Fig. 3.1
(a) Similar spindle-shaped phenotypes among orbital fibroblasts, dermal fibroblasts, and fibrocytes (hematoxylin and eosin, ×20). (b) Fibrocytes from individuals with GD display cell surface receptor CD34. (1) Immunofloresence staining of CD34 in TAO-derived tissue (inset as negative control). (2) Absence of CD34 expression in healthy orbital tissue (inset as positive control). (c) Orbital fibroblasts from individuals with and without TAO display similar receptors as fibrocytes, as shown by flow cytometric analysis with anti-CD34 and anti-Col I antibodies. (Reprinted with permission; Douglas et al. Increased generation of fibrocytes in thyroid-associated ophthalmopathy, Copyright 2010. The Endocrine Society)
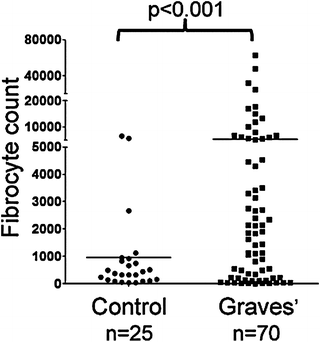
Fig. 3.2
Abundance of fibrocytes cultivated from PBMCs of individuals with GD (5,268 ± 1,260 fibrocytes per 106 PBMCs, n = 70) and from healthy individuals (954 ± 329 fibrocytes per 106 PBMCs n = 25) (p < 0.001). (Reprinted with permission; Douglas et al. Increased generation of fibrocytes in thyroid-associated ophthalmopathy, Copyright 2010. The Endocrine Society)
The mechanism through which TSH and TSI up-regulate IL-6 production in fibrocytes has been at least partially solved recently. Raychaudhuri and colleagues [45] found that the induction of IL-6 is completely independent of adenylate cyclase activation or the generation of cAMP. In these cells, TSH activates PDK1, AKT/PKB and PKC pathways, and disruption of any of these pathways attenuates the induction of IL-6. Further, the isoenzyme, PKCβII is expressed and utilized in mediating the responses to TSH in fibrocytes. The study further compared the induction of IL-6 in fibrocytes with those occurring in orbital fibroblasts derived from patients with GD and TED. It would appear that as circulating fibrocytes transition to CD34+ orbital fibroblasts, PKCβII expression and usage disappear and that PKCμ becomes expressed. Further, following the cellular transition, PKCμ signaling then becomes an indispensible component of the signaling to IL-6 expression. When TED-derived orbital fibroblasts which uniquely contain CD34+ cells are isolated from their CD34− counterparts by flow cytometry-based cell sorting, expression of PKCμ is extinguished and PKCβII again becomes detectable and thus the profile of PKC isoenzymes reverts to that of the circulating fibrocyte. IL-6 up-regulation following TSH treatment in fibrocytes and orbital fibroblasts results both from a coordinate activation of the IL-6 gene promoter and from enhanced IL-6 mRNA stability. Further, the transcriptional activity of the IL-6 gene can be traced to a CREB binding element extending from −213 to −208 nt, and to an NF-κB site extending from −78 to −62 nt. Thus TSHR signaling appears to involve a robust, cyclic AMP-independent set of pathways in non-thyroid epithelial cell types such as fibrocytes. Armed with these insights, it may be possible to interrogate multiple potentially attractive therapeutic targets for interrupting IL-6 expression in the context of GD and TED.
Fibrocytes Express Multiple “Thyroid-Specific Proteins”
Another interesting aspect of the CD34 fibrocyte is its expression of multiple “thyroid-specific” proteins. Fernando and colleagues [46] reported that those cultivated from circulating peripheral blood mononuclear cells express not only TSHR but also low levels of Tg mRNA. Levels of the transcript are considerably lower than those found in thyroid tissue. Tg resolved on polyacrylamide gel electrophoresis as a 305 kDa protein. Both 125Iodine and 35S methionine can be incorporated into Tg expressed by fibrocytes. siRNAs targeting Tg attenuated the synthesis of the protein. Tg gene promoter is active in fibrocytes. Compared with Tg levels found in fibrocytes, those in orbital fibroblasts from donors with GD were substantially lower. When GD-orbital fibroblasts were sorted into pure CD34+ and CD34− populations, Tg mRNA expression segregated to CD34+ fibroblasts. Tg expression is considerably greater in CD34+ fibroblasts than in the mixed cell population found in parental cultures. Two additional thyroid-specific proteins have been detected in the protein expression repertoire of fibrocytes, namely sodium iodide symporter (NIS) and thyroid peroxidase (TPO) [47]. Levels of NIS and TPO mRNAs are substantially lower than those found in thyroid tissue and FRTL-5 cells in culture. However they are adequately abundant to result in translation into detectable protein. They are considerably higher than those detected in dermal fibroblasts or orbital fibroblasts from healthy individuals and those with TED. The expression of multiple autoantigens suggested that a specific transcription factor might be involved, namely the autoimmune regulator protein (AIRE) [44]. AIRE is a major determinant of intra-thymic education that allows the negative selection of autoreactive thymocytes. In fibrocytes, AIRE mRNA and protein are detectable but at extremely low levels compared to thymus. Interruption of AIRE expression using small interfering RNAs resulted in substantial reductions in the steady state mRNA levels of TSHR, Tg, TPO, and NIS (Fig. 3.3). Further, knocking down AIRE expression also reduced levels of PAX8 and TTF1, the two transcription factors closely associated with expression of these proteins in thyroid epithelium. To confirm the importance of AIRE in this specific context, fibrocytes cultivated from an individual with APS 1 syndrome were also interrogated for levels of thyroid proteins and found to be significantly below those found in an unaffected first degree relative. AIRE expression in fibrocytes is the consequence of an active AIRE gene promoter and remarkably stable AIRE mRNA. The implications of these findings to the biology of fibrocytes and the tissues which they infiltrate include the suggestion that AIRE is an important component of extra thyroidal autoimmunity related to peripheral loss of tolerance to thyroid-associated proteins such as thyroglobulin, TPO, and TSHR. Further, they suggest that fibrocytes may be capable of synthesizing thyroid hormones or their derivatives. The array of self-antigens expressed by fibrocytes does not appear to be limited to those associated with autoimmune thyroid disease. Fernando et al. reported recently that IA-2 and ICA69 can also be detected in these cells, findings with implications for type 1 diabetes mellitus [48]. In addition to their expression of these proteins, fibrocytes express high levels of MHC Class II and can present antigens to T cells [49].
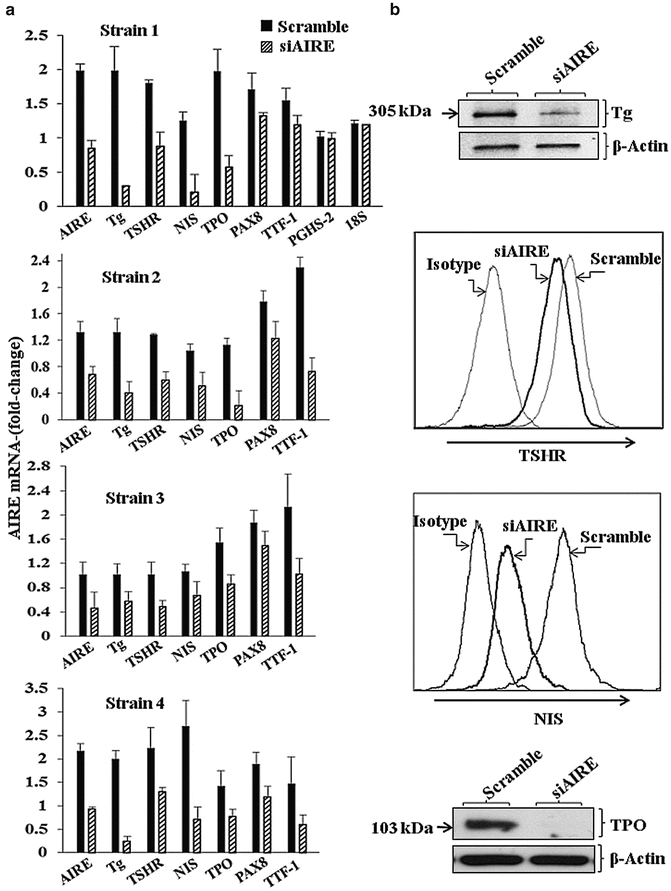

Fig. 3.3
Knocking down AIRE expression reduces Tg, TSHR, NIS, TPO, PAX8, and TTF-1 but not PGHS-2 or 18S RNA. (a) Fibrocytes from four different donors were treated with scrambled (control) siRNA or one targeting AIRE. Data are expressed as mean ± SD of three independent determinations. (b) Fibrocyte cultures were transfected with control siRNA or AIRE-targeting siRNA. Radiolabeled Tg, unlabeled TSHR, NIS, and TPO proteins were knocked-down by interrupting AIRE expression. (Reprinted with permission; Fernando et al. Copyright 2014. The Endocrine Society)
Could Identification of Fibrocytes as Participants in TED Disclose Additional Therapeutic Targets?
Despite substantial efforts to understand better the mechanisms responsible for the initiation of GD and the factors that overarch involvement of the orbit in TED, current knowledge remains mired in uncertainty. Much of this can be attributed to an absence of preclinical models that faithfully recapitulate the human disease. These deficits have resulted in substantial unmet need in how we treat this disease. Especially during the active phase when surgery is frequently discouraged, medical options are limited. Nonspecific anti-inflammatory agents, including corticosteroids, nonsteroidal agents (PGHS-2 inhibitors), and antimetabolites may lessen the symptoms directly caused by inflammation. But none of these agents is thought to alter the clinical course of TED. More recently, targeted strategies, many repurposed from other diseases, have been used on an ad hoc basis. Anti-CD20 monoclonal antibody (rituximab) is the currently best-studied among these and the results of two separate prospective trials are eagerly awaited. Other biologics, such as those that interrupt cytokine networks, have yet to be studied in a manner that could lead to conclusions about their efficacy. Their roles in the therapy of active TED remain to be established. Further, all of these candidate agents are associated with potentially serious side effects. Thus, we continue to rely on surgical intervention for ocular rehabilitation.
Recognition of the potential involvement of fibrocytes in GD and TED widens the horizons for future inquiry into disease mechanisms. It may be possible to directly target specific aspects of their behavior as potential mediators of the immune reactivity systemically as well as within the orbit space. To be certain, TED is a complex autoimmune condition with numerous molecular and cellular underpinnings. We are only now beginning to understand the underlying processes. Further insights into the potential roles fibrocytes play in the disease, especially the initial phases, should bring us closer to developing more effective treatments.
Acknowledgments
The valuable assistance of Yao Wang in identifying reference material for this chapter is gratefully acknowledged as is the great help provided by Ms. Justyna Piernicka. This work was supported in part by National Institutes of Health grants EY008976, EY011708, DK063121, Core Center for Vision grant EY007003 from the National Eye Institute, an unrestricted grant from Research to Prevent Blindness, and the Bell Charitable Foundation.
References
1.
Werner SC, Ingbar SH, Braverman LE, Utiger RD. Werner and Ingbar’s the thyroid: a fundamental and clinical text. 7th ed. Philadelphia: Lippincott-Raven; 1996. p. 1124.
3.
Wang Y, Smith TJ. Current concepts in the molecular pathogenesis of thyroid-associated ophthalmopathy. Invest Ophthalmol Vis Sci. 2014;55:1735–48.PubMedCentralPubMedCrossRef
4.
Douglas RS, Gupta S. The pathophysiology of thyroid eye disease: implications for immunotherapy. Curr Opin Ophthalmol. 2011;22:385–90.PubMedCentralPubMedCrossRef
5.
Kazim M, Goldberg RA, Smith TJ. Insights into the pathogenesis of thyroid-associated orbitopathy: evolving rationale for therapy. Arch Ophthalmol. 2002;120:380–6.PubMedCrossRef
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


