CHAPTER 123 Disorders of the Thyroid Gland
Physiology of the Thyroid Gland
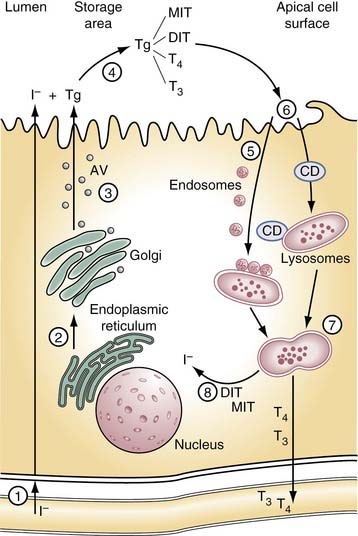
The thyroid gland produces two hormones, 3,5,3′-triiodothyronine (T3) and 3,5,3′,5′-tetraiodothyronine or thyroxine (T4). Both are iodinated derivatives of tyrosine. Hormone production depends on an external iodine supply and on intrathyroidal mechanisms for concentrating ingested iodide and then incorporating it into the tissue-specific protein, thyroglobulin. The thyroid gland is unique within the endocrine system in having a large extracellular space, the follicular lumen, which is used for storage of the hormones and their precursors. As hormone is needed by the organism, thyroglobulin is retrieved by the cell, where the biologically active hormones are released from thyroglobulin before being passed into circulation (Fig. 123-1).
Iodide Transport
A daily dietary intake of at least 100 µg of iodine per day is required in humans to ensure adequate production of thyroid hormone. In North America, the average daily intake is higher than this, largely because of the use of iodine as a food additive.1 In many parts of the world, however, consumption is significantly below the minimum level, and iodine deficiency is the leading cause of thyroid-related disorders.
The thyroid normally concentrates iodide 20-fold to 40-fold over the extracellular space and against an electrical gradient of approximately 40 mV. Key to this trapping action is a protein located in the basal membrane of the thyroid cell known as the sodium/iodide symporter (NIS).2 NIS couples with the influx of Na+ down its electrochemical gradient with the simultaneous influx of I− up its electrochemical gradient. An Na+,K+-ATPase acts to maintain the Na+ gradient. Iodide travels down its electrochemical gradient to the apical surface of the thyrocyte, where it is incorporated into thyroglobulin. More recent evidence suggests an apical membrane protein, pendrin, aids in releasing iodide into the follicular lumen.3 Mutations in the gene and coding for this protein are responsible for the common hereditary disorder Pendred’s syndrome, which is associated with mild hypothyroidism, goiter, and hearing loss.3 Mutations in the gene coding for NIS have been identified in patients with iodide trapping defects, a rare cause of congenital hypothyroidism.2
Thyroglobulin
Thyroglobulin is essential to thyroid physiology. It is a tissue-specific protein that serves as a matrix for the synthesis of hormone and as a vehicle for its storage.4 The human thyroglobulin gene has been cloned and is located on the long arm of chromosome 8Q24. Thyroglobulin is a large dimeric glycoprotein of approximately 660 kD, consisting in humans of two identical polypeptide chains each of 2750 amino acids. About 10% of its weight is carbohydrate, and about 0.1% to 1% is iodine. Synthesis and maturation of thyroglobulin follow a pathway typical of proteins destined for secretion. The polypeptide chain is synthesized on the surface of the rough endoplasmic reticulum. As that passes through a series of intracellular compartments, it undergoes important post-translational modifications before reaching the follicular lumen.5 Carbohydrate units are added to the polypeptide chain as it is translocated into the lumen of the rough endoplasmic reticulum. Folding and dimerization of the polypeptide chain occurs within this compartment, aided by folding enzymes and a group of proteins known as molecular chaperones. Perturbations of this process result in block of protein transport beyond this point and cause congenital hypothyroidism.5 Under normal circumstances, the properly folded thyroglobulin dimers migrate to the Golgi complex, where processing of the carbohydrate units are completed. Mature but as of yet uniodinated thyroglobulin is transferred from the Golgi complex to the apical cell surface in small vesicles.
Iodination and Thyroperoxidase
Newly formed thyroglobulin and iodide meet at the apical cell surface, where hormone synthesis occurs. This process includes (1) the oxidation of iodide; (2) its subsequent transfer to thyrosyl residues on thyroglobulin, producing monoiodotyrosine (MIT) and di-iodotyrosine (DIT); and (3) coupling of two iodotyrosine molecules, either one each of MIT and DIT to form T3 or two of DIT to form T4. Thyroperoxidase (TPO), an enzyme present in the apical cell membrane, is responsible for each of these steps.6 Hydrogen peroxide (H2O2), required in the iodinating and coupling reactions, is generated at the apical membrane by a reduced nicotinamide adenine dinucleotide phosphate (NADPH) oxidase.7 Mutations in the TPO gene have been found in patients with congenital hypothyroidism caused by defective organification. Abnormalities in H2O2 generation seem to be more rare.
Under normal circumstances, iodide, when trapped, is rapidly incorporated into thyroglobulin so that little free iodide exists within the thyroid gland at any given time. The extent to which thyroglobulin is iodinated depends on the thyroid’s iodide supply. At a level of 0.5% iodine, the thyroglobulin dimer in humans contains on average 5 residues of MIT, 5 of DIT, 2.5 of T4, and 0.7 of T3 of a total of 132 residues of tyrosine.4
Hormone formation involves the coupling of two residues of iodotyrosine within the thyroglobulin polypeptide chain. At the hormonogenic site, the “acceptor” DIT receives the iodinated phenol ring of the “donor” iodothyrosyl (either MIT or DIT) located at some distal site on the polypeptide chain. In the process, the alanine side chain of the donor remains behind, now presumably in the form of hydroalanine. Iodination in vitro of low iodine human thyroglobulin indicates that certain thyrocele sites are favored for early iodination, and that three or four major sites exist for hormone formation. The most important hormonogenic site is located five residues from the amino terminal thyroglobulin, whereas a second major site is located three residues from the carboxy terminal. The locations of donor thyrosyls are incomplete. To date, only one has been identified in human thyroglobulin, and this resides in the amino terminal region of the molecule.4
Storage and Release of Hormone
Hormone release is initiated by the retrieval of thyroglobulin from the follicular lumen. Under stimulatory conditions in some species, this process may occur by macropinocytosis. Pseudopods form at the thyrocytes’ apical surface and engulf thyroglobulin as large colloid droplets. Under physiologic conditions in most species, however, including humans, thyroglobulin is retrieved by micropinocytosis into small vesicles. It is then passed through the endosome-lysosomal system, where the combined action of several acid proteases, including cathepsins B, D, and L, and lysosomal dipeptidase 1, releases the hormones and their iodotyrosine precursors from the polypeptide backbone.4 Evidence suggests the iodo-amino acids may be preferentially cleaved first, but ultimately thyroglobulin is broken down into amino acids or small peptides within the lysosomes.
When released from thyroglobulin, the thyroid hormones and their precursors enter the cytosol; there MIT and DIT are deiodinated by an iodotyrosine-specific deiodinase, and the released iodide re-enters the iodine pool. Some T4 is deiodinated to T3 before it is released into the circulation by 5′-iodothyronine deiodinase similar to that found in peripheral tissue.8 The mechanism by which T4 and T3 are released from the thyrocyte is unknown, but more recent evidence suggests a carrier protein may be involved.9
Circulating Thyroid Hormones
Less than 1% of circulating thyroid hormones exist as free iodo-amino acids. The remainder are bound in reversible, noncovalent linkage to one of several plasma proteins.10 In humans, the most important of these is thyroxine-binding protein (TBG), accounting for approximately 70% of circulating hormone. The TBG molecule has one hormone-binding site with a very high affinity for T4 and lower affinity for T3. A second plasma protein transthyretin accounts for approximately 10% of circulating T4 and T3. Each transthyretin molecule has two hormone-binding sites, but the affinity of the first is lower than that of TBG, and that of the second site is very low for both hormones. Albumin also serves as a thyroid hormone transport protein. Although it has low affinity, its abundance allows it to account for 10% to 20% of bound circulating hormone.
The bound hormones are in equilibrium with the minute fraction of free circulating hormone that is available for use in peripheral tissue. Under euthyroid conditions, approximately 0.2% of T4 and about 0.3% of T3 in circulation is unbound. The larger free-to-bound ratio of T3 to T4 is caused by the lower affinity of TBG for T3.11 To date, no change in thyroid state has been attributed to abnormalities in these hormone-binding proteins, despite their apparent role in thyroid function homeostasis.
Metabolism of Thyroid Hormones
T4 must first be deiodinated to T3 to exert most of its biologic actions. Because little T3 is directly synthesized on thyroglobulin, this transformation becomes an important step in hormonogenesis. Three iodothyronine deiodinases are present in mammals.8 These are membrane-bound enzymes that are closely related structurally and are distinguished by the presence of selenocysteine at their active sites. Each has distinctive substrate preferences, activity characteristics, inhibitor sensitivities, and relative tissue specificity. Type I and II deiodinases, through their combined action, are responsible for generating approximately 80% of the total T3 production.
Type I deiodinase is the primary source of circulating T3 and is found in liver, kidney, and thyroid (where it is activated by TSH) tissues, and to a lesser extent in other tissues. Type I deiodinase is positively regulated by thyroid hormones and is greatly reduced under pathophysiologic states, such as starvation and nonthyroidal illnesses. It is inhibited by the antithyroid drug propylthiouracil. Type II deiodinase is present primarily in the central nervous system, the pituitary, the placenta, and the skin, and has more recently been found in the thyroid.12 Its major role is thought to be in the local production of T3, but it may also contribute to circulating T3. In contrast to type I deiodinase, the type II enzyme is negatively regulated by thyroid hormone and is unaffected by propylthiouracil. Type III deiodinase inactivates T4 and T3 by inner ring deiodination in the five position, forming reversed T3. The enzyme is present in the adult brain, skin, and placenta, and is present in high levels in fetal tissues, where it is thought to be important in protecting developing tissue from excess levels of thyroid hormone.13
Control of Thyroid Function
The anterior pituitary is the primary internal regulator of thyroid function, influencing virtually all phases of thyroid metabolism.14 It secretes TSH, also known as thyrotropin, which is a 28- to 30-kD lipoprotein consisting of two subunits, α and luteinizing hormone. The α subunit is common to the pituitary hormones follicle-stimulating hormone and luteinizing hormone and to chorionic gonadotropin. The α subunit is unique to TSH, however, and is responsible for the binding of the hormone to its receptor in the basal membrane of the thyroid cell. On interaction with TSH, the receptor, a member of a family of G protein–coupled receptors, undergoes conformational changes that activate one or two regulatory pathways. Most TSH effects are mediated by the activation of the cyclic adenosine monophosphate (cAMP) pathway; others involve the Ca2+/phosphatidylinositol cascade. The pathway used to elicit a given effect may vary among species. TSH stimulates the efflux of iodide into the follicle and the resorption of colloid into the cell within minutes. Later effects include increased expression of the NIS, thyroglobulin, and TPO genes; stimulation of H2O2 production; promotion of glycosylation; and increased production of T3 relative to T4.
Circulating levels of TSH are controlled by the opposing influences of thyroid hormone and of thyrotropin-releasing hormone (TRH) from the hypothalamus.15 The latter is a modified tripeptide secreted to the anterior pituitary by way of the hypothalamohypophyseal portal system. TRH binds to the plasma membrane of the thyrotrope and stimulates the release of TSH and the expression of its gene. Levels of circulating TSH are under strict control by the thyroid in a classic negative feedback system. As levels of thyroid hormone increase in response to TSH stimulation, T4 and T3 block the TRH-stimulated release of TSH in the thyrotrope. The thyroid hormones also act indirectly by inhibiting TRH gene expression in the hypothalamus.
Withdrawal of iodide from the diet leads to a rapid decrease in serum T4 and an increase in serum TSH. Serum T3 levels initially are unaffected, but eventually decline with prolonged withdrawal. In response to TSH stimulation, the thyroid increases iodide uptake and organification, alters the distribution of iodo-amino acids within thyroglobulin by increasing the ratios of MIT to DIT and T3 to T4, and increases the intrathyroidal conversion of T4 to T3 by type I and II deiodinases.8 With prolonged iodine deficiency, TSH-stimulated cell proliferation eventually leads to goiter.
Antithyroid Agents
Antithyroid drugs can inhibit thyroid hormone synthesis secretion or metabolism.16 Common agents and their major actions are summarized in Table 123-1. Numerous agents used in the treatment of nonthyroidal illnesses may have profound effects on thyroid hormone production. Notable among these are the iodinated radiocontrast agents that are potent inhibitors of thyroid hormone deiodination, and can interfere with hepatic uptake of T4 and binding of T3 to nuclear receptors.17 The antiarrhythmic agent amiodarone, which is also heavily iodinated, elicits similar alterations in thyroid hormone metabolism and action. Lithium, used in the treatment of bipolar illness, is a potent inhibitor of thyroid hormone release and acts by blocking thyroglobulin endocytosis.18
Table 123-1 Drugs Used in Hyperthyroidism
| Drug | Usual Starting Dose |
|---|---|
| Propylthiouracil | 200 mg PO tid |
| Methimazole | 20 mg PO bid |
| Propranolol | 10-40 mg PO qid |
| Saturated solution of potassium iodide (SSKI) | 1-2 drops PO qd-tid |
| Compound solution of iodine (Lugol’s solution) | 2-5 drops PO qd-tid |
| Dexamethasone | 2 mg PO qid |
| Prednisone | 40-60 mg po qd |
| Ipodate | 1 g PO qd |
| Lithium | 300-450 mg PO tid |
| Perchlorate | 1 g PO qd |
| Cholestyramine | 2-4 g PO bid-qid |
| Colestipol | 5 g PO one to five times a day |
| Octreotide | 50-100 µg SC bid-tid |
| Diltiazem | 120 mg PO tid |
Thyroid Hormone Mechanism of Action
The thyroid has multiple effects on development, growth, and metabolism. The effects on development are widespread phylogenetically and can be dramatically observed during the course of amphibian metamorphosis. The appropriate levels of thyroid hormone during fetal and neonatal stages in humans are crucial for the normal maturation of the central nervous system, muscle, bone, and lung. In severe cases of thyroid hormone deficiency during this period, the syndrome of cretinism results with its associated mental retardation, deafness, mutism, and stunted growth.19 Similarly, an excess of thyroid hormone during these critical developmental periods can result in neurologic abnormalities. The metabolic effects of thyroid hormone seem to be confined to birds and mammals, presumably evolving in response to the increased metabolic pressures of thermogenesis. Oxygen consumption and the metabolism of proteins, carbohydrates, and fats all are under thyroid hormone control.
Most effects of thyroid hormone are now believed to be exerted by interactions with specific nuclear thyroid hormone receptors, resulting in the altered expression of specific genes.20 T4 has little affinity for the nuclear receptors and first must be converted to T3 to be effective. The receptors themselves belong to a large superfamily of nuclear receptors, which includes the steroid hormones, retinoic acid, and vitamin D. The thyroid hormone receptors are closely related isoforms, despite being encoded by two different genes (α and β).
Thyroid Function Studies
Circulating Thyroid Hormone Measurement
Elevated total T4 levels may also occur when there is production of endogenous antibodies to T4, especially in patients with Hashimoto’s thyroiditis or other autoimmune disorders, and occasionally in patients with Waldenström’s macroglobulinemia associated with a benign monoclonal gammopathy.21 Another condition of elevated total T4 level is peripheral resistance to thyroid hormone. Individuals with this condition may have goiter, and they may be hyperactive.22 Patients with this disorder are euthyroid. Although rarely found, this disorder has led to inappropriate treatment for hyperthyroidism.
The most widely used measurement of thyrometabolic status is measurement of serum free T4 by equilibrium dialysis.23 When measured by the dialysis method, free T4 is not affected by changes in binding protein concentrations or by nonthyroidal illness. This method is cumbersome and expensive, and it is not routinely performed. Commercial free T4 levels are most commonly measured by immunoassay techniques, but their reliability is suboptimal because they may be affected by illness or significant changes in binding proteins.24 The clinical usefulness of free T4 measurements by any method may be limited.25
Although the thyrometabolic status is best reflected by the free T4 level, from a clinical standpoint, an index or estimate of free T4 is generally adequate. The free T4 index is obtained by multiplying the serum total T4 and an indirect assessment of thyroglobulin. Serum thyroglobulin is generally estimated by one of two methods—the thyroid uptake test and the T3 uptake test.25 The thyroid uptake test is directly proportional to thyroglobulin levels in serum, whereas the T3 uptake test is inversely proportional to thyroglobulin levels.26 The result, by use of either method, is that variances in serum thyroglobulin levels are largely eliminated, and the calculated free T4 index accurately reflects actual free T4 status. Extreme changes in thyroglobulin levels, or the presence of the severe nonthyroidal illness, may result in poor correlation between calculated and measured free T4 levels.
The principles used for obtaining the serum T3 are to determine the severity of hyperthyroidism and to confirm the diagnosis of suspected thyrotoxicosis in cases in which serum T4 levels are normal or equivocal. In addition, the serum T3 may be indicated in evaluating patients with autonomously functioning thyroid adenomas, in whom so-called T3 toxicosis may be present. Such patients may have normal or borderline elevated serum T4 levels along with suppressed serum TSH levels.27
Serum Thyrotropin Measurement
Until approximately 10 years ago, virtually all clinical TSH assays were performed by radioimmunoassay. By the mid-1980s, many commercial laboratories began using more sensitive immunometric TSH methods with either monoclonal or polyclonal antibodies. Functional sensitivity of these assays represented a 10-fold improvement in sensitivity over radioimmunoassay methods. More recently, nonisotopic immunometric TSH assays have been developed with a chemiluminescent label. These newer assays have a 10-fold greater sensitivity than the early immunometric TSH assays and are 100 times more sensitive than radioimmunoassay methods. These latest TSH assays, with a sensitivity of 0.01 mU/L, are currently termed third-generation TSH assays and represent the most sensitive method for detecting TSH level.28
The clinical application of TSH detection may be summarized as follows:
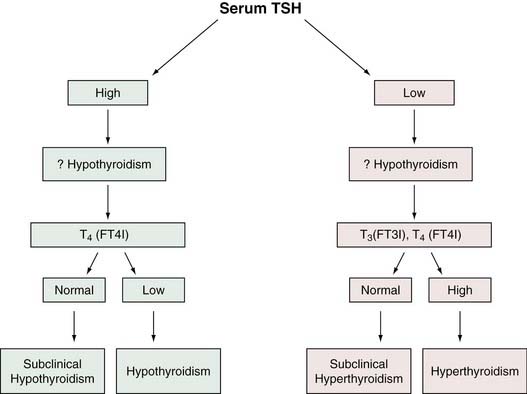
The fact that serum TSH is abnormal in hypothyroidism and hyperthyroidism would seem to make it ideally suited as a screen of thyroid status because, with rare exceptions, a normal TSH level would suggest normal thyroid hormone homeostasis. Experience in ambulatory individuals suggests that a normal TSH virtually excludes the possibility of thyroid dysfunction.34 In addition, the serum TSH level is more sensitive than the serum T4 level as a test for thyroid dysfunction because TSH can detect subclinical thyroid disorders in which serum total T4 (and T3) is usually normal. As a result of advances in TSH methodology, measurement of circulating thyroid hormones may become assigned to a second line of assessment of suspected thyroid dysfunction. Many investigators believe that the serum TSH is preferable as a screening method for thyrometabolic status.11,29 Figure 123-2 is an algorithm for the use of TSH level in the evaluation of thyroid function.
Serum Thyroglobulin Measurement
Thyroglobulin is elevated in the serum of patients with nearly all types of thyroid disorders, limiting its usefulness as a diagnostic test. Its greatest clinical value is in managing patients with well-differentiated thyroid carcinoma. An elevated or increasing thyroglobulin level (after initial surgical and ablation therapy) suggests persistence or recurrence of tumor.35 Thyroglobulin is measured by either radioimmunoassay or immunometric technique. Although antithyroid antibodies may cause interference with accurate thyroglobulin measurement in 10% of individuals, in these patients measurements of thyroglobulin and antithyroglobulin antibodies may be used concurrently to provide information regarding the tumor status.35
Thyroid Antibody Status
Circulating antithyroid antibodies, specifically antimicrosomal (AMA) and antithyroglobulin (ATA) antibodies, are usually present in patients with autoimmune thyroid disease.36 Since the introduction of immunoassay techniques, the term antithyroperoxidase (anti-TPO) has become interchangeable with AMA. AMAs are detectable in more than 90% of patients with chronic autoimmune thyroid disease; nearly 100% of patients with Hashimoto’s thyroiditis and more than 80% of patients with Graves’ disease have positive titers.37 Although ATAs are more specific than AMAs, they are less sensitive, and they are not as useful in the detection of autoimmune thyroid disease.38 Elevated levels of AMA are also frequently positive in various other organ-specific autoimmune diseases, such as lupus, rheumatoid arthritis, autoimmune anemia, Sjögren’s syndrome, type 1 diabetes mellitus, and Addison’s disease.39
Approximately 15% of adults (especially women) in the United States have elevated AMA titers.31 Prevalence of positive AMA titers increases with age, as does the incidence of primary hypothyroidism. The presence of a positive AMA titer should alert one to the possibility of hypothyroidism. Individuals with positive AMA and elevated TSH levels, even with normal serum total T4 levels (subclinical hypothyroidism), had a 3% to 5% per year likelihood of clinical hypothyroidism developing.40 In this manner, determination of AMA levels may be useful in the diagnosis of individuals with suspected autoimmune thyroid disease and in providing prognostic information when used in conjunction with TSH levels.
Thyroid-Stimulating Antibody Measurement
The immunopathogenesis of Graves’ disease was first suspected in the mid-1950s, when it was observed that injecting sera of patients with Graves’ disease into rats produced a prolonged uptake of radioactive iodine in the rat thyroid glands. The term long-acting thyroid stimulator (LATS) was coined.41 Later, LATS was characterized as a 7S immunoglobulin, and in recent years several assays have been developed for the detection of LATS, or thyroid-stimulating antibodies. Two methods are commonly used; one depends on generation of cAMP, and the other is a radioreceptor method that relies on the TSH binding inhibitory properties of the immunoglobulin. The cAMP-generating assay is termed thyroid-stimulating immunoglobulin, and it is detectable in 90% to 95% of hyperthyroid patients with Graves’ disease. The other assay detects stimulating and blocking antibodies termed TBII; it is detected in 85% of patients with hyperthyroid Graves’ disease.42 Thyroid-stimulating antibody measurements are not indicated for the routine diagnostic evaluation of suspected Graves’ disease, but they may be useful when the diagnosis of Graves’ disease is not evident.