Chapter 7 Electrogenesis of the Electroretinogram
Introduction
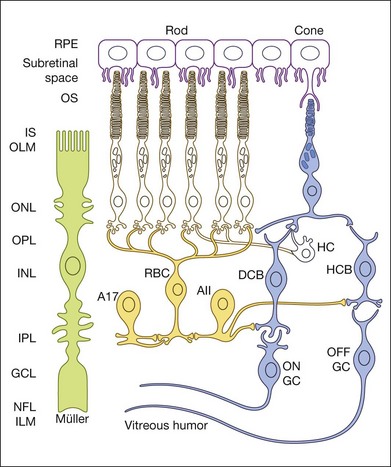
This chapter will review current knowledge of the cellular origins and mechanisms of generation of the various ERG component waves, progressing from distal retina to proximal retina. As will be described, the ERG is generated by radial currents arising either directly from retinal neurons, or as a result of the effect on retinal glia of changes in extracellular potassium concentration ([K+]o) brought about by retinal neuronal activity. Our understanding of the electrogenesis of the ERG was initially based on studies in a variety of cold-blooded vertebrate and as well as some mammalian species, described in more detail in previous reviews.1,2 Studies in a nonhuman primate model (macaque monkey) whose retina and ERG are very similar to that of humans (Fig. 7.1),3 and particularly those over the past two decades, have improved our understanding of the electrogenesis of the ERG in humans. This work will be highlighted wherever possible in this chapter. Clinical applications of the ERG will be described in the following chapter (Chapter 8, Clinical electrophysiology). Another recent review has examined the electrogenesis of a common animal model for retinal disease, the mouse.3
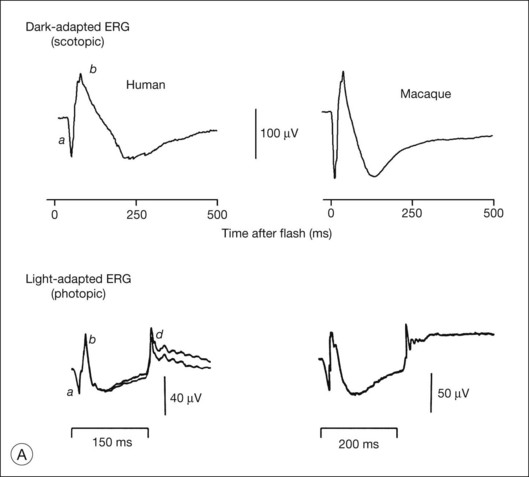
Fig. 7.1 Dark- and light-adapted full-field flash electroretinograms (ERGs) of human subjects and macaque monkeys. (A) Top: Dark-adapted (scotopic) ERGs in response to brief high-energy flashes from darkness occurring at time zero for a normal human subject (left) and an anesthetized macaque monkey (right). The stimulus energy was ~400 sc td/s. Bottom: Light-adapted (photopic) flash ERGs in response to longer-duration flashes on a rod-saturating background for a normal human subject (left) and a macaque monkey (right). For the human subject the stimulus was a 150-ms white full-field flash of 4.0 log ph td presented on a steady background of 3.3 log sc td. For the macaque, the same stimulus was used, but the flashes were 200 ms in duration. (Adapted from Sieving PA, Murayama K, Naarendorp F. Push–pull model of the primate photopic electroretinogram: a role for hyperpolarizing neurons in shaping the b-wave. Vis Neurosci 1994;11:519–32.) (B) ERG recording setup: recordings are made using a traditional ganzfeld bowl (left) or more modern light-emitting diode-based full-field stimulator. Burian-Allen and DTL fiber electrodes4 are illustrated. ERG recordings are amplified and sent to a computer for averaging, display, and analysis.
Generation of extracellular potentials: general concepts
An important factor affecting a given cell’s contribution to the ERG is its orientation in the retina. Radially oriented neurons in the retina (photoreceptors and bipolar cells) and glial cells (Müller cells and retinal pigment epithelial (RPE) cells) make larger contributions to the ERG than cells that are oriented more irregularly or laterally (e.g., horizontal and amacrine cells) (Fig. 7.2). The currents around cells that underlie the ERG enter the ECS at one retinal depth (the current source), and return into the cell at another depth (the current sink), creating a current dipole. Although most of the extracellular current flowing from source to sink traverses the ECS within the retina, some travels extraretinally – through the vitreous humor, extraocular tissues, sclera, choroid, the high resistance of the RPE (R-membrane), and back into the neural retina.
The polarity and amplitude of the recorded ERG will depend upon the location of the active and reference electrodes. In noninvasive studies, a common location for the active electrode is on the cornea via a contact lens electrode, e.g., as illustrated in Fig. 7.1B, a Burian-Allen electrode, or jet electrode, or another type of surface conductive electrode (e.g., a DTL fiber electrode,4 H–K loop,5 or gold foil electrode). For comfort, skin electrodes, which yield smaller signals, are sometimes used instead. In invasive studies of ERG components in animals, the active electrode may be positioned anywhere in the current path, including at different retinal depths, near particular cell types. The reference electrode also can be positioned anywhere in the path, but is often placed behind the RPE in studies of isolated retina, or retrobulbar in intact eyes. In noninvasive and clinical applications, the reference is positioned either under the eyelids, such as the speculum of the Burian-Allen electrode for bipolar recording between the contact lens electrode and the reference on the same eye, or remote from the eye (e.g., on the temple) for monopolar recording. The exact position of the remote reference is of minor consequence except for possible contamination of the retinal signal by other sources.
Other factors that influence the magnitude of the contribution to the ERG of a particular cell type include stimulus conditions, such as the strength of the stimulus and its wavelength (spectrum), the background illumination (that determines adaptation level of the retina), the duration and spatial extent of the stimulus, and the location of the stimulus within the visual field, as these stimulus parameters have different effects on the responses of the different cells. For example, the relative contributions of various cell types are different under dark-adapted (scotopic) and light-adapted (photopic) conditions when rod and cone pathways, respectively, are involved in generating responses. Spatially extended diffuse stimuli, i.e., full-field (ganzfeld) flashes that fill the retina evenly, using stimulators such as those illustrated in Fig. 7.1B, are commonly used to elicit the major ERG waves (a-, b-, and d-waves) from photoreceptors and bipolar cells. Contributions from these cells generally increase with the area of the retina stimulated as the number of cells, and hence the total extracellular current, is increased. In contrast, contributions of retinal ganglion cells (and other cells with antagonistic regions within their receptive fields) to the full-field flash ERG will be limited by the strength of surround antagonism. For photopic ERGs, particularly from subjects with trichromatic color vision, such as the macaque and human, stimulus wavelength also affects contributions from cells whose responses are dependent upon spectral antagonism.6,7
Spatial buffering by glial cells
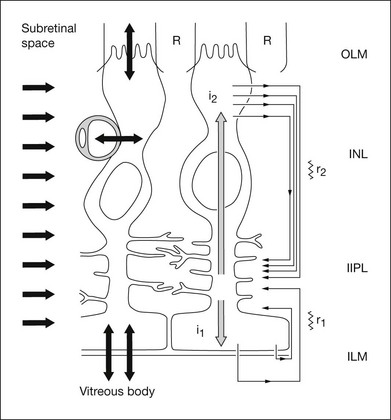
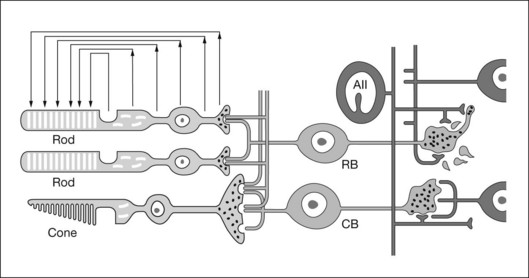
[K+]o spatial buffering is important for maintaining the electrochemical gradients across cell membranes necessary for normal neuronal activity and for minimizing the changes in local [K+]o that occur as a consequence of neuronal activation. Membrane depolarization leads to the leak of K+ from neurons, causing [K+]o to be elevated, particularly in synaptic layers of the retina (Fig. 7.3); membrane hyperpolarization leads to reduced [K+]o as the leak conductance is reduced, but the Na+-K+ ATPase in the membranes continues to pump K+ into (and Na+ out of) cells. K+ from the ECS enters the Müller cells via inwardly rectifying K+ channels and is carried radially as an intracellular (spatial buffer) current to regions of lower [K+]o. Thus a current loop is set up: the current inside the Müller cell is carried by K+ and, to complete the circuit, the dominant extracellular ions, Na+ and Cl–, carry the extracellular return current. Because the magnitude of the [K+]o changes depends upon the integral of K+ flow rate into the ECS, ERG components that reflect this glial current will be slower than components that reflect the currents around neurons. This “slowing” would be equivalent to low-pass filtering of the neuronal signal.
The electrical properties of the Müller cell membrane are important for the creation of spatial buffer currents. The membrane is selectively permeable to K+,8,9 but the K+ conductance is not distributed evenly over the cell surface. Instead, it is concentrated in the vicinity of extracellular sinks (i.e., the vitreous body, subretinal space, and blood vessels). This regional distribution facilitates “K+ siphoning” from synaptic areas where [K+]o is high, to those regions of high K+ conductance where [K+]o is lower.10,11 In mouse retina, as indicated in Fig. 7.3, strongly inward rectifying Kir 2.1 channels have been localized to synaptic layers (small arrows on the left) where K+ moves from the ECS into Müller cells, whereas less strongly rectifying Kir 4.1 channels at the extracellular sinks allow K+ to leave the Müller cell.12
Approaches for determining the origins of the electroretinogram
Intraretinal depth recordings
A microelectrode positioned at some locus in the retinal ECS records a field potential called the “local” or “intraretinal” ERG.13 The recorded potential reflects electrical activity of the cells located near the microelectrode tip, and when a local stimulus, such as a small spot of light, is used, the local activity will be the entire signal. However, when full-field diffuse flashes are used, currents can be sufficiently large to produce a corneal ERG simultaneously with the local ERG. Local potentials with a similar timecourse to that of the corneal ERG components can be helpful in locating the cells of origin. However, this type of analysis has some complications:
1. Field potentials that spread over long distances will superimpose in space and time, making it difficult to locate the cells of origin with certainty.
2. Retinal resistivity varies between and within retinal layers,14,15 which causes currents passing through layers of different resistance to set up complex voltages.
Both of these problems occur in the intact eye when a scleral reference is used, and the local signal recorded with a microelectrode is contaminated by the diffuse ERG, due to the high resistance of the RPE and sclera. Some of this contamination can be eliminated by using a vitreal reference.16
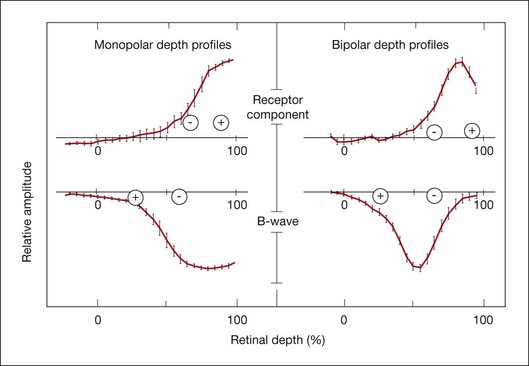
Current source density (CSD) or “source-sink” analysis can provide a solution to these problems. Local field potentials are measured and analyzed, but in addition, radial resistance is taken into account to obtain direct estimates of radial current.17 The result is a spatiotemporal profile of relatively well-localized current sources and sinks that can be compared with the retinal structure (layers) and physiology. CSD analysis has elucidated the origins of particular ERG components (e.g., for the bipolar cell origin of the b-wave).18,19
For ERG components believed to depend specifically on glial K+ spatial buffer currents, intraretinal depth recordings with ion-selective microelectrodes have been used to locate the retinal layer(s) where neurally induced changes of [K+]o were largest and most similar in timecourse to a particular component.20–25 Application of barium (Ba2+) to block Kir channels in glial cell membranes,26,27 and the ERG components dependent upon the spatial buffer currents, or genetic inactivation of Kir4.1 channels in mice,28 have been used to provide evidence for the role of Müller cells in generating certain slow waves of the ERG.
Correlation of ERG with single-cell recordings
Correlations of the ERG with single-cell electrophysiology are most useful when the light-evoked currents from a particular cell type are the primary determinant of an ERG component, as is the case for rod photoreceptors and the currents around the photoreceptor that generate the scotopic a-wave,29 or rod bipolar cells and the scotopic b-wave in mammals.30,31 Correlation also may be useful for identifying the origin of a response property, such as oscillatory potentials (OPs) in the ERG, and the light-evoked oscillatory behavior in amacrine cells as a possible source for the potentials. However, if currents from several cell types contribute to a local field potential, the relationship between field potential and local cellular responses may be difficult to determine without using other tools, such as pharmacologic agents.
Pharmacologic dissection
The use of pharmacologic agents that have specific effects on cellular functions has been very helpful in determining origins of ERG components. In Granit’s classical pharmacologic study of the dark-adapted ERG of the cat, he observed that components disappeared sequentially during induction of ether anesthesia.32 He called the components “processes” and numbered them in the order of disappearance: PI, the positive c-wave, was first to leave, then PII, the positive b-wave, disappeared, and finally, PIII, the negative a-wave. We now know that these processes correspond roughly to RPE, bipolar, and photoreceptor cell contributions to the ERG respectively. The terms PII and PIII are still used.
In recent years, much has been learned about retinal microcircuitry and biophysics, including information at cellular and molecular levels about retinal neurotransmitters (their identity, release mechanisms, and receptors), signal transduction cascades, ion channels, and other cellular proteins. This knowledge has allowed better use of pharmacologic tools in isolating ERG components and in interpreting experimental observations. For example, specific knowledge about glutamatergic neurotransmission in the retina and appropriate agonists and antagonists for specific receptors has improved our understanding of the major waves of the ERG, including the ERG in primates.33–35 Use of the voltage-gated Na+ channel blocker tetrodotoxin (TTX) has made it possible to identify ERG components resulting from the Na+-dependent spiking activity of inner retinal cells.
Modeling of cellular responses and ERG components
As our understanding of the function of retinal cell types has improved, it has been possible to develop quantitative models that predict the light responses of those cells, and to apply the models to the analysis of the ERG. Models based on suction electrode recordings from single photoreceptor outer segments have been used to predict the leading edge of the a-wave,36–38 although more proximal portions of the photoreceptor cell will also participate in its generation.39 The models have been extended to predict the leading edge of the scotopic b-wave.30 Models of stimulus–response relations of specific retinal cells can be used to analyze amplitude versus energy curves obtained from ERG measurements into components related to the different cell types.40,41
Standard ERG tests in the clinic
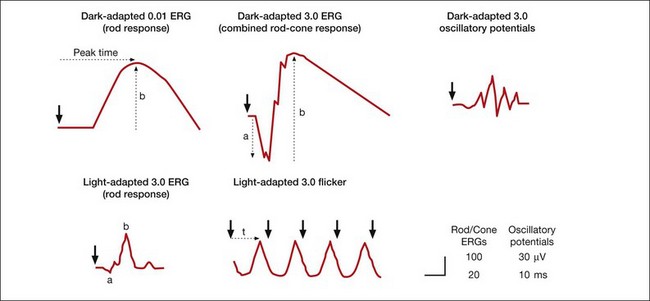
Standard and more specialized tests for use in the clinic that examine key aspects of light- and dark-adapted retinal (and more central visual) function have been described in various publications by the International Society for the Clinical Electrophysiology of Vision (ISCEV), with the most recent update for the flash and flicker ERG in 2008.42 The “standard” tests advocated by ISCEV for basic ERG testing are listed in Box 7.1, and typical responses to these tests are illustrated in Figure 7.4. The standards were developed so that ERGs recorded in clinics around the world would be comparable. The ISCEV publications, which now cover several other ERG tests as well, i.e., the ones with an asterisk in Box 7.1, describe basic technology and clinical protocols.
Box 7.1
Standard and more specialized electroretinogram (ERG) tests
Standard ERG tests described by ISCEV standard for full-field clinical electroretinography (2008 update);42 all numbers are stimulus calibrations in cd/s/m2
Dark-adapted 0.01 ERG (“rod response”)
Dark-adapted 3.0 ERG (“maximal or standard combined rod–cone response”)
Dark-adapted 3.0 oscillatory potentials (“oscillatory potentials”)
Light-adapted 3.0 ERG (“single-flash cone response”)
Light-adapted 3.0 flicker ERG (“30-Hz flicker”)
Recommended additional response: either dark-adapted 10.0 ERG or dark-adapted 30.0 ERG
Specialized types of ERG and recording procedures
Early receptor potential (ERP)
Scotopic threshold response (STR), negative and positive
Photopic negative response (PhNR)
Long-duration light-adapted ERG (ON–OFF responses)
Chromatic stimulus ERG (including S-cone ERG)
Dark and light adaptation of the ERG
Dark-adapted and light-adapted luminance response analyses
Saturated a-wave slope analysis
Distal retinal components: slow PIII, C-wave, fast oscillation trough, and light peak
After the onset of a step of light, the early waves of the dark-adapted ERG, the a- and b-waves, are followed by the c-wave and then by a succession of slower responses that include the fast oscillation trough (FOT), which is a negative deflection, and the light peak, which is a large slow positive deflection (Fig. 7.5A). Because these responses are so slow, lasting seconds to minutes, patients cannot keep their eyes steady long enough for them to develop. Therefore, in the clinic, these slower responses are generally recorded by using electro-oculography.
The electro-oculogram (EOG) is an eye movement-dependent voltage recorded between electrodes placed near the eye at the inner and outer canthus. The patient is asked to look back and forth between a pair of fixation lights separated by 30° of visual angle, situated in a ganzfeld bowl. The source of the voltage is a corneofundal potential, also called the “standing potential” that renders the cornea positive with respect to the back of the eye. Light-evoked changes in the EOG reflect changes in the transepithelial potential (TEP) of the RPE. These changes have been studied experimentally in human and animal preparations using direct current electroretinography (dc-ERG43), and these studies will be reviewed briefly here.
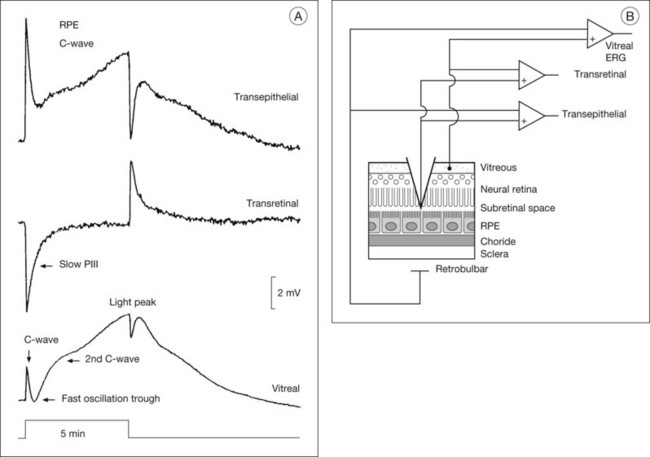
Electrogenesis of the c-wave, FOT, and light peak of the dc-ERG involves ion concentration changes in the subretinal space between photoreceptors and the RPE that in turn produce slow membrane responses in the Müller and RPE cells that face the space. The Müller and/or RPE component voltages overlap in time and sum to produce the recorded dc-ERG components. The (sub)component voltages from Müller cells and RPE have been recorded in anesthetized animals by placing a microelectrode in the subretinal space, and simultaneously recording the potentials across neural retina and the RPE, as illustrated in the schematic in Fig. 7.5B. Such experiments have provided a good understanding of the origins and mechanisms of generation of the c-wave and other slow potentials from distal retina.
c-Wave
The cornea-positive c-wave that follows the b-wave is the sum of two major (sub)component voltages: a cornea-negative voltage, generated by the neural retina, and a cornea-positive voltage of similar latency and timecourse, generated by the RPE (Figs 7.5 and 7.6). The c-wave is cornea-positive when the RPE component is larger than the neural retinal component. If the two components are equal in amplitude, the c-wave will be absent, as observed in some monkeys.44
There is long-standing evidence that two components of opposite polarity form the ERG c-wave. For example, intravenous injection of sodium iodate in rabbit, which poisons primarily the RPE, abolishes the cornea-positive c-wave and leaves a cornea-negative potential,45 as occurs in vitro when recording from an isolated neural retina preparation.46 Microelectrode recordings in retinas of several species,43 including monkey,47 have confirmed the presence of the two components. An example of such recordings in intact cat eye is shown in Fig. 7.6. The component from the neural retina is commonly termed slow PIII, to distinguish it from fast PIII, the photoreceptor current. The component from the RPE is the RPE c-wave.
Both slow PIII and the RPE c-wave are responses to the light-evoked decrease in [K+]o in the subretinal space that occurs in response to intense light stimulation of the dark-adapted retina. When measurements of [K+]o were made with ion-selective microelectrodes, either in intact eyes or in vitro preparations, the timecourse of the [K+]o decrease was found to predict that of the ERG c-wave and its component parts (Fig. 7.6). Blocking K+ conductance (via Kir channels) with various agents eliminated both the slow PIII48 and the RPE c-wave.49
Müller cell contribution (slow PIll)
Intraretinal recording at various depths50 have shown that slow PIII is generated by a radially oriented current across the neural retina. A Müller cell generator, rather than a neuronal generator, was suggested because slow PIII persisted after treatment with aspartate, a nonselective glutamate agonist, to suppress all responses of postreceptoral neurons.51
Studies in amphibia and mammals have shown that slow PIII is initiated when the distal ends of the Müller cells are passively hyperpolarized by a photoreceptor-dependent decrease in subretinal [K+]o. This sets up a transretinal “K+ spatial buffer” current,25 and the current drop across the extracellular resistance produces the slow PIII voltage.50,52,53 The slow hyperpolarization recorded in Müller cells was observed to be similar in timecourse to both the subretinal [K+]o decrease and to slow PIII.20,21,53 Further, when Ba2+ was used to block Müller cell Kir channel conductances, slow PIII was suppressed but there was little effect on the light-evoked subretinal K+ decrease.22,27,54 Finally, slow PIII was not present in ERGs of mice with Kir 4.1 channels, the dominant Kir channels in Müller cells, genetically inactivated.28
Distal versus proximal PIII
Intraretinal depth recordings in isolated rabbit retina have identified a component of similar timecourse and polarity to slow PIII that is eliminated by aspartate, and therefore, unlike slow PIII, is generated by cells proximal to the photoreceptors.55 Proximal PIII is now thought to originate from Müller cell K+ currents that flow in the same direction in the retina as slow PIII currents. However, the proximal PIII currents are initiated by an increase in [K+]o due to neuronal activation in proximal retina, rather than the decrease in [K+]o in the subretinal space. The term “proximal PIII” is not commonly used now that responses have been identified that are Müller cell, or perhaps astrocyte-mediated responses to [K+]o changes in proximal retina, e.g., STR and PhNR (described in later sections).
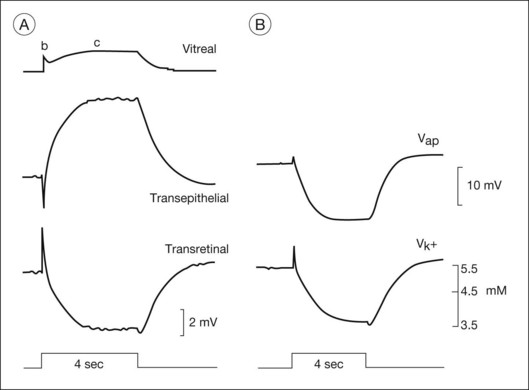
Retinal pigment epithelial component
The RPE c-wave is a cornea-positive potential that reflects an increase in the TEP of the RPE, a major component of the standing potential of the eye. The TEP exists because the apical and basal membranes of RPE cells are electrically separated by high-resistance tight junctions that encircle the monolayer of cells (the “R membrane”). The TEP is equal to the difference between the apical (Vap) and basal (Vba) membrane potentials.43 Vap is generally more hyperpolarized than Vba, making the TEP cornea-positive. During c-wave generation in response to an increase in light, the TEP increases (becomes even more positive). This is initiated by a hyperpolarization of the apical membrane, and passive shunting of current to the basal membrane, resulting in a (smaller) hyperpolarization of basal membrane, and a greater difference in potential between the two membranes.43
As was observed for Müller cells, the slow hyperpolarization of the apical membrane, with its large K+ conductance,56 and the RPE c-wave have a timecourse that is very similar to the subretinal [K+]o decrease, as illustrated in Fig. 7.6. In an isolated RPE preparation (where only the apical bath [K+] was altered), Oakley et al.57 demonstrated that the RPE c-wave was due solely to the [K+]o decrease.
The fast oscillation trough
The FOT (usually measured by EOG) is a change in the corneoretinal potential – it decreases and increases in synchrony with an alternating light/dark stimulus. The response in the dc-ERG that corresponds to the EOG decrease (trough) also is termed the FOT. The FOT response to maintained illumination follows the c-wave peak, and, when a light peak occurs, it appears as a dip between the c-wave and the light peak (Fig. 7.5A).
The FOT originates from both neural retina and RPE. It involves recovery of Müller and RPE cells from their peak polarizations as subretinal K+ reaccumulates following the reduction in concentration caused by light. This recovery may be greater than predicted by the reaccumulation, particularly for the RPE component. Light initially elicits a hyperpolarization of the apical membrane that increases the TEP and then produces a delayed basal hyperpolarization that decreases the TEP. This extra decrease in TEP underlies most of the cornea-negative potential of the FOT.43
The ionic mechanisms of the basal membrane hyperpolarization involve Cl– conductances.58 In intact sheets of RPE/choroid from human fetal eyes, the Miller lab distinguished two types of basal membrane Cl– channels: a 4,4’-diisothiocyanostilbene-2, 2’-disulfonate (DIDS)-inhibitable Ca2+-sensitive Cl– channel, and a cyclic AMP-dependent channel that is inhibited by DIDS as well as by 5-nitro-2-(3phenylpropylamino) benzoate (NPPB), which identifies it as a cystic fibrosis transmembrane regulator (CFTR) channel.59 In CF patients, the FOT, but not the light peak (see below) is reduced, implicating CFTR in generation of the FOT.
The light peak
Maintained illumination causes a slow increase in the standing potential in the dc-ERG called the light peak that can be recorded as a slow oscillation of the EOG (Fig. 7.6). Intraretinal recordings in several species,43,58 including monkey,47 have shown that this cornea-positive potential originates solely from an increase in the TEP (Fig. 7.5A). Intracellular RPE recordings localized the origin of the increase to a slow depolarization of the basal membrane caused by an increase in basal Cl– conductance. In both chick RPE and human RPE cell sheets the Cl– conductance increase was suppressed by DIDS.58,59 In mouse, the light peak is also dependent on a Cl– conductance, and it is regulated by voltage-dependent Ca2+ channel CaV1.3 subunits.60
Although the light peak voltage originates from the RPE basal membrane, it is then initiated in neural retina via the photoreceptors.61 Light stimulation leads to a change in concentration of a “light peak substance” which then affects the basal membrane via a second-messenger system. The identities of the “light peak substance” and the second messenger(s) involved in producing the light peak are unresolved. Although dopamine affected the light peak in the perfused cat eye,62 studies in chick did not support its being the “light peak substance.”61 Epinephrine also has been proposed as a candidate, and a role for ligands binding to adrenergic alpha-1 receptors on the apical membrane is likely.59 Cyclic AMP has been investigated as a second messenger in light peak generation but, as described above, it may be involved in generation of the FOT, rather than the light peak.59
Origin of the A-wave
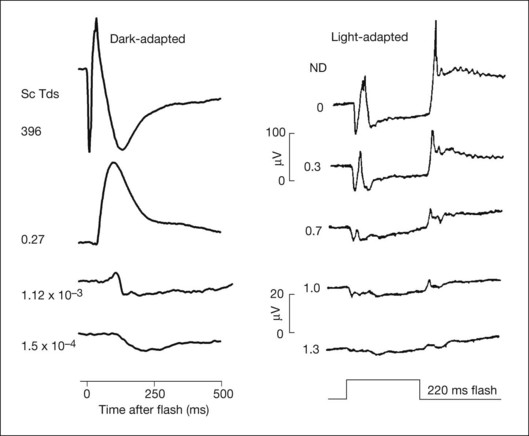
Fig. 7.7 shows the dark- and light-adapted flash ERG of macaque monkey, a good animal model for studying the origins of the human response, as illustrated in Fig. 7.1. The a-wave in the dark-adapted ERG is the initial negative wave that occurs in response to strong stimuli from darkness (Fig. 7.7, top left column) and it is primarily rod-driven (scotopic), but contains a cone contribution when flashes are very strong. When the background is rod-saturating the a-wave is cone-driven (photopic; right column). Under both dark- and light-adapted conditions, the a-wave is truncated by the rise of the positive-going b-wave that originates primarily from ON bipolar cells,30 as reviewed below. The slow negative wave in the dark-adapted ERG in response to the weakest stimuli, called the STR, is not the a-wave, but instead is initiated by amacrine and/or ganglion cell activity. This is known because STR is eliminated when postreceptoral activity is blocked pharmacologically.63,64
The a-wave as a reflection of rod and cone receptor photocurrent
Early intraretinal depth studies in intact cat eyes13,65 found that the signal at the timecourse of the a-wave was largest in the vicinity of the photoreceptors. The case for a receptoral origin for PIII was strengthened by microelectrode recordings in macaque monkey retina, with the inner retinal circulation clamped to suppress retinal activity proximal to the photoreceptors.66
Penn and Hagins67 in CSD analyses of the isolated rat retina demonstrated that light suppressed the circulating (dark) current of the photoreceptors, and proposed that this suppression was seen in the ERG as the a-wave. Figure 7.8 provides a schematic of the photoreceptor layer current from a review by Pugh et al.68 The figure shows that, in the dark, cation channels (Na+, Ca2+, and Mg2+) in the receptor outer segment (ROS) are open, current flows into the ROS (a current sink with respect to the ECS), and K+ leaks out of the inner segment (a current source), creating dipole current. This dipole current produces a corneal (and vitreal) potential that is positive with respect to the scleral side of the retina. Suppression of the dark current reduces the cornea-positive potential, creating the negative-going a-wave. Consistent with this view, as illustrated in Fig. 7.9, intraretinal recordings and CSD analyses in the intact macaque retina have localized current sources and sinks for a local potential, corresponding to the a-wave, to the distal third of the retina.18
Hood and Birch38 sought to relate the timecourse of the leading edge of the ERG a-wave directly to the leading edge of the photoreceptor outer-segment response to light. They demonstrated that both the linear and the nonlinear (i.e., saturating) behavior of the leading edge of the a-wave in the human ERG could be predicted by a model of photoreceptor function derived from in vitro suction electrode recordings of currents around the outer segments of primate rod photoreceptors.69 Subsequently a simplified kinetic model of the leading edge of the photoreceptor response (in vitro current recordings) that took into account the stages of the biochemical phototransduction cascade was developed by Lamb and Pugh.70 This model could be fit to the leading edge of the human a-wave generated by strong stimuli and it has been used in noninvasive studies of human rod36,71 and cone photoreceptor72 function. Recent analyses suggest that the a-wave generated in response to strong stimuli includes additional currents associated with more proximal regions of the photoreceptor cell that form a sharp “nose” on the leading edge of the response that quickly relaxes to the level of the photocurrent.39
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree