Endoscopic Evaluation and Treatment of CSF Leaks
Pete S. Batra
HISTORY OF CSF LEAK SURGERY
The history of surgery for cerebrospinal fluid (CSF) rhinorrhea has witnessed evolution from an entity historically managed by neurosurgeons via open approaches to one largely in the rhinologic domain amenable to endoscopic techniques. The initial description of CSF leak management was provided by Dandy (1) in 1926, who repaired a case of pneumocephalus resulting from a frontal dural tear suffered from trauma via a frontal craniotomy approach. In 1948, Dohlman (2) reported the first extracranial repair of cribriform plate CSF leak using muscle and nasal mucosa via a nasoorbital incision. In 1952, Hirsch (3) repaired a sellar CSF leak resulting from resection of a pituitary tumor by a transnasal transseptal approach. In 1981, Wigand (4) provided the initial description of endoscopic repair of CSF leaks; he repaired six intraoperative leaks encountered during 300 ethmoidectomy procedures. In 1996, Lanza et al. (5) reported endoscopic repair of 36 skull base defects, codifying the endoscopic technique as the method of choice for treatment of CSF rhinorrhea.
CSF PHYSIOLOGY AND INTRACRANIAL PRESSURE BASICS
Normal CSF is a clear colorless fluid nearly iso-osmotic to plasma. CSF serves several key functions, including protecting the central nervous system, providing an alternate circulation for distribution of micronutrients, and mediating intracranial compliance in physiologic and pathologic states. Majority of CSF is produced from the choroid plexus of the ventricular system (70%). The remainder is produced extrachoroidally by capillary ultrafiltration (18%) and metabolic water production (12%). In an adult, the total CSF volume equals approximately 140 to 150 mL. The ventricles hold about 30 mL, while the brain and spinal subarachnoid spaces contain 80 and 30 mL, respectively. Traditional teaching holds that CSF production occurs at a constant rate of 500 mL daily. However, quantitative magnetic resonance (MR) imaging has estimated daily production rates as high as 650 mL based on variations with circadian rhythm, with maximal production at 2:00 am and nadir at 6:00 pm. CSF absorption is a hydrostatic pressure-dependent event that occurs at the arachnoid villi juxtaposed to the dural sinuses. These villi act as pressure gradient one-way valves that transmit CSF into the venous system when the intracranial pressure (ICP) increases 3 to 6 cm H2O greater than venous pressure (6,7).
Normal ICP ranges from 5 to 15 cm H2O at the lumbar cisterns with the patient in a supine position. ICP is a dynamic measurement with activities, such as change in position, straining, and sneezing, transiently increasing it to 40 cm H2O. Further, the pressure can vary depending on time of day, patient age, respiratory phase, and cardiac cycle. Sustained increases or frequent spikes in the 20 to 30 cm H2O range can result in neurologic symptoms and requires measures to reduce the ICP. For example, patients with ICP exceeding 20 cm H2O in the setting of traumatic brain injury is strongly associated with mortality (6).
ETIOLOGY
A number of important etiologies may result in CSF rhinorrhea. The most common include accidental trauma, surgical trauma, congenital, tumors, and spontaneous (or idiopathic). Though earlier studies cited accidental trauma as the most common etiology, recent reports have noted spontaneous CSF leaks to be the most common cause of CSF leakage. Banks et al. (8) reported on 193 cases repaired over 21 years; the most common etiologies included spontaneous (40%), iatrogenic trauma after endoscopic sinus surgery (ESS) (26%) or endoneurosurgical procedures (20%), accidental trauma (8%), and congenital (4%). Careful understanding of these etiologies is a prerequisite
prior to devising a comprehensive diagnostic and management strategy for effective surgical repair of CSF rhinorrhea.
prior to devising a comprehensive diagnostic and management strategy for effective surgical repair of CSF rhinorrhea.
Accidental Trauma
The reported incidence of posttraumatic CSF leaks ranges from 2% to 4% in cases of blunt head injury. Eighty percent of leaks will occur within 48 hours after injury, and 95% will manifest in 3 months. However, a small but important subset may have a delayed presentation months, or even years, after the initial injury. Though 70% will present with active CSF rhinorrhea, the remainder will only develop pneumocephalus or ascending meningitis as part of the initial presentation. The most common locations of skull base fractures in descending order include the ethmoid-cribriform plate region, posterior table of the frontal sinus, orbital roof, and sphenoid sinus. The fractures can range from linear fractures to massive communication of the entire skull base, based on the extent of traumatic force. Concomitant facial fractures and intracranial lesions, such as cerebral contusion and epidural or subdural hematoma, are not uncommon.
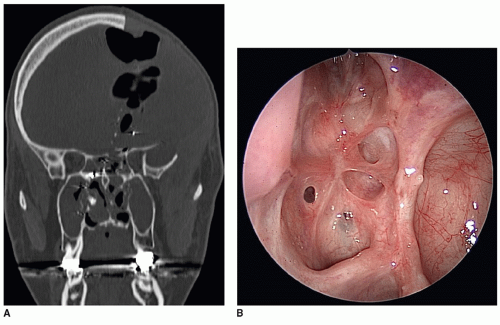
Approximately 60% to 70% of traumatic leaks will close with conservative measures, such as bed rest, stool softeners, and lumbar drain (LD). However, the dura does not regenerate in these cases and the skull base may only be covered by a thin monolayer of nasal mucosa or fibrous tissue. Further, this layer may break down over time by sinonasal inflammatory processes or by the pulsatile effect of the brain. The risk of ascending meningitis in this setting has been noted in 30% to 40% of cases. This underscores the need for a proactive strategy with early detection and repair of persistent posttraumatic CSF leaks. All cases of suspected trauma should undergo high-resolution thin-cut computed tomography (CT) imaging to delineate the extent of skull base fracture. The management strategy should be highly individualized given the heterogeneity of these fractures. Cases with linear disruption of the ethmoid or sphenoid roof are good candidates for an endoscopic approach. Cases with extensive skull base disruption, especially with associated skull fractures or intracranial injury, may require an open neurosurgical approach. Penetrating skull base trauma from gunshot wounds or other projectile missiles can also result in significant skull base comminution and may be associated with extensive granulation tissue in the path of the foreign body (Fig. 45.1A and B). Close multidisciplinary coordination among subspecialty services is imperative to optimize care in these challenging cases (9,10).
Surgical Trauma
Surgical trauma from ESS or neurosurgical procedures represents the most common causes of iatrogenic CSF leaks. With the advances in image-guided surgery and sophisticated surgical instrumentation, the risk profile of ESS
has improved dramatically with the general incidence in the 0% to 3% range. In a recent review of 6,908 endoscopic sinus surgeries at a single institution, 32 (0.46%) intraoperative CSF leaks were reported (11). Approximately 70% of the leaks were evident at the time of surgery, while 30% manifested postoperatively. The side distribution was right in 56%, left in 41%, and bilateral in 3% of cases. The leaks were commonly to result in hands of residents and novice attending surgeons. Further, asymmetry and low-lying skull base have been recognized as potential risk factors for iatrogenic leaks.
has improved dramatically with the general incidence in the 0% to 3% range. In a recent review of 6,908 endoscopic sinus surgeries at a single institution, 32 (0.46%) intraoperative CSF leaks were reported (11). Approximately 70% of the leaks were evident at the time of surgery, while 30% manifested postoperatively. The side distribution was right in 56%, left in 41%, and bilateral in 3% of cases. The leaks were commonly to result in hands of residents and novice attending surgeons. Further, asymmetry and low-lying skull base have been recognized as potential risk factors for iatrogenic leaks.
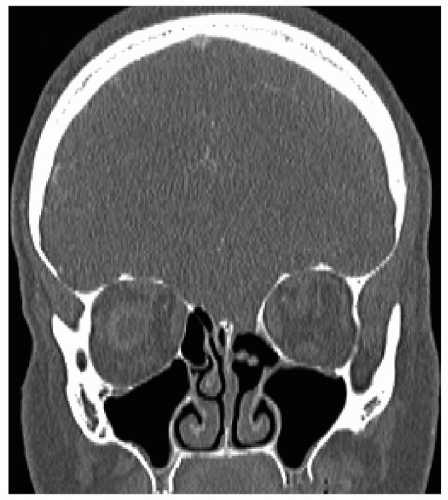
The two most common locations for CSF rhinorrhea include the lateral lamella of the cribriform plate (LLCP) and posterior ethmoid roof as it transitions to the sphenoid sinus. LLCP represents the thinnest portion of the skull base with mean thickness of 0.5 mm; this decreases 10-fold at the ethmoidal sulcus, the entrance of the anterior ethmoid artery into the olfactory fossa. The region is more at risk on the right side, especially in right-handed surgeons, given tendency to drift medially during the ethmoid dissection. Aggressive resection of the vertical attachment of the middle turbinate to the skull base also risks injury to the cribriform plate. The posterior ethmoid roof is at risk in scenarios where the maxillary sinus is extensively aerated; this excessive pneumatization encroaches more medially and superiorly, resulting in inversely proportional decrease in total inferior to superior height of the posterior ethmoid. High entry into the basal lamella poses a significant risk of injury to the posterior ethmoid roof (Fig. 45.2). The size of the skull base defect can vary from millimeters to greater than 2 cm, especially in cases of use of powered instrumentation. Underlying dural and intraparenchymal brain injury may result from breach of the skull base boundary, especially in cases of significant anatomic disorientation and delay in recognition of this violation.
A variety of neurosurgical procedures can result in intraoperative CSF leaks. Historically, rhinologic involvement in this setting has been limited to addressing inadvertent postop leaks resulting from open transsphenoidal hypophysectomy or transcranial approaches to the anterior skull base (ASB) for neoplasm resection. Paraclinoid aneurysm clipping complicated by postop CSF rhinorrhea also represents another clinically challenging scenario given the narrow confines of the opticocarotid recess in the sphenoid sinus. In rare cases, a CSF leak from lateral skull base surgery may manifest as CSF rhinorrhea as it egresses via the eustachian tube. Management of the leak at the primary site represents the treatment of choice, though eustachian tube closure may be required in select cases.
The advent of advanced endoscopic techniques has greatly facilitated the surgical management of skull base neoplasms. Endoneurosurgical procedures, including transcribriform and transethmoid approaches to the anterior cranial fossa and transclival approaches to the posterior fossa, may result in large skull base defects with high-flow CSF leaks. This surgical paradigm involves close coordination between rhinology and neurosurgery for access to the skull base, combined tumor resection, and planned repair of the skull base defect with multilayered closure or vascular pedicled flap. Thus, the iatrogenic leaks in these cases are expected sequelae of the surgical procedure and addressed as an integral component of the surgery.
Spontaneous
Spontaneous CSF leaks represent an important clinical subset of leak patients whose prevalence may be increasing. No obvious definable etiology such as trauma, surgery, or neoplasm can be identified in this group. Emerging data have demonstrated that these represent a distinct clinical entity, being typically observed in middle-aged females with elevated body mass index. Common presenting symptoms include headaches, pulsatile tinnitus, and vertigo, along with CSF rhinorrhea. CT imaging may demonstrate a broadly attenuated skull base or arachnoid pit formation, while MR imaging may show partially or completely empty sella. This group also has the highest rate of encephalocele formation, being noted in 50% to 100% of cases. Many of these patients may have elevation of ICP or carry an underlying diagnosis of benign intracranial hypertension (BIH). The etiology of elevated ICP is not known but may be related to impaired CSF absorption at the arachnoid villi. Chronic elevation of ICP results in transmission of hydrostatic forces along the areas of least resistance such as the cribriform plate or lateral pterygoid recess of the sphenoid sinus causing erosion of the skull
base and ensuing herniation of meninges or brain and associated CSF rhinorrhea. Alternately, the forces may be exerted at the optic nerve sheath causing papilledema and visual loss (12,13).
base and ensuing herniation of meninges or brain and associated CSF rhinorrhea. Alternately, the forces may be exerted at the optic nerve sheath causing papilledema and visual loss (12,13).
Spontaneous CSF leaks represent the most challenging group for surgical repair and have been historically associated with the highest failure rate. This may in part be related to technical hurdles from a broadly, thinned skull base and associated encephalocele formation, along with underlying elevated ICP. The current surgical algorithm entails meticulous repair of the skull base defect and management of associated intracranial hypertension. Methods to accomplish the latter include administration of oral acetazolamide (ACTZ) to reduce the ICP. Ventriculoperitoneal (VP) shunting may be considered in severe cases.
Congenital
Congenital encephaloceles are herniation of meninges and/or brain through a skull base defect that maintain an intracranial connection. Though occipital lesions comprise 70% of these lesions, the otolaryngologist is most likely to encounter sincipital (20%) or basal (10%) encephaloceles. Sincipital encephaloceles are further subdivided into nasofrontal, nasoethmoidal, and naso-orbital lesions that exit anterior to the cribriform plate, typically presenting as a mass over the nose, glabella, or forehead. Basal encephaloceles present as an intranasal mass, with the defect being at the level of the cribriform plate or just posterior in the superior or lateral sphenoid. The mean age of presentation is 6 years, with common symptoms being nasal obstruction, CSF rhinorrhea, meningitis, or facial mass with hypertelorism.
Timely repair of the skull base defect is imperative to stop the CSF leak, to minimize risk of meningitis, to alleviate nasal obstruction, or to prevent further facial deformity. Traditional approaches have relied on combined extra- and intracranial repair by otolaryngology and neurosurgery. The advent of endoscopic techniques has facilitated minimally invasive resection of encephaloceles with layered skull base reconstruction and avoidance of external incisions with high success rate. Nonetheless, the technical challenges should not be understated given the narrow confines of the pediatric sinonasal anatomy and need for appropriate rigid scopes and instrumentation. In select cases, surgical management may need to be deferred till age of 2 to 3 years to allow for adequate facial growth to accommodate an endoscopic approach (14,15).
Tumors
Skull base neoplasms can result in CSF rhinorrhea in rare circumstances. Primary brain parenchymal or pituitary neoplasms can cause extensive bone erosion at the skull base with a resultant CSF leak. Conversely, aggressive sinonasal malignant neoplasms can invade the skull base and cause a CSF leak. Moreover, intracranial neoplasms may cause communicating or noncommunicating hydrocephalus, leading to CSF leakage. Management of the underlying neoplasm and elevated ICP are imperative in these scenarios. CSF leak repair, if clinically warranted, should only be undertaken after the underlying etiology is addressed.
PATIENT WORKUP
Patient History
The classic presentation of a CSF leak involves unilateral clear rhinorrhea that worsens with straining or bending over. Many patients with a spontaneous leak may recall a history of antecedent viral upper respiratory infection or significant sneezing or coughing as the inciting event. Patients may report a salty or metallic taste from posterior drainage of the CSF rhinorrhea. Headaches are a common symptom from sudden changes in the ICP from the CSF leakage. Careful history should ascertain for previous trauma and sinonasal or neurosurgical procedures. In select cases, sudden onset of meningitis may be the only presenting symptom of a skull base defect without associated CSF rhinorrhea. It is imperative for the otolaryngologist to evaluate for defects in the skull base as a potential etiology of unexplained meningitis. Equally importantly, any patient with an active CSF leak should be carefully counseled on the warning signs of meningitis until successful closure is performed.
Physical Examination
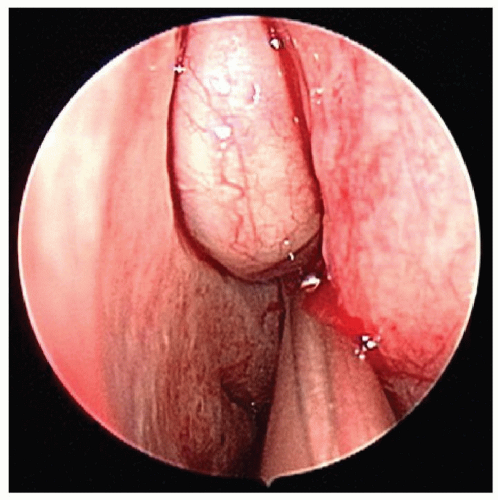
Complete head and neck examination is an integral component of the evaluation. Patient may be able to produce the clear rhinorrhea by leaning forward. The otologic evaluation may reveal a middle ear effusion suggestive of a possible temporal bone leak via the eustachian tube. An ophthalmologic examination may be necessary if papilledema from BIH is suspected. Rigid nasal endoscopy is crucial for accurate diagnosis of CSF rhinorrhea. Endoscopy may demonstrate a glistening pulsatile mass in the olfactory cleft (Fig. 45.3). Further, pooling of clear secretions in the nasal cavity may be noted that rapidly fills with suctioning.
Laboratory Testing
Historically, glucose strip testing has been employed as a rapid test to assay for CSF rhinorrhea. False positives are common given the reducing substances in nasal mucus. It represents a nonsensitive and nonspecific study and should not be relied upon in the contemporary diagnostic strategy for the CSF leaks. β2-Transferrin is a protein produced by the neuraminidase activity in the brain and is detectable almost exclusively in CSF. Low levels of β2-transferrin are found in the perilymph of the cochlea and aqueous and vitreous
humor of the eye. It is a highly sensitive and specific noninvasive test that has been used successfully as a primary screening modality for suspected CSF leakage. Minimal volume of 0.5 mL is required for the assay, which may prove problematic in patients with slow or intermittent leaks. Further, the test is only performed at select laboratory facilities across the country, thus resulting in a delay of a few days to confirm diagnosis. The result should be interpreted with caution in patients with liver failure due to derangements in serum protein levels in this pathologic state. β-trace protein is synthesized primarily by the epithelial cells of the choroid plexus and is found in concentrations 35-fold higher in CSF than in plasma. It may represent a quantitative alternative to β2-transferrin for testing for CSF rhinorrhea. The test is available in Europe for commercial usage. It must be remembered that these biochemical tests can only prove the presence of CSF in the tested specimen, but are unable to directly demonstrate the CSF leakage or leak site (16).
humor of the eye. It is a highly sensitive and specific noninvasive test that has been used successfully as a primary screening modality for suspected CSF leakage. Minimal volume of 0.5 mL is required for the assay, which may prove problematic in patients with slow or intermittent leaks. Further, the test is only performed at select laboratory facilities across the country, thus resulting in a delay of a few days to confirm diagnosis. The result should be interpreted with caution in patients with liver failure due to derangements in serum protein levels in this pathologic state. β-trace protein is synthesized primarily by the epithelial cells of the choroid plexus and is found in concentrations 35-fold higher in CSF than in plasma. It may represent a quantitative alternative to β2-transferrin for testing for CSF rhinorrhea. The test is available in Europe for commercial usage. It must be remembered that these biochemical tests can only prove the presence of CSF in the tested specimen, but are unable to directly demonstrate the CSF leakage or leak site (16).