Chapter 21 Dynamic Risk Group Analysis for Differentiated Thyroid Cancer
Introduction
As described in detail in Chapters 17, Molecular Pathogenesis of Thyroid Neoplasia, 18, Papillary Thyroid Cancer 19, Papillary Thyroid Microcarcinoma, 28, Chernobyl and Radiation-Induced Thyroid Cancer, and 29, Familial Nonmedullary Thyroid Cancer, a relatively small set of clinical-pathologic factors has been used in a wide variety of staging systems (e.g., TNM AJCC, MACIS, AMES, AGES) to predict the risk of death in differentiated thyroid cancer.1,2 The most important of these factors include age of the patient at diagnosis, histology, size of the tumor, completeness of resection, and the presence of metastatic disease at presentation. However, in clinical practice, comprehensive initial risk stratification requires a review of all available data that are usually obtained as part of initial diagnosis and therapy (Table 21-1). Integration of all of these factors allows for an initial estimation of the risk of death and the risk of recurrence.
Table 21-1 Important Factors to Be Used in Initial Risk Stratification
| Preoperative Findings | Physical examination Vocal cord function Cross-sectional imaging (if obtained) |
| Intraoperative Findings | Presence of gross extrathyroidal extension Involvement of major structures in the neck Completeness of tumor resection Lymph node metastases |
| Pathology Findings | Specific histology Vascular invasion Significant extrathyroidal extension Molecular characterization |
| Laboratory Findings | Postoperative serum thyroglobulin |
| Nuclear Medicine Findings | RAI scans FDG PET scans |
However, although each of the major staging systems can risk stratify with respect to disease-specific mortality, they are much less effective at predicting the risk of recurrence.3 Additionally, these staging systems fail to adequately incorporate the impact of initial therapy (other than completeness of resection).4 And because it is quite likely that our initial therapies have a major impact on the final outcome, the failure to incorporate these treatment variables into the commonly used staging systems will result in an inappropriately low estimate of risk in the few “low-risk” patients who do not respond to initial therapy and also an inappropriately high estimate of risk for the few “high-risk” patients who have an excellent response to initial therapy.
But more important, the current staging systems present a static representation of the patient at the time of presentation that does not change over time.5,6 Clearly in clinical care, our risk estimates change over time depending on the response to therapy for individual patients. For example, a “low-risk” patient who develops a rapidly rising thyroglobulin over time or new pulmonary nodules would forever be classified as “low risk” based on initial staging when in reality the patient is now at increasing risk of developing structurally identifiable progressive disease and his or her risk of death from thyroid cancer is increased by virtue of these new clinical findings. It is both the failure to account for the effect of initial therapy and the static nature of our current staging systems that result in the relative inability of these staging systems to guide long-term follow-up recommendations.
The importance of accurate, ongoing risk stratification cannot be overemphasized. In fact, many of the major recommendations concerning the most important clinical aspects of patients care provided in the updated ATA guidelines on the management of thyroid cancer and thyroid nodules are based on an assessment of individualized risk (Table 21-2).7 Therefore, implementation of the American Thyroid Association (ATA) guidelines into clinical practice requires a thorough understanding of the risk of death and risk of recurrence for each individual patient we evaluate and treat.
Table 21-2 ATA Cancer Recommendations Relating to Individual Risk
| ATA Recommendation | Management Issue |
|---|---|
| 26 | Extent of initial surgery |
| 27 | Indications for prophylactic central neck dissection |
| 29 | Indications for completion thyroidectomy |
| 31 | Initial staging systems to be used |
| 32 | Indications for RAI ablation |
| 37 | Selecting dose for RAI ablation |
| 40 | Goal TSH |
| 41 | Indications for external beam radiation |
| 45 | Follow-up approach to low-risk patients |
| 46 | Indications for diagnostic whole body radioiodine scan (WBS) in low-risk patients |
| 47 | Indications for diagnostic WBS in high-risk patients |
| 48 | Frequency, indications for follow-up ultrasound |
| 49 | Goal TSH in follow-up |
| 59 | Indications for systemic therapy/additional therapies |
| 77 | Management of persistent disease |
Initial Risk Stratification
Although initial risk stratification using the current staging systems has some limitations, it nonetheless provides a starting point for ongoing risk stratification. We agree with the recommendations provided in the updated ATA guidelines on the management of thyroid cancer and thyroid nodules which recommend using both (1) the AJCC TNM system to estimate the risk of death from thyroid cancer and (2) a separate postoperative clinical-pathologic staging system to improve predictive ability and to plan follow-up for patients with differentiated thyroid cancer.7 Because none of the commonly used initial staging systems adequately predicts the risk of recurrence, the ATA guidelines recommended a reasonable, but unproven, three-tiered staging system to estimate risk of recurrence7 (see Box 21-1 and Chapter 18, Papillary Thyroid Cancer).
Box 21-1 ATA Risk of Recurrence Group 2009
Low-Risk Patients
• No local or distant metastases
• All macroscopic tumor has been resected
• No tumor invasion of locoregional tissues or structures
• Tumor does not have aggressive histology (e.g., tall cell, insular, columnar cell carcinoma) or vascular invasion
• If 131I is given, there is no 131I uptake outside the thyroid bed on the first posttreatment whole-body RAI scan (RxWBS)
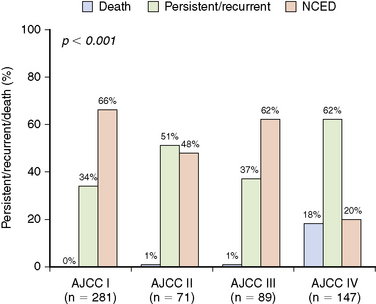
We recently evaluated the ability of both the AJCC TNM system and the newly proposed ATA risk of recurrence stratification system to predict the risk of recurrence in a cohort of 588 patients with differentiated thyroid cancer followed at our center for a median of 7 years after total thyroidectomy and RAI ablation.8 Consistent with previous reports,9,10 the AJCC TNM system does not adequately risk stratify with respect to risk of recurrence or risk of biochemical or structural persistent disease (Figure 21-1). As would be expected, 26 of the 28 patients who died in this cohort were AJCC stage IV (1 stage II and 1 stage III patient also died of disease). However, the risk of having persistent or recurrent disease was very similar across AJCC stages I, II, and III. So although AJCC staging is useful for predicting risk of death, it does not provide adequate predictions regarding the risk of recurrence or persistent disease, which is required to plan follow-up studies.
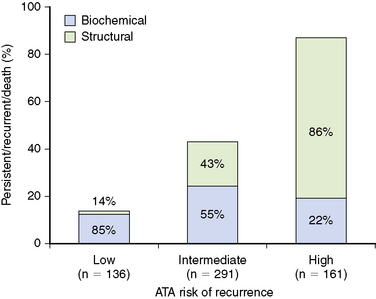
However, the ATA three-tiered system for predicting risk of recurrence performed much better. In our cohort, 23% of the patients were classified as low risk, 50% intermediate risk, and 27% high risk for recurrence based on the ATA system.8 As can be concluded from Figure 21-2, patients classified as low risk have a 14% risk of having persistent or recurrent disease, whereas intermediate-risk patients have a much higher likelihood (44%) of having persistent or recurrent disease. As would be expected, the high-risk patients have an 86% likelihood of having persistent or recurrent disease. Interestingly, the persistent/recurrent disease in the low-risk patients was a biochemical diagnosis (thyroglobulin) without structural correlate in 85% (see Figure 21-2). However, the persistent/recurrent disease was associated with a structural correlate in 45% of the intermediate-risk group and 78% of the high-risk group. Therefore, the ATA risk stratification system identifies both the risk of having persistent/recurrent disease and the likelihood that the recurrent/persistent disease will be either a biochemical or structurally evident recurrence.
Response to Therapy Assessment
Although these initial risk assessments provide the basis for ongoing risk stratification, it is clear that new data obtained during follow-up could significantly alter these initial risk estimates.6 Table 21-3 provides a sample of the types of data that are used to modify our initial risk estimates.12–20 Regardless of initial AJCC TNM stage or ATA risk group classification, rising values of serum Tg or Tg antibodies would be expected to increase the risk of developing progressive or newly identifiable structural disease. Conversely, declining Tg values, undetectable stimulated Tg values, negative neck ultrasonography, and other cross-sectional imaging would all tend to decrease the risk of disease recurrence and death.
Table 21-3 Response to Therapy Variables
| Change in serum thyroglobulin over time Change in serum Tg antibodies over time Results of stimulated thyroglobulin determinations Results of follow-up neck ultrasound Results of RAI scanning Other cross-sectional imaging Results of FDG PET imaging Physical examination |
We had previously proposed a response to therapy assessment scheme that classifies patients as having an excellent, acceptable, or incomplete response to initial therapy (Table 21-4).5,8 In this system, the response to therapy for each patient is determined on the basis of the standard follow-up testing that is usually done in differentiated thyroid cancer. Patients with an “excellent response” to therapy have no evidence of disease with negative cross-sectional imaging and undetectable stimulated thyroglobulin values. RAI scanning, if done, shows only normal uptake in the thyroid bed without evidence of metastatic disease. We classified patients as having an “acceptable response” to therapy if they had low-level persistent thyroglobulinemia (suppressed Tg < 1 ng/mL, stimulated Tg < 10 ng/L), or nonspecific changes on cross-sectional imaging, that though not abnormal, were not completely normal. We used the term acceptable response because in our practice these patients are followed with cautious observation and are not immediately retreated. Finally, patients who clearly failed our initial therapies were classified as having an “incomplete response.” These patients had persistently elevated or rising serum Tg during the first 2 years of follow-up or had new structural disease identified within that same time period. We reasoned that regardless of initial risk classification, patients who had excellent response to therapy should be at a lower risk of recurrence/persistent disease than what had originally been predicted.
Table 21-4 Categories of Response to Treatment
| Excellent Response | Acceptable Response | Incomplete Response |
|---|---|---|
| All the following | Any of the following | Any of the following |
| Suppressed and stimulated Tg < 1 ng/mL Neck ultrasonography without evidence of disease Cross-sectional or nuclear medicine imaging negative (if performed) | Suppressed Tg < 1 ng/mL and stimulated Tg ≥ 1 and < 10 ng/mL Neck ultrasound with nonspecific changes or stable subcentimeter lymph nodes Cross-sectional or nuclear medicine imaging with nonspecific changes, although not completely normal | Suppressed Tg ≥ 1 ng/mL or stimulated Tg ≥ 10 ng/mL Rising Tg values Persistent or newly identified disease on cross-sectional or nuclear medicine imaging |
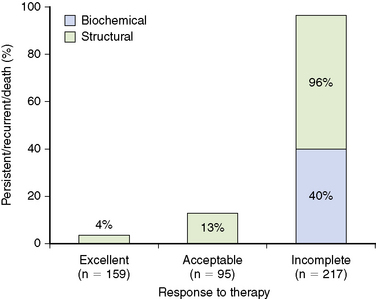
Using the 471 patients who had stimulated Tg values available from same follow-up cohort of 588 patients with differentiated thyroid cancer followed at our center for a median of 7 years after total thyroidectomy and RAI ablation described earlier, we validated our proposed response to the therapy staging system.8 As Figure 21-3 shows, regardless of initial risk stratification, patients demonstrating an excellent response to therapy have only a 4% likelihood of having persistent/recurrent disease, whereas patients who have an acceptable response to therapy have a 13% risk of having persistent/recurrent disease (in both cases, most of the persistent/recurrent disease was biochemical without structural correlate). As expected, the incomplete response to therapy patients had a 96% chance of having persistent/recurrent disease with over 50% chance of having structurally identifiable disease.
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree