megalocornea or anterior megalophthalmos, an X-linked recessive trait that consists of megalocornea, iris and angle abnormalities, and lens subluxation with early cataract formation; and (c) buphthalmos in infantile glaucoma (3). In keratoglobus, the protuberant thin cornea appears enlarged clinically but usually has a normal diameter. There seems to be no entity of “megaloglobus” in which the entire globe is congenitally enlarged with a normal intraocular pressure.
glaucoma, spherophakia and/or lens subluxation), which requires lifelong annual examinations for early detection, and cataracts, which often appear in the fourth decade and may require the use of vitrectomy-type instruments during extraction, because the lenses are subluxated or dislocated (6,7). Due to enlarged anterior segment, custom-designed intraocular lenses may be required for lens stability and visual rehabilitation (8).
Table 10.1 DIFFERENTIAL DIAGNOSIS OF ENLARGED CORNEA | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
may produce focal stromal edema (acute hydrops) and heal spontaneously in weeks to months (Fig. 10.8B). Minor blunt trauma to the eye or to the head may rupture the thin cornea and sclera, so the ophthalmologist must counsel the parents of these children to provide a safe environment and protective spectacles or eye guards. Amblyopia is often severe because of the high myopia, a problem diminished by carefully fitted spectacles or contact lenses. A scleral lens may be tried if contact lenses are not successful. In patients with severe thinning and anterior bulging of the cornea, surgical treatment may be contemplated. Given the diffuse corneal thinning to the limbus, surgical repair is problematic at best. A large limbus-to-limbus onlay lamellar keratoplasty or epikeratoplasty to both reinforce the corneal integrity and provide a more normal curvature can be performed. A “tuck-in” lamellar keratoplasty (central lamellar keratoplasty with intrastromal tucking of the peripheral flange) is another technique that can be performed in these patients. If the central cornea is scarred, typically from a previous episode of hydrops, a subsequent central visual penetrating keratoplasty can be performed (9,10,11,12,13,14).
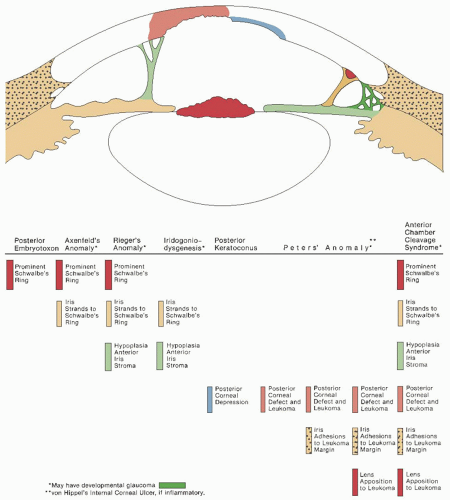
not seem to be appropriate in the description of abnormalities that occur during embryogenesis and are of neural crest differentiation (22,23,24,25).
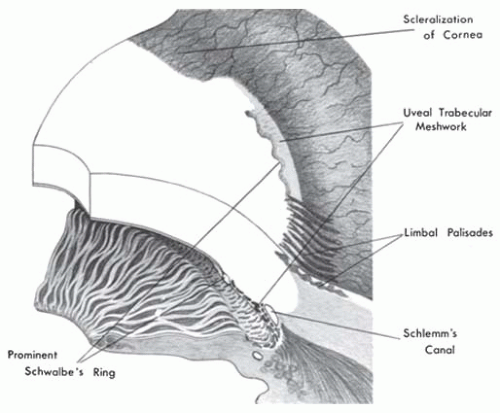
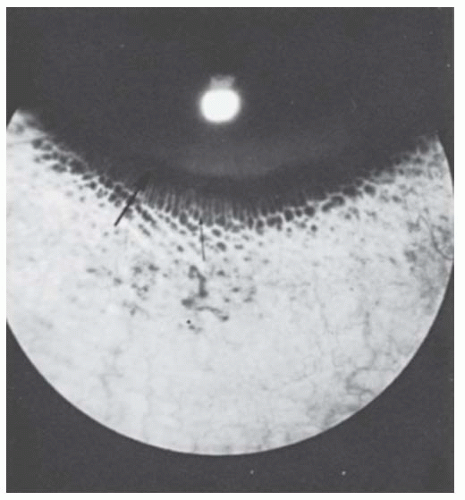
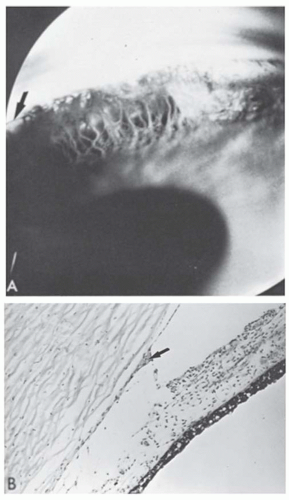
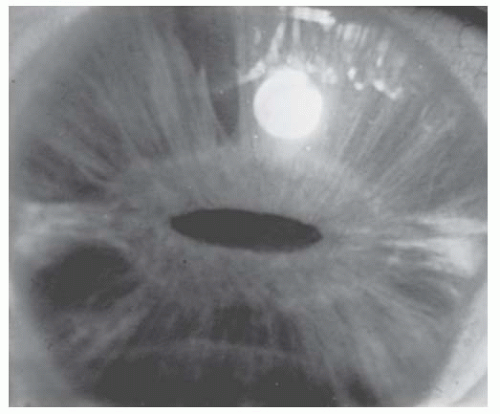
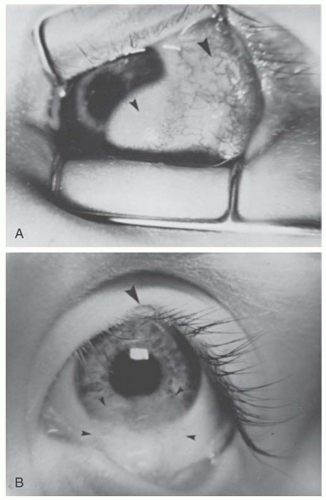
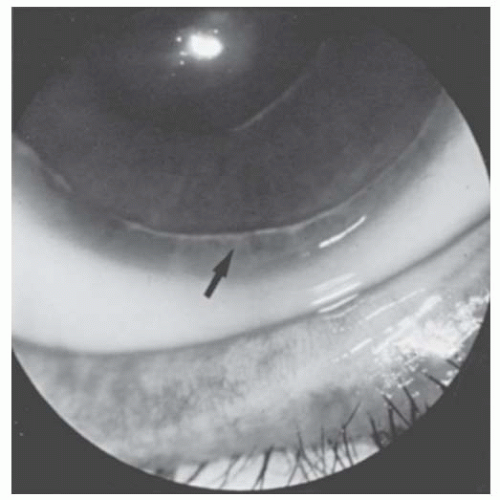 FIGURE 10.13. Rieger’s anomaly. Close-up of 6 o’clock limbus of eye in Figure 10.12. Iris processes (arrow) extend to the irregular prominent Schwalbe’s ring. |
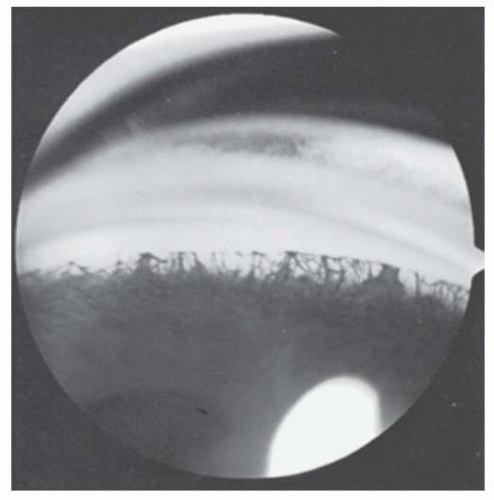 FIGURE 10.14. Angle in Rieger’s anomaly. Gonioscopic appearance of eye in Figures 10.12 and 10.13, showing iris processes extending to the prominent Schwalbe’s ring. |
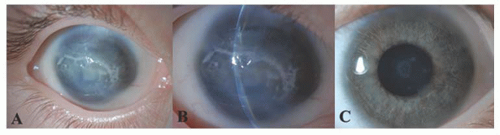
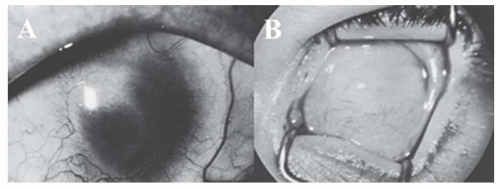
degenerative type (40). Histopathologically, abnormalities include an irregularly thickened epithelial basement membrane, focal disruption of Bowman’s layer, stromal irregularity, and a multilaminar Descemet’s membrane that contains wide-spacing material and focal excrescences (41). Scanning electron microscopy was used to evaluate a cornea with posterior keratoconus, revealing no excrescences of Descemet’s membrane or endothelial tags (42). Given these findings, Al-Hazzaa et al. (42) suggest there may be a subset of posterior keratoconus patients who do not fall into the category of anterior segment dysgenesis abnormalities.
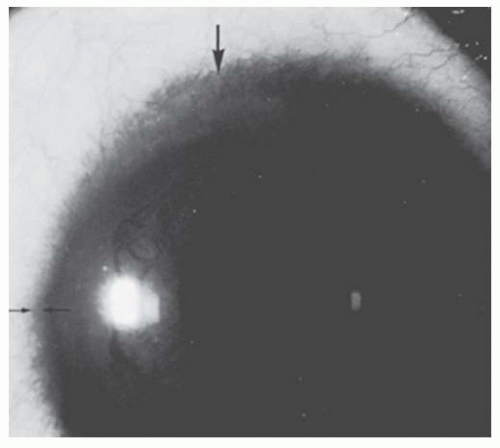
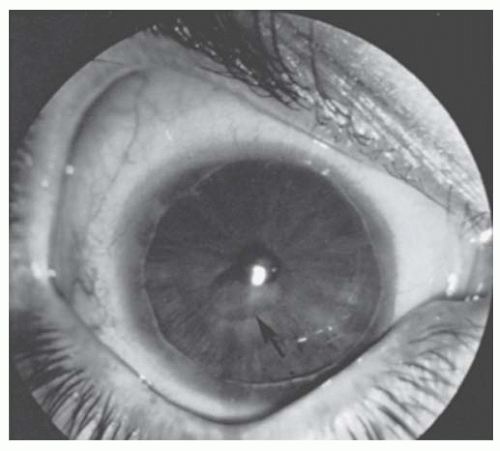
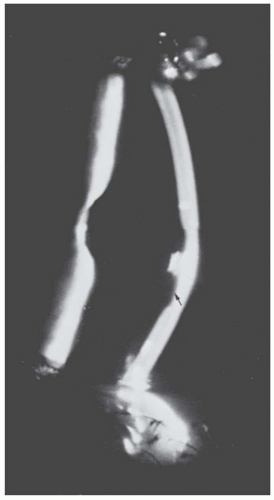 FIGURE 10.18. Posterior keratoconus. Slit-lamp view of the eye in Figure 10.12, showing the depression in the posterior corneal surface (arrow). The cornea overlying it is clear, and no iris processes extend to its margin. |
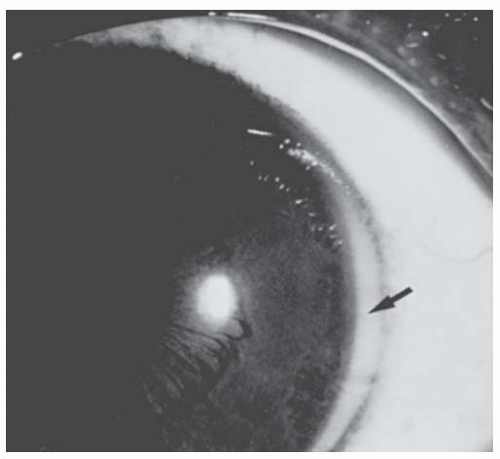
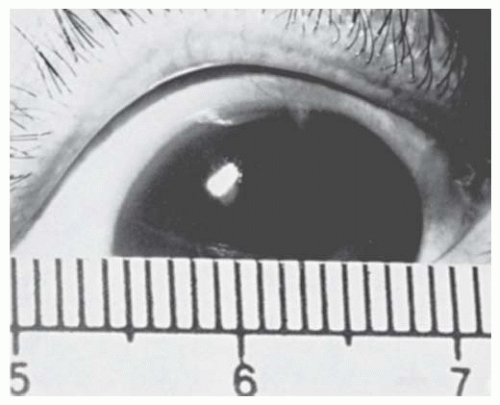
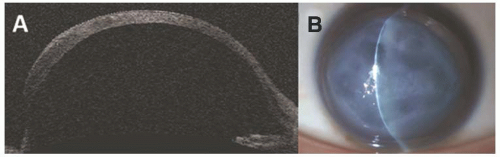
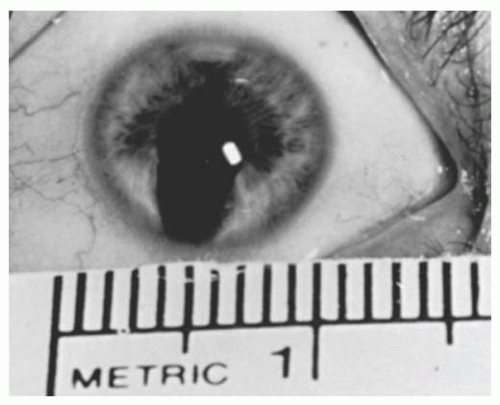
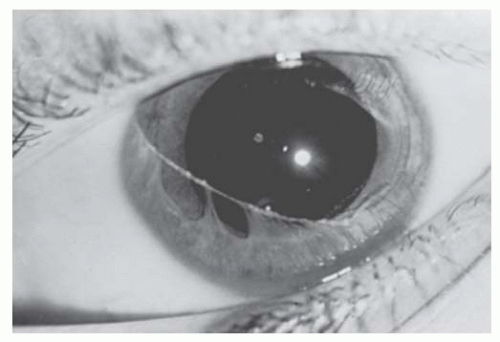
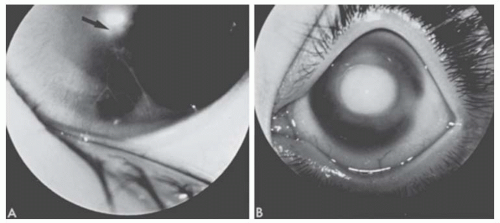 FIGURE 10.19. A: Peters’ anomaly. A mild form showing attenuated iris adhesions to the border of a small corneal opacity (arrow). This was present bilaterally in this 9-month-old white girl. B: Peters’ anomaly. This 10-month-old white girl had bilateral congenital central corneal opacities. During penetrating keratoplasty, iris adhesions were found extending from the pupillary margin to the borders of the opacity. An anterior polar cataract was present. (Courtesy of Harold Koller, MD.) The histopathologic findings are equally varied but usually include thickening or fragmentation of Bowman’s layer, disorganization of stromal architecture, central absence of Descemet’s membrane and endothelium (both of which are present peripherally), and central iridocorneal adhesions (Figs. 10.20 and 10.21C, D). |
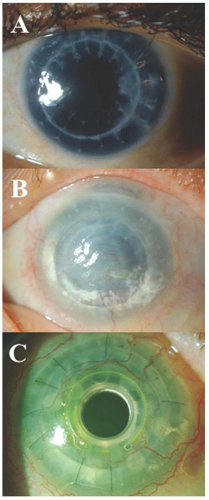
inheritance is also seen. Patients with sporadic aniridia are at risk for WAGR syndrome (Wilms tumor, aniridia, genitourinary abnormalities, mental retardation) and should undergo routine surveillance for kidney disorders. Aniridia is due to mutation at locus 11p13 of the paired box gene 6 (PAX6). It is usually associated with keratopathy, cataract, glaucoma, foveal hypoplasia and strabismus. Dental, musculoskeletal and developmental delays are systemic abnormalities frequently associated with aniridia. Keratopathy is thought to be due to an abnormally differentiated epithelium, abnormal cell adhesion, impaired healing response and limbal stemcell deficiency leading to conjunctivalization of cornea. It begins as vascularized thickening of the cornea at the periphery, which gradually advances centrally. Recurrent corneal erosions lead to subepithelial fibrosis causing corneal opacification. Corneal opacification that occurs due to recurrent erosions is caused by deficiency in matrix metalloproteinase 9 (regulated by PAX6), which is responsible for normal cell remodeling and wound healing. Penetrating keratoplasty for visual rehabilitation is generally unsuccessful because of recurrent surface breakdown. Keratolimbal allograft and Boston keratoprosthesis have proven to be effective for long-term visual rehabilitation (Fig. 10.26) (60,61,62).
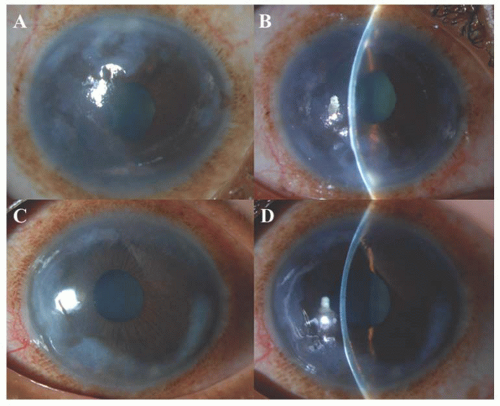
with cornea plana (<38D) and a slightly shallow anterior chamber, which may be a risk factor for the development of secondary glaucoma (66). It should be differentiated from arcus juvenilis, which is devoid of vessels and has a clear lucid interval between the limbus and the corneal opacification.
Table 10.2 STUMPED: DIFFERENTIAL DIAGNOSIS OF NEONATAL CORNEAL OPACITIES | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
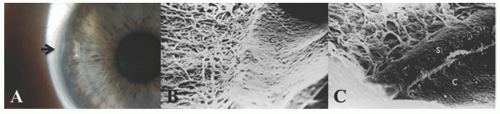
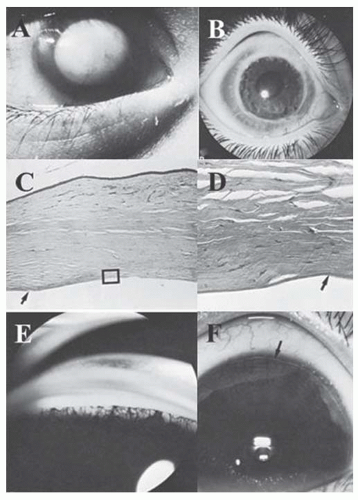
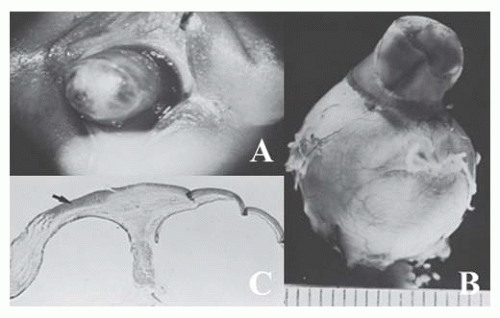
rarely its occurrence in families has been reported. The clinical picture is highly variable, however. The masses may be multiple, bilateral, confined to the cornea alone, minutely small, or large enough to obscure the entire cornea (Fig. 10.29). The dermoid extends into the corneal stroma and sclera but seldom occupies the full thickness and only rarely grows into the angle. Hair is not always present on the surface. Dermoids have been classified into three grades: Grade-1, most frequent type and is small (up to 5 mm) and isolated. Grade-2, is much larger and may cover the entire corneal surface and may extend into deeper layers of the cornea. Grade-3, most severe and rare, it replaces the entire anterior segment (74).
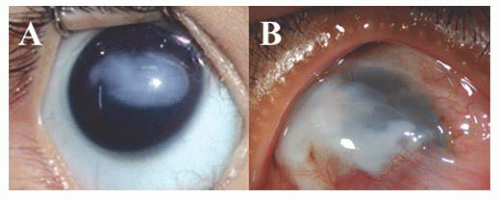
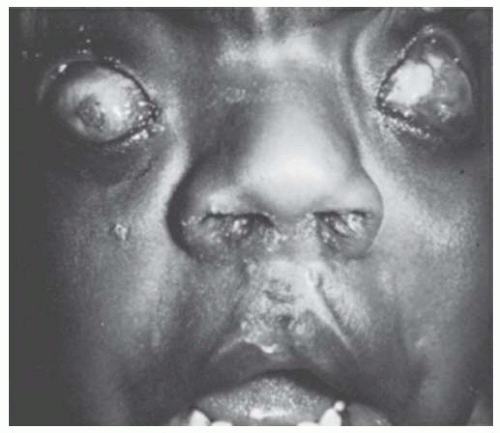
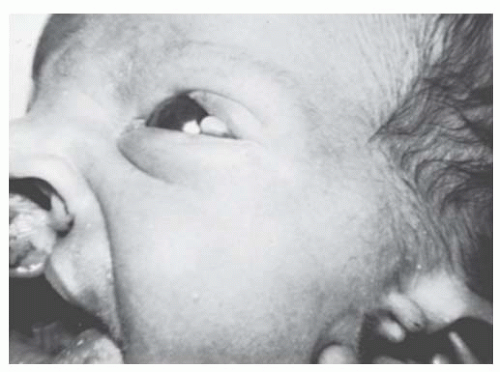 FIGURE 10.30. Goldenhar’s (oculoauriculovertebral dysplasia) syndrome. The limbal dermoid and preauricular skin tags are present, in addition to a cleft lip. (Courtesy of Robison D. Harley, MD.) |
systemic abnormalities, especially cardiovascular, renal, genitourinary, and gastrointestinal defects. Goldenhar syndrome occurs sporadically.
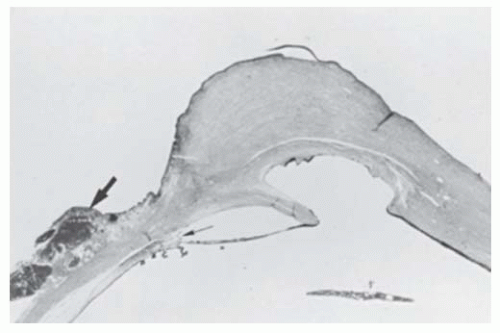
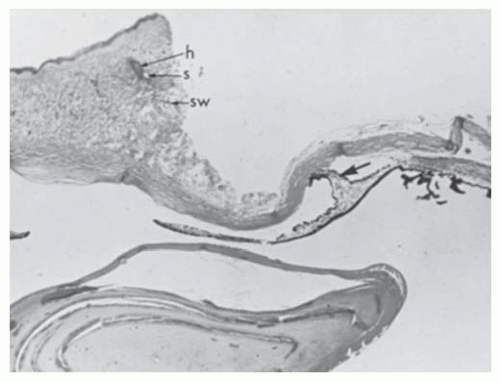 FIGURE 10.33. Corneal dermoid, central posterior corneal defect, and iris-corneal adhesion. The left eye of the patient shown in Figure 10.32. Anterior cornea is replaced by vascularized connective tissue containing hair follicle (h), sebaceous gland (s), and sweat gland (sw). A biopsy has been taken for diagnostic purposes, leaving a defect. Descemet’s membrane is present peripherally but absent centrally. An iris adhesion (arrow) is present centrally. Angle structures are disorganized (Hematoxylin ×4). |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree