Incidence
Epistaxis is a common problem ranging from minor traces of blood or clot to isolated massive bleeds that may be life-threatening. Most patients have self-limiting episodes that do not require medical care and thus the true incidence is unknown. In spite of this, there is an estimated global lifetime incidence of 60% (
1). From an otolaryngologic standpoint, epistaxis may account for up to 33% of emergency admissions with a median age of 70 years (
2). The overall age of onset describes a bimodal distribution with an increased incidence in childhood followed by a peak incidence in the sixth decade. While younger individuals overwhelmingly present with minor bleeding derived from the anterior septum, older patients are far more likely to present with a severe acute bleed. Of note, epistaxis in children under 2 years old is rare and should raise the suspicion of an underlying illness or child abuse (
3).
Some studies have also suggested a seasonal variation in the incidence of epistaxis with episodes being more common in the fall and winter. While this may reflect an overall decrease in the ambient humidity during the colder months these findings have not been universally accepted (
4).
Etiology
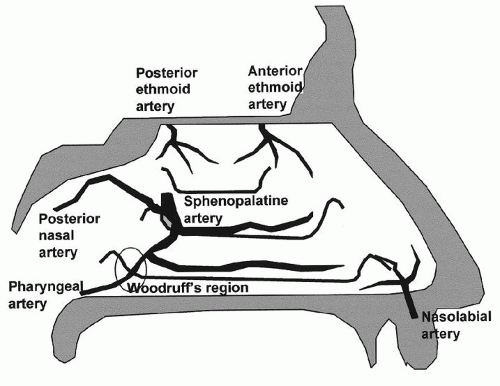
When considering the management and prognosis of patients with epistaxis, it is useful to classify the bleeding site into anterior or posterior. There is no precise anatomic distinction between the two; however, anterior bleeds are more common (
5) and tend to originate on the septum while posterior bleeds are generally higher volume and arise laterally from branches of the sphenopalatine artery (SPA). Bleeding may also be classified as primary (or idiopathic), which accounts for the majority of cases, or secondary, which results from known local or systemic factors. Spontaneous mucosal bleeding may occur in all age groups but is particularly problematic in children as this region is subject to mucosal desiccation, irritant exposure, and digital trauma. While the advent of the endoscopic era has greatly improved our ability to identify and manage the specific site of bleeding, many patients presenting to a general emergency department will continue to lack a definitive diagnosis and thereby remain in the idiopathic category.
Secondary epistaxis refers to bleeding resulting from a known cause (
Table 32.1). Any disorder that disrupts the mucosal epithelium or impairs clotting and platelet function may result in nosebleeds. Management of secondary epistaxis requires addressing the underlying etiology and thus it is useful to subcategorize it into local or systemic causes.
There are many local causes of epistaxis including sinonasal tumors, granulomatous disease, and septal perforation; however, the majority are iatrogenic or posttraumatic. In adolescent males with unilateral epistaxis and nasal obstruction, the presence of a juvenile nasopharyngeal angiofibroma should be ruled out (
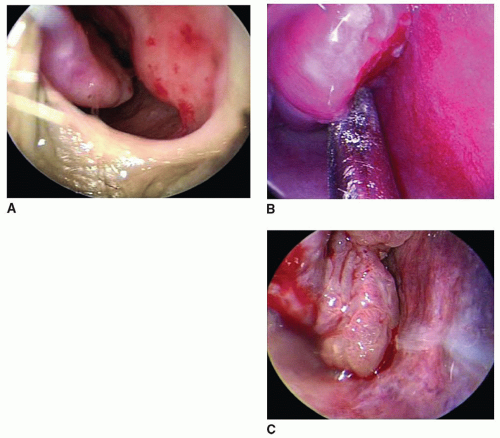
6). Postoperative bleeding following endoscopic skull base surgery also deserves special consideration not only because of the potential for exposure of critical neurovascular structures but also for the fact that blind packing may potentiate a neurologic catastrophe. Isolated vascular lesions such as hemangiomas, pyogenic granulomas, and telangiectasias are another local source of epistaxis that can sometimes be difficult to identify but may result in recurrent large volume bleeding (
Fig. 32.1).
In patients with epistaxis and multiple cutaneous, lip, oral, and intranasal telangiectasias, the diagnosis of hereditary hemorrhagic telangiectasia (HHT) must be considered (
Table 32.2). This entity represents both a diagnostic and
therapeutic challenge as patients may have underlying lifethreatening intracranial, gastrointestinal (GI), hepatic, and pulmonary arteriovenous malformations, (AVMs) which must be concomitantly evaluated. A recent international guidelines statement also recommended genetic testing of all patients to allow for identification of family members who have not yet met the diagnostic criteria for HHT (
7). Due to the presence of multiple local sources of potential bleeding, HHT patients often require repeated and aggressive epistaxis interventions, which is covered later in the chapter.
Systemic causes of epistaxis typically relate to disorders or medications resulting in impaired clotting or platelet function including renal failure, NSAID, and salicylate use. For example, low-dose aspirin has been shown to increase the risk of epistaxis when compared with placebo (19.1% vs. 16.7% respectively) (
8). Warfarin use may also
predispose to epistaxis, particularly when the INR is not tightly controlled. Epistaxis patients should also be questioned regarding the use of over-the-counter supplements including garlic, ginkgo, or ginseng, which can result in a mild systemic coagulopathy (
9).
Inherited bleeding diatheses, the most common of which being hemophilia A followed by von Willebrand disease, and hematologic malignancies are less common causes of epistaxis although they should be considered in any broad differential diagnosis. Hepatobiliary disease may also result in coagulopathy related epistaxis secondary to a reduction in the production of vitamin K-dependent factors (prothrombin, factors VII, IX, and X, proteins C and S).
While hypertension exacerbates epistaxis, it has not been shown to be an independent causative factor (
10). When hypertension is suspected in the setting of epistaxis, it should be investigated and managed appropriately.