diminishing anteriorly and posteriorly.5 This suggests that global layer fibers may bifurcate and rejoin in myomyous junctions along the length of each EOM, and is supported by the finding that EOM fibers are substantially shorter than overall EOM length.14 However, the literature on this fundamental question is inconsistent. The putative myomyous junctions were not evident in a study in which individual fibers were traced along the EOM length for multiple different human and monkey rectus EOMs.6 A careful study of serially sectioned human and monkey orbits indicated that the number of orbital layer fibers remains constant along the length of each EOM, suggesting that each global layer fiber extends the entire length of the EOM.5
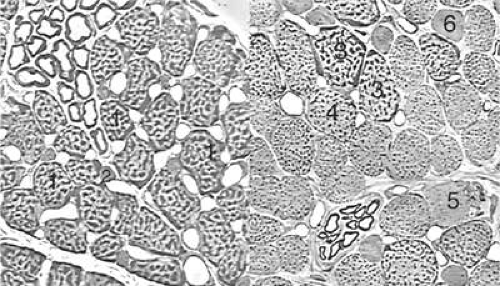
The orbital layer of each EOM contains two distinct muscle fiber types (Fig. 1.3A).15 About 80% of fibers in the orbital layer of each EOM are fast, twitch-generating, singly innervated fibers (SIFs) resembling mammalian skeletal muscle fibers, whereas 20% are multiply innervated fibers (MIFs) that either do not conduct action potentials or do so only in their central portions.15 SIFs are relatively small in diameter, and their abundant mitochondria impart a dark staining characteristic with Masson trichrome. The metabolism and blood supply of orbital SIFs are tailored to their unique mechanical loading and nearly continuous activity. Orbital SIFs are specialized for intense oxidative metabolism and fatigue resistance (5). The vascular supply in the orbital layer is high,2,16 about 50% greater in humans than the well-perfused global layer. The orbital layers of human rectus EOMs can be distinguished from the global layers during MRI by the more rapid perfusion of the former after bolus injection of intravenous gadodiamide contrast.17
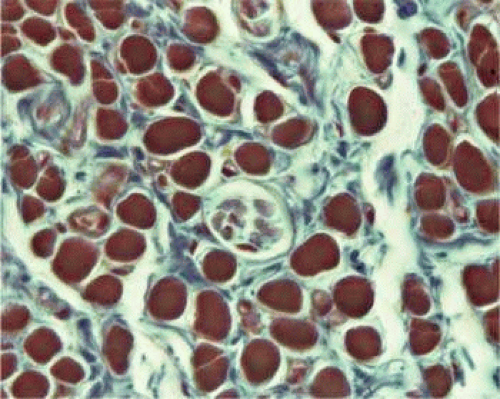
Orbital SIFs express unique myosin isoforms, perhaps related to the requirements of fast twitch capability against continuous loading, since alterations in EOM activity patterns can change EOM-specific myosin heavy chain gene expression is adapted to EOM activity patterns.17 The function of the relatively sparse and primitive orbital MIFs remains unclear. The global layer also contains MIFs that either do not conduct action potentials or do so only in their central portions and not near their origins or insertions. Global layer MIFs end as myotendinous cylinders termed palisade endings, many of which are innervated and have been proposed to have a proprioceptive function,18,19,20,21 although more recent evidence suggests that palisade endings are more likely to have a motor function.22 The MIFs resemble fibers found in amphibians and are capable of generating slow contractions without twitches. Orbital MIFs may have a proprioceptive function. Also found in the orbital layer are muscle spindles, cylinders of connective tissue containing a handful of small muscle fibers (Fig. 1.4). Since in skeletal muscles spindles are proprioceptive organs sensing EOM length, the spindles of the orbital layer appear suited to signal the length of the orbital layer and thus the position of the corresponding connective tissue pulley.
The global layer contains one MIF and three designated types of SIFs that form a continuum distinguished by their density of mitochondria (Fig. 1.3B). The largest and most granular SIF is very similar to the orbital SIF, having almost as many mitochondria, whereas the other two SIFs have correspondingly fewer mitochondria. Global MIFs are still smaller, contain fewer mitochondria, and correspond to the orbital MIFs. No spindles are present in the global layer, but the anterior tendonous termination of the global layer of the rectus EOMs contains palisade endings. Palisade endings are distributed along the width of each rectus tendon near the insertion, and while formerly believed to function as proprioceptive organs,23 have immunochemical features more typical of motor effector structures.22 The possible role of palisade endings in proprioception is controversial on multiple grounds.24
Extraocular muscles, like all muscles, generate tension through the interaction of the proteins actin and myosin. A wide variety of isoforms of myosin are found in EOMs, with the predominant one in orbital SIFs, EOM-specific myosin, being found only in EOMs.3,15 Myosin isoforms vary along the length of individual EOM fibers. Neonatal and embryonic myosin isoforms persist throughout adult life at the anterior and posterior ends of SIFs. This persistence of immature myosins may be related to the capability of EOMs to adjust their total number of sarcomeres, a process that presumably occurs at the ends of the fibers.25 Variation in myosin expression may be the reason for the unique susceptibility of the EOMs to certain disorders such as endocrine myopathy, as well as their resistance to other disorders such as muscular dystrophy.26
Origins and Paths of the Striated Extraocular Muscles
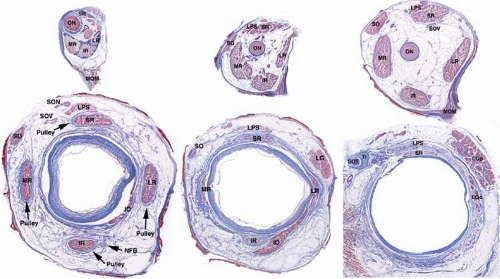
The rectus EOMs originate in the orbital apex from a common fibrous ring surrounding the optic nerve called the annulus of Zinn (Fig. 1.5, upper left). The SO muscle originates from the periorbita of the superonasal orbital wall slightly more anteriorly (Fig. 1.5, upper center). The rectus EOMs course anteriorly through loose areolar lobules of orbital fat without mechanical constraint on their paths until they enter their connective tissue pulleys, described below, that form sheathes as the EOMs penetrate posterior Tenon fascia (Fig. 1.5, lower left). The SO muscle remains tethered to the periorbita via connective tissues as it courses anteriorly, then thins to become continuous with its long, thin tendon (Fig. 1.5, lower center; Fig. 1.6). The concentric SO orbital layer terminates posterior on a peripherally located sheath (Fig. 1.5, lower right). Both the SO sheath and tendon pass through the trochlea, a cartilaginous rigid pulley attached to the superonasal orbital wall (Fig. 1.6). The trochlea typically calcifies in older people. After reflection in the trochlea, the SO tendon passes beneath the SR, thins, and flattens as it spreads out to its broad scleral insertion posterolaterally on the globe. Anterior to the trochlea, the SO sheath inserts on the nasal border of the superior rectus (SR) pulley. The IO muscle originates much more anteriorly from the periorbita of the inferonasal orbital rim adjacent to the anterior lacrimal crest and runs laterally to enter its connective tissue pulley immediately inferior to the IR at the point the IO penetrates the Tenon fascia (Fig. 1.5, lower right).
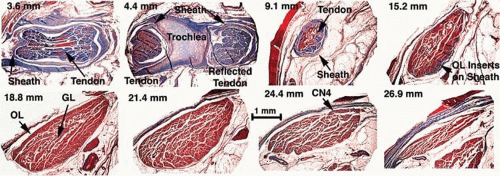 Figure 1.6. Superior oblique (SO) tendon, trochlea, and muscle belly in Masson trichrome–stained coronal sections of 17-month-old human. Numbers in the upper left of each panel indicate distance in mm from the anterior corneal surface. 3.6 mm. SO tendon is sectioned at its point of reflection and surrounded by the dense SO sheath that also reflects at the trochlea. 4.4 mm. Cartilage of the trochlea is visible between posterior (right) and anterior (left) intersections of the SO tendon with the plane of sectioning. Note that the SO sheath is also sectioned posterior and anterior to the trochlea. 9.1 mm. Posterior to the trochlea, the SO tendon is thin and enveloped by a thick sheath. 15.2 mm. The SO muscle belly is evident from the bright red staining. Orbital layer (OL) fibers at the peripheral portion at right are seen inserting on connective tissues contiguous with the thinner sheath. In sections not shown, OL fibers insert into the sheath around the entire circumference of the SO muscle. The global layer (GL) fibers transition to form the SO tendon. 18.8 mm. Small darker-staining OL fibers surround the larger brighter-staining GL fibers in the core of the SO belly. 21.4 mm. The midorbital portion of the SO has the largest cross section and exhibits a central GL and peripheral OL. 24.4 mm. The trochlear nerve (CN4) enters the SO at its superior orbital surface and forms a dense arborization within the muscle. 26.9 mm. The SO muscle thins near its origin from the bony orbital wall. The medial rectus muscle is seen below the SO. This section corresponds roughly to the upper middle row in Figure 15. |
The long axes of the bony orbits are angled about 23° laterally from the midsagittal plane (Fig. 1.7). The general configuration of the rectus EOMs and the SO is that of a cone, although it should be clear from Figure 1.5 that in the posterior orbit this configuration is incomplete, without interconnections among adjacent EOMs. Despite the implications of other texts,27 there exists no “muscle cone” of connective tissue forming bridges among the adjacent rectus EOM bellies in the mid to deep orbit. As they continue anteriorly, the rectus EOMs thin to become straplike bands about 10 mm in width, and ultimately their global layers become continuous with tendons that insert on the globe.
Pulleys of Extraocular Muscles
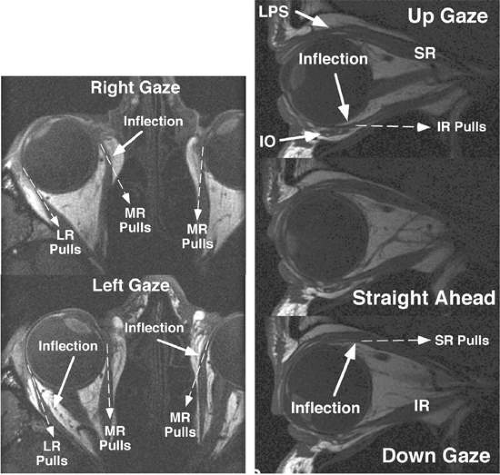
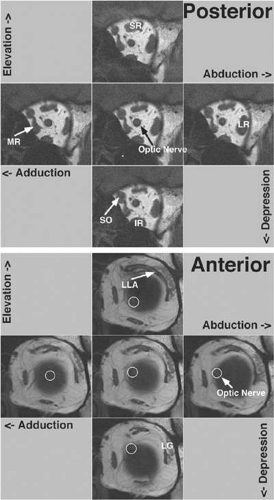
An important insight from contemporary tomographic imaging of EOMs is that they do not follow straight-line paths from their origins to their scleral insertions as implicitly and explicitly (but nevertheless incorrectly) indicated by older and even many modern textbooks. Discrete inflections in horizontal rectus EOM paths are obvious on axial images in abduction and adduction (Fig. 1.8A). These inflections occur more anteriorly in relaxation of the involved EOM and more posteriorly during its contraction. The paths of the vertical rectus EOMs are similarly inflected, being obviously not straight lines (Fig. 1.8B). The inflections in Figure 1.8 that bow rectus EOM paths toward the orbital wall are related to the pulley sleeves but do not correspond to the pulley rings described later. Inflections owing to pulley rings lie more anteriorly. The rectus EOMs in fact do follow linear paths from the annulus of Zinn to discrete points in the anterior orbit, paths that are remarkably stable even during the largest possible shifts of gaze in every direction. The stability of anterior rectus EOM paths is illustrated in Fig. 1.9 (lower), where despite substantial ocular abduction, adduction, supraduction (elevation), and infraduction (depression), only the junction of the optic nerve with the globe is seen to shift relative to the orbit. Posteriorly in the orbit (Fig. 1.9, upper), the EOMs also maintain stable paths but the cross sections of contracting EOMs increase, whereas those of relaxing EOMs decrease. These changes in cross section are dynamic indications of rectus contractility, as well as contractility of the SO28,29 and IO.28,30 Atrophic EOMs exhibit marked reduction in cross section and fail to demonstrate a contractile increase in cross section in their field of action.31,32,33 In monkeys following experimental trochlear neurectomy, prominent neurogenic atrophy of the SO muscle develops in 5 weeks but is confined to the global layer only.34,35
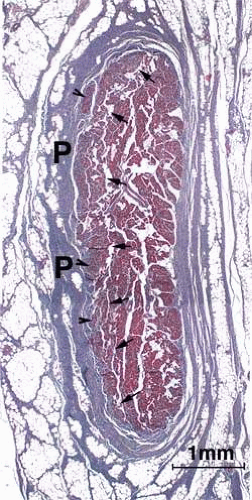
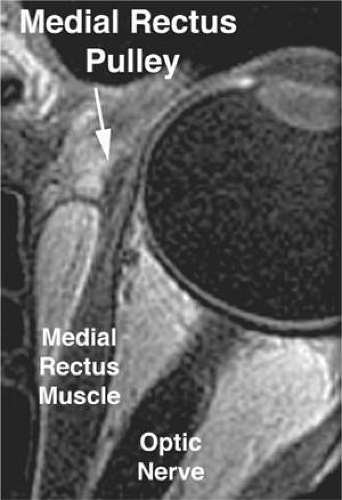
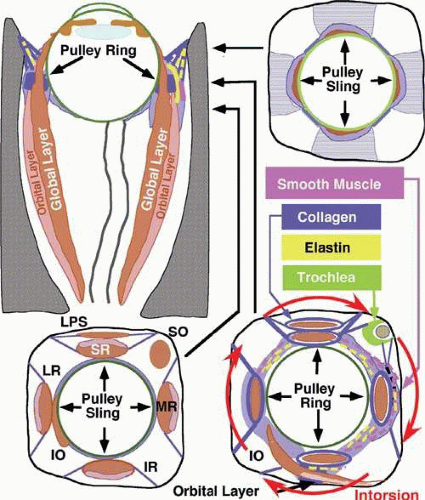
The discrete points in the anterior orbit where the EOM paths are inflected constitute the functional, mechanical pulleys, whose structure will be described in detail below. The orbital pulleys redirect EOM forces, but should not be misunderstood to be precisely structured like machine pulleys consisting of wheels with bearings.36 Anterior to the pulleys, rectus EOM paths shift, sharply when necessary, to follow the scleral insertions in eccentric positions of gaze. The pulleys thus act as functional mechanical insertions of the rectus EOMs. The pulling direction of each EOM is thus defined by the line segment connecting the EOM’s scleral insertion to the respective pulley, so that the location of the pulley influences EOM action profoundly. Rectus pulleys consist of discrete rings of dense collagen encircling the EOM and are about 2 mm long, coaxial with less substantial collagenous sleeves around the EOM (Figs. 1.2, 1.10, and 1.11). Anteriorly, these sleeves thin to form slings convex to the orbital wall, and more posteriorly the sleeves thin for form slings convex toward the center of the orbit. The anterior pulley slings are also known as the intermuscular septum. The collagenous pulley ring forms the fulcrum of the pulley that inflects the EOM path. Not surprisingly given this pivotal mechanical role, electron microscopy demonstrates the fibrils of pulley collagen to have an unusual criss-crossed configuration suited to high internal rigidity.36 Elastic fibers in and around pulleys6,37,38,39,40 provide reversible extensibility essential to these resilient tissues.41 Reversible extensibility, particularly in connective tissue bands that connect the pulleys to bony anchors on the orbital rim, means that the pulleys are suspended under elastic tension that tends to draw them anteriorly. Bands of smooth muscle in the pulley suspensions (Fig. 1.11), and particularly the peribulbar muscle connecting the nasal aspects of the vertical rectus pulleys, provide a means of active generation of modest modulatory force on the pulleys.37,38,39 The most prominent smooth muscle is the inframedial orbital muscle extending between the MR and IR pulleys.38 However, large shifts in pulley positions probably require forces exceeding those developed by smooth muscles.
The overall structural organization of the pulleys is illustrated in Figure 1.12. The anterior pulley slings, forming what has also been termed the intermuscular membrane, are routinely dissected at strabismus surgery. Note that the orbital layers of each rectus EOM insert on their respective pulleys and act to oppose the passive tension produced by the elastic suspensions of the pulleys from the anterior orbit. The pulley suspensions have also been described as “check ligaments,”42 a term that unfortunately fails to clearly describe their mechanical functions. The region of the pulleys and their sleeves is endowed with dense connective tissue that constrains EOM paths. Posterior Tenon fascia forms a socket for the globe, suspended from the orbital rim like a drumhead. The peripheral part of posterior Tenon fascia is very dense with collagen, elastin, and smooth muscle in circumferential distributions. The pulley sleeves penetrate this part of posterior Tenon fascia. The central part of posterior Tenon fascia is thin and compliant, so that the optic nerve, the ciliary nerves, and ciliary vessels can pass through it easily despite large gaze shifts. Note that posterior to the pulleys, there exists no connective tissue to constrain EOM paths. There is no “muscle cone,” only loose, areolar lobules of fat among the EOMs, vessels, and nerves of the posterior orbit. Unnecessary surgical dissection of the pulley rings or of posterior Tenon fascia is to be avoided, for in addition to liberating deep orbital fat, this will damage the essential function of the pulleys and promote adherence to the globe and scarring of these dense connective tissues that can severely limit normal eye movements.
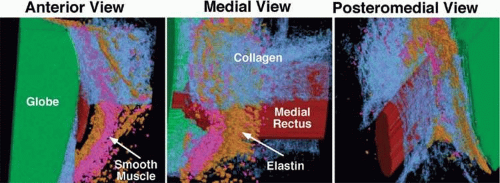
Detailed views of the three-dimensional structure of rectus pulleys can be obtained by computer-assisted reconstruction of serial, coronal histologic sections stained for specific tissue constituents such as collagen, elastin, and an α-actin specific to smooth muscle.38 Such reconstructions, when manipulated using computerized virtual reality techniques, enable orbital tissues to be visualized from perspectives not otherwise possible. Figure 1.11 shows a posteromedial view of the MR pulley, as if seen from the surgically inaccessible center of the orbit, demonstrating its sleevelike character. Figure 1.11 also provides a surgically possible anterior view of the MR pulley region, in which the collagen in the inferior half of the pulley has been rendered transparent to better display the embedded elastin and the extensive peribulbar smooth muscle.
The foregoing anatomic relationships can, with the benefit of the preceding insights, be readily demonstrated in gross dissections and in surgical exposures. Following typical conjunctival incision and engagement of a rectus muscle on a surgical hook, the white anterior pulley slings come immediately into view. These tissues play little role in constraining EOM path, although they are related to the more posterior pulley rings. The anterior pulley slings can, with the occasional exception of the lateral levator aponeurosis at the superior border of the LR, be posteriorly displaced by blunt dissection, although sharp dissection is sometimes unavoidable despite its undesirability. After this posterior displacement, fine fibrous bands constituting the insertion of the orbital layer of the EOM on the glistening white pulley suspension can be visualize.6,10 (Fig. 1.13A). This insertion site is intimately related to the mechanically effective site of the pulley, the connective tissue ring, as better demonstrated in cadaveric preparations such as Figure 1.13B. In such preparations, the anterior anchoring point of the MR pulley may be confirmed to be at the anterior lacrimal crest. With detachment of the orbital layer from the MR pulley, the pulley ring may be stretched under high tension to demonstrate the complete ring in posterior Tenon fascia (Fig. 1.13B). It should be noted that unless the insertion of the orbital layer on the pulley is severed (generally an unnecessary surgical maneuver), the MR cannot through surgical accident slip posteriorly through the pulley to become “lost” in the deep orbit. All the other rectus EOMs are similarly protected against loss by their orbital layer attachments to their pulleys, and in the case of the IR and LR, to the adjacent IO.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree