Purpose
To update information on cytomegalovirus (CMV) retinitis in patients with acquired immunodeficiency syndrome (AIDS) and to integrate information on its pathogenesis and clinical outcomes.
Design
Literature review.
Methods
Selected articles from the medical literature, particularly large epidemiologic studies, including the Johns Hopkins Cytomegalovirus Retinitis Cohort Study, the Longitudinal Study of the Ocular Complications of AIDS, and the Cytomegalovirus Retinitis and Viral Resistance Study, were reviewed. Clinical information is discussed in light of knowledge on CMV, its pathogenesis, and its interactions with human immunodeficiency virus (HIV).
Results
Cytomegalovirus uses several mechanisms to evade the immune system and establish latent infection in immunologically normal hosts. With immune deficiency, such as late-stage AIDS, CMV reactivates, is disseminated to the eye, and establishes a productive infection, resulting in retinal necrosis. HIV and CMV potentiate each other: CMV accelerates HIV disease, and CMV retinitis is associated with increased mortality. Randomized clinical trials have demonstrated the efficacy of treatments for CMV retinitis. Systemically administered treatment for CMV retinitis decreases AIDS mortality. Highly active antiretroviral therapy (HAART) effectively suppresses HIV replication, resulting in immune recovery, which, if sufficient, controls retinitis without anti-CMV therapy. Resistant CMV, detected in the blood, correlates with resistant virus in the eye and is associated with worse clinical outcomes, including mortality. Host factors, including host genetics and access to care, play a role in the development of CMV retinitis.
Conclusions
Clinical outcomes of CMV retinitis in patients with AIDS are dependent on characteristics of the virus and host and on HIV-CMV interactions.
Epidemiology of Cytomegalovirus Retinitis
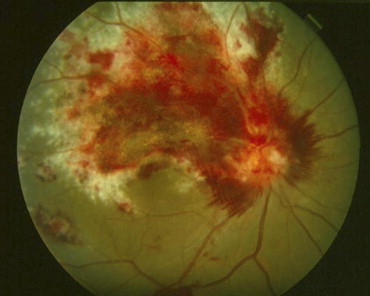
Prior to the advent of the acquired immunodeficiency syndrome (AIDS), cytomegalovirus (CMV) retinitis ( Figure 1 ) was a rare disease seen primarily among transplant patients, but also among those with congenital CMV infection and other forms of immune compromise. It was estimated to affect ∼1% of patients undergoing renal transplant and ∼0.5% of those undergoing bone marrow transplant. It became evident early in the AIDS epidemic that CMV retinitis was a frequent opportunistic infection among patients with AIDS, and that it typically occurred in patients with CD4+ T cells (helper T cells) <50 cells/μL. Epidemiologic studies in the era before highly active antiretroviral therapy (HAART) reported that the incidence of CMV retinitis among patients with CD4+ T cells <50 cells/μL was 0.20/person-year (PY), and that the lifetime probability of a patient with AIDS getting CMV retinitis was ∼30%. Although other end-organ CMV disease, such as colitis, esophagitis, cerebritis, and pneumonitis, occurred in patients with AIDS, the eye was the organ affected in ∼80% of patients with AIDS and CMV disease. By the mid-1990s CMV retinitis was the most frequently encountered intraocular infection at major urban medical centers in the United States.
The primary risk factor for CMV retinitis appeared to be the level of immunodeficiency, as determined by CD4+ T cells, but other risk factors contributed. These other factors included the level of CD8+ T cells (cytotoxic T cells); retinal vascular damage, as evidenced by the microangiopathy in the retina (known as AIDS retinopathy or HIV retinopathy); and bacteremia from Mycobacterium avium complex (MAC). In a case-control study of patients with AIDS matched for CD4+ T cells, those with CMV retinitis had significantly lower CD8+ T cells (152 cells/μL) than did those without CMV retinitis (296 cells/μL, P < .001). In the Johns Hopkins CMV Retinitis Cohort Study, the presence of HIV microangiopathy, as evidenced by cotton-wool spots, increased the odds of CMV retinitis 1.46-fold ( P < .05). In a study by the UCLA CMV Retinitis Study Group, the large majority of small (and presumably early) CMV lesions (73%) were adjacent to retinal blood vessels. These data suggest the importance of vascular damage to the risk of CMV retinitis in patients with AIDS and are consistent with data suggesting that the method of entry of CMV into the retina is through vascular endothelial cells (see below). In the Multicenter AIDS Cohort Study, the relative risk for CMV retinitis among patients with MAC bacteremia was 3.94 ( P < .0001), and in the Johns Hopkins CMV Retinitis Cohort the relative odds were 3.04 ( P < .05).
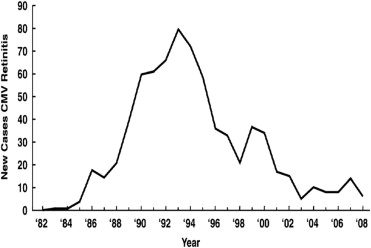
The development of HAART profoundly altered the AIDS epidemic as it resulted in control of replication by the human immunodeficiency virus (HIV), improved immune function (as evidenced by rises in CD4+ T cells), a marked decrease in the incidence of opportunistic infections, and improved survival. With the advent of HAART, the incidence of CMV retinitis has decreased by 80% to 90%, although that decrease has leveled off ( Figure 2 ), and CMV retinitis continues to occur. HIV infection now is a chronic disease with survival after infection with HIV estimated at >14 years. However, because of late testing among many high-risk patients, approximately one-third of patients with newly diagnosed HIV infection in the United States still will progress to AIDS within one year of the diagnosis of HIV infection, resulting in an ongoing population at risk for CMV retinitis.
Pathogenesis of Cytomegalovirus Disease
Cytomegalovirus is a ubiquitous herpes family virus, and seroprevalence surveys estimate that ∼50% of the general population is latently infected. Certain populations, such as men who have sex with men, have a higher rate of latent infection (estimated at >90%). In immunologically normal hosts, acquired CMV typically manifests as a mild, self-limited, flu-like illness, followed by a latent infection. The site of latency is thought to be granulocytes. Cytomegalovirus uses several of its protein products to prevent the presentation of CMV antigens in conjunction with HLA class I molecules and thereby prevent clearance of CMV-infected cells by CD8+ T cells. Although natural killer (NK) cells recognize downregulation of HLA class I by virally infected cells and thereby clear them, CMV produces an HLA class I homologue, which allows CMV-infected cells to evade NK cell surveillance. With immune compromise, CMV can reactivate and be hematogenously disseminated to target organs. The large majority of patients with newly diagnosed CMV retinitis have evidence of systemic CMV replication, detected either by culture in either the blood or urine (∼80%) or by polymerase chain reaction (PCR) amplification of blood specimens to detect CMV DNA in the blood (∼60%). The entry of CMV into the eye appears to be through retinal blood vessels, and pathology studies have shown infection of vascular endothelial cells at the edge of the lesion. Cytomegalovirus retinitis results in full-thickness retinal necrosis of infected areas. The end result of infection is retinal destruction of infected areas. Cytomegalovirus resides at the edge of the lesion, and unless there is immune recovery or anti-CMV therapy is administered, CMV spreads into contiguous retina, resulting in enlarging lesions. As lesions enlarge ever-increasing amounts of visual field are lost, and when the optic nerve or fovea is damaged there is a loss of visual acuity.
Cytomegalovirus and HIV Interactions
Cytomegalovirus and HIV interact to promote both infections. Cytomegalovirus and HIV transactivate each other in vitro, resulting in increased yields of both viruses. Dual infection of individual retinal cells of autopsy eyes from patients with AIDS and CMV retinitis with both CMV and HIV has been demonstrated. Cytomegalovirus produces cytokine homologues that bind to host receptors and downregulate the immune system. One key cytokine homologue appears to be an interleukin (IL)-10 homologue (cmv IL-10h). IL-10 is a human cytokine that inhibits T helper type-1 (Th1) immune responses, which are key to cell-mediated immunity. Cytomegalovirus also produces chemokine receptors, which bind chemokines and inhibit the recruitment of inflammatory and immune cells, and (as noted above) it interferes with natural killer cells, thereby inhibiting the host’s ability to clear viruses. Studies of populations with low seroprevalence of CMV infection, such as neonatal and transfusion-acquired HIV infection, demonstrate that CMV co-infection (as evidenced by positive serology), even in the absence of end-organ CMV disease, accelerates the development of AIDS and shortens the time from acquisition of HIV to AIDS.
Mechanisms of Visual Loss
The primary cause of visual loss in patients with CMV retinitis is necrotic damage to the fovea or optic nerve from CMV infection ( Table 1 ). In the era before HAART, the second-leading cause of visual loss was retinal detachment, whereas in the HAART era it was cataract, in part because of the reduced incidence of retinal detachment in the HAART era. Macular edema was uncommon in the pre-HAART era but is a more common cause of visual impairment in the HAART era, particularly among patients with immune recovery uveitis (see below).
| Cause (% of Eyes) | Pre-HAART Era 20/200 or Worse | HAART Era | |
|---|---|---|---|
| 20/50 or Worse | 20/200 or Worse | ||
| Macula or optic nerve disease | 54-84 | 63.1 | 69.8 |
| Cataract | No data | 22.0 | 28.6 |
| Retinal detachment | 36-63 | 12.9 | 16.7 |
| Macular edema | <5 | 12.4 | 11.1 |
| Immune recovery uveitis | Not applicable | 4.3 | 3.2 |
| Epiretinal membrane | <5 | 3.3 | 4.1 |
a From Thorne and associates. Used with permission. Only data on causes of blindness (20/200 or worse) are available from the pre-HAART era.
Pathogenesis of Cytomegalovirus Disease
Cytomegalovirus is a ubiquitous herpes family virus, and seroprevalence surveys estimate that ∼50% of the general population is latently infected. Certain populations, such as men who have sex with men, have a higher rate of latent infection (estimated at >90%). In immunologically normal hosts, acquired CMV typically manifests as a mild, self-limited, flu-like illness, followed by a latent infection. The site of latency is thought to be granulocytes. Cytomegalovirus uses several of its protein products to prevent the presentation of CMV antigens in conjunction with HLA class I molecules and thereby prevent clearance of CMV-infected cells by CD8+ T cells. Although natural killer (NK) cells recognize downregulation of HLA class I by virally infected cells and thereby clear them, CMV produces an HLA class I homologue, which allows CMV-infected cells to evade NK cell surveillance. With immune compromise, CMV can reactivate and be hematogenously disseminated to target organs. The large majority of patients with newly diagnosed CMV retinitis have evidence of systemic CMV replication, detected either by culture in either the blood or urine (∼80%) or by polymerase chain reaction (PCR) amplification of blood specimens to detect CMV DNA in the blood (∼60%). The entry of CMV into the eye appears to be through retinal blood vessels, and pathology studies have shown infection of vascular endothelial cells at the edge of the lesion. Cytomegalovirus retinitis results in full-thickness retinal necrosis of infected areas. The end result of infection is retinal destruction of infected areas. Cytomegalovirus resides at the edge of the lesion, and unless there is immune recovery or anti-CMV therapy is administered, CMV spreads into contiguous retina, resulting in enlarging lesions. As lesions enlarge ever-increasing amounts of visual field are lost, and when the optic nerve or fovea is damaged there is a loss of visual acuity.
Cytomegalovirus and HIV Interactions
Cytomegalovirus and HIV interact to promote both infections. Cytomegalovirus and HIV transactivate each other in vitro, resulting in increased yields of both viruses. Dual infection of individual retinal cells of autopsy eyes from patients with AIDS and CMV retinitis with both CMV and HIV has been demonstrated. Cytomegalovirus produces cytokine homologues that bind to host receptors and downregulate the immune system. One key cytokine homologue appears to be an interleukin (IL)-10 homologue (cmv IL-10h). IL-10 is a human cytokine that inhibits T helper type-1 (Th1) immune responses, which are key to cell-mediated immunity. Cytomegalovirus also produces chemokine receptors, which bind chemokines and inhibit the recruitment of inflammatory and immune cells, and (as noted above) it interferes with natural killer cells, thereby inhibiting the host’s ability to clear viruses. Studies of populations with low seroprevalence of CMV infection, such as neonatal and transfusion-acquired HIV infection, demonstrate that CMV co-infection (as evidenced by positive serology), even in the absence of end-organ CMV disease, accelerates the development of AIDS and shortens the time from acquisition of HIV to AIDS.
Mechanisms of Visual Loss
The primary cause of visual loss in patients with CMV retinitis is necrotic damage to the fovea or optic nerve from CMV infection ( Table 1 ). In the era before HAART, the second-leading cause of visual loss was retinal detachment, whereas in the HAART era it was cataract, in part because of the reduced incidence of retinal detachment in the HAART era. Macular edema was uncommon in the pre-HAART era but is a more common cause of visual impairment in the HAART era, particularly among patients with immune recovery uveitis (see below).
| Cause (% of Eyes) | Pre-HAART Era 20/200 or Worse | HAART Era | |
|---|---|---|---|
| 20/50 or Worse | 20/200 or Worse | ||
| Macula or optic nerve disease | 54-84 | 63.1 | 69.8 |
| Cataract | No data | 22.0 | 28.6 |
| Retinal detachment | 36-63 | 12.9 | 16.7 |
| Macular edema | <5 | 12.4 | 11.1 |
| Immune recovery uveitis | Not applicable | 4.3 | 3.2 |
| Epiretinal membrane | <5 | 3.3 | 4.1 |
a From Thorne and associates. Used with permission. Only data on causes of blindness (20/200 or worse) are available from the pre-HAART era.
Immune Recovery and Immune Recovery Uveitis
Highly active antiretroviral therapy consists of combination therapy, generally with three or more drugs, including at least one potent antiretroviral drug, such as a protease inhibitor. In the industrialized world HAART became widely used in the mid-1990s and changed the course of HIV management. For the first time, clinicians were able to obtain substantial and sustained suppression of HIV replication, typically measured as the amount of HIV RNA in the blood, also known as HIV “load” or “viral load.” With suppression of HIV replication, the decline in immunity, measured as the decline in CD4+ T cells, could be forestalled, and those with impaired immunity could experience immune recovery, measured as a rise in CD4+ T cells. The incidence of AIDS among patients with HIV infection declined, as fewer patients progressed to end-stage HIV infection, and the incidence of opportunistic infections also declined. Immunologic studies demonstrated the restoration of specific immunity to pathogens, such as CMV, with immune recovery, although the recovery generally took three to six months. AIDS mortality declined, and the prevalence of patients alive with AIDS and earlier stages of HIV infection increased. Resistance of HIV to the drugs in the HAART regimen, particularly among patients heavily pretreated with single drugs in the pre-HAART era, could limit immune recovery. In addition, failure to fully reconstitute the immune system, resulting in partial restoration of immunity or lacunae in the immune repertoire, has been demonstrated. These problems would result in some patients remaining at risk for opportunistic infections, but they appear to be decreasing as HAART use expanded and with the introduction of additional antiretroviral drugs.
Patients with CMV retinitis started on HAART, restarted on HAART, or having their regimen changed often experienced sufficient immune recovery to stop anti-CMV therapy without the CMV retinitis relapsing, documented by numerous case series. The recommended threshold for interrupting anti-CMV therapy is a CD4+ T cell count over 100 cells/μL. Because there is a delay in restoring specific immunity after a rise in CD4+ T cells, the CD4+ T cell count should be above 100 cells/μL for a minimum of three to six months before stopping anti-CMV therapy. Case series have suggested that among patients with immune recovery, discontinuation of anti-CMV therapy, and subsequent failure of immune recovery, relapse of the CMV retinitis occurs when CD4+ T cells have declined again to <50 cells/μL.
With the advent of HAART and immune recovery, a new problem arose, immune recovery uveitis (IRU). Immune recovery uveitis is one of the immune recovery inflammatory syndromes (IRIS), which have been described in other organs and with other opportunistic infections (such as mycobacterial infections). Patients with immune compromise and CMV retinitis usually have either no or mild anterior chamber and vitreous cellular reactions, which are usually less among patients treated with anti-CMV therapy. Immune recovery uveitis is characterized by the new occurrence of or an increase in anterior chamber or vitreous inflammatory reaction (cells) in the face of immune recovery, most often a vitritis. Uveitis-related structural complications, such as macular edema, also occur and can compromise vision. Immediately after the introduction of HAART, widely disparate rates of IRU were reported, from 0.83/PY to 0.11/PY. Initially the reasons for this disparity were unclear, but subsequent investigations suggested a substantial role for intravitreal cidofovir in these disparate rates. The use of intravitreal cidofovir is a major risk factor for IRU (relative odds = 19), and the highest rate of IRU was reported from the clinic that pioneered the investigation of intravitreal cidofovir therapy as treatment for CMV retinitis. In the late 1990s to early 2000s the prevalence of IRU among patients with CMV retinitis was reported as 14% in the Longitudinal Study of the Ocular Complications of AIDS (LSOCA) cohort. It was hypothesized that IRU might represent a partial restoration of immunity, particularly among those who could not develop full benefit from HAART because of prior antiretroviral treatment or prolonged sustained immunodeficiency with resultant partial immune restoration from HAART. Supporting this latter concept is the apparent decrease in the incidence of IRU among patients diagnosed with CMV retinitis and then experiencing immune recovery in the HAART era ( Figure 3 ), which now is estimated to be 0.04/PY.
There is an issue on the timing of starting a patient on HAART who has active CMV retinitis. In general, HAART treatment is started as soon as possible in patients with opportunistic infections. However, one historically controlled study suggested that early introduction of HAART in a patient with CMV retinitis before completing induction therapy for CMV resulted in a higher incidence of IRU (71%) than among those who had suppressed retinitis before starting HAART (31%, P = .01), and a greater severity of IRU. These data suggest that all patients with CMV retinitis, even HAART-naïve patients with small peripheral lesions, should be treated for the CMV to reduce the incidence of IRU. Furthermore, these data suggest that a delay in initiating HAART until after the retinitis had been controlled might be beneficial. However, this approach must be counterbalanced by the risk of other opportunistic infections if antiretroviral therapy is delayed. One randomized trial in which HAART was started approximately two weeks after opportunistic infection therapy vs approximately six weeks demonstrated a significantly greater risk of AIDS-related events in the latter group. Furthermore, there was no increase in IRIS events in the latter group, although CMV retinitis was not specifically studied. Because the rate of IRU is low (0.04/PY) and because CMV replication is controlled within one to two weeks, a delay of no more than two weeks would seem prudent.
Treatment of Cytomegalovirus Retinitis
Treatment of CMV retinitis is influenced by the fact that all available treatments are virostatic and do not eliminate the virus. A histopathologic study of treated CMV retinitis demonstrated the presence of viral DNA at the border of the lesion, but ineffective assembly of intact virions. With cessation of therapy viral assembly resumes, as does clinical retinitis. Therefore, unless there is immune recovery, chronic, lifelong therapy to prevent relapse is required for patients with AIDS. Three United States Food and Drug Administration (FDA)-approved drugs are available for the treatment of CMV retinitis: ganciclovir (Cytovene; Roche Pharmaceuticals, Nutley, New Jersey, USA), foscarnet (Foscavir; AstraZeneca Pharmaceuticals LP, Wilmington, Delaware, USA), and cidofovir (Vistide; Gilead, Foster City, California, USA). A fourth drug, fomivirsen (Vitravene; Isis Pharmaceuticals, Inc, Carlsbad, California, USA), an anti-sense aptamer, was FDA-approved and then withdrawn from the market when the incidence of CMV retinitis declined after the introduction of HAART. Ganciclovir can be administered by intravenous infusion; by surgical placement of a sustained-release implant into the vitreous (Vitrasert; Bausch & Lomb Pharmaceuticals, Inc, Tampa, Florida, USA), which typically lasts at least six months; or orally with the prodrug valganciclovir (Valcyte; Roche Pharmaceuticals). An oral formulation of ganciclovir, with poor bioavailability, largely has been replaced by valganciclovir. Foscarnet and cidofovir are administered intravenously. Ganciclovir and foscarnet also are administered off-label as intravitreal injections. Cidofovir was investigated as an intravitreal drug, but its use largely has been discontinued because of a narrow therapeutic-toxic window and problems with uveitis and hypotony. The treatment of CMV retinitis has been the subject of numerous clinical trials ( Table 2 ), sponsored both by the pharmaceutical industry and by the National Institutes of Health (NIH).
| Trial Name/Design | Year Published | Treatments | Main Results |
|---|---|---|---|
| Foscarnet vs observation | 1991 | Foscarnet | Foscarnet effective |
| Ganciclovir vs observation for peripheral retinitis | 1993 | Ganciclovir | Ganciclovir effective |
| Foscarnet Ganciclovir CMV Retinitis Trial (FGCRT) | 1992 | Foscarnet vs ganciclovir | Foscarnet and ganciclovir equivalent for controlling CMV retinitis; foscarnet associated with a lower mortality |
| CMV Retinitis Retreatment Trial (CRRT) | 1996 | Foscarnet vs ganciclovir vs combination | Combination treatment superior to monotherapy for relapsed retinitis |
| Monoclonal Antibody CMV Retinitis Trial (MACRT) | 1997 | Anti-CMV monoclonal antibody vs placebo as adjunct to drug therapy | Antibody ineffective and associated with increased mortality |
| HPMPC Peripheral CMV Retinitis Trial (HPCRT) | 1997 | Cidofovir vs observation for peripheral retinitis | Cidofovir effective |
| Cidofovir peripheral CMV retinitis trial | 1997 | Cidofovir vs observation for peripheral retinitis | Cidofovir effective |
| Ganciclovir implant trial | 1994 | Ganciclovir implant vs observation for peripheral CMV retinitis | Ganciclovir implant effective |
| Ganciclovir implant vs ganciclovir trial | 1997 | Ganciclovir implant vs intravenous ganciclovir | Implant more effective for controlling retinitis but higher rate 2nd eye & visceral disease |
| Ganciclovir implant vs implant & oral ganciclovir vs intravenous ganciclovir | 1999 | Ganciclovir implant vs implant & oral ganciclovir vs intravenous ganciclovir | Implant & oral ganciclovir more effective than implant alone for preventing 2nd eye & visceral disease & similar to intravenous ganciclovir |
| Ganciclovir Cidofovir CMV Retinitis Trial (GCCRT) | 2001 | Ganciclovir implant & oral ganciclovir vs cidofovir | Two regimens equivalent for controlling retinitis |
| Valganciclovir vs intravenous ganciclovir for induction | 2002 | Valganciclovir vs intravenous ganciclovir | Valganciclovir similar to intravenous ganciclovir for induction therapy |
Systemically administered anti-CMV therapies are given initially at higher doses to control the infection (known as induction) for two to three weeks and then at lower doses to prevent relapse of the clinical disease (maintenance or secondary prophylaxis). Although systemically administered therapies initially control the retinitis, with time, the retinitis relapses, requiring repeated cycles of induction and maintenance; and unless there is immune recovery, there is a progressive shortening of the time to relapse.
The evaluation of the efficacy of anti-CMV therapies required the development of methodologies and outcomes suitable for clinical trials. The primary outcome of nearly all trials of anti-CMV therapies has been the time to progression, defined as the movement of the border of a CMV lesion one-half disc diameter in distance, along a front one-half disc diameter in size, or the occurrence of a new lesion one-quarter disc area in size. Meta-analysis of clinical trials with long-term follow-up demonstrated that retinitis progression is a reasonable surrogate marker for subsequent visual loss. Because the administration of intravenous therapies (the first ones developed) required a central line and because of the differential methods of administering the different drugs, masked treatment administration typically was not feasible, so clinical trials employed a reading center to read standardized retinal photographs with graders masked as to treatment assignment. Studies of reading center performance demonstrated that reading center grading of retinal photographs detected retinitis progression earlier than clinicians. A study of two reading centers’ performance grading the same set of photographs demonstrated that although the two reading centers might differ on an occasional grading (κ = 0.55), overall they gave very similar results for time to progression analyses of the entire study population (medians 65 vs 69 days).
In order to determine if a new treatment for CMV retinitis was effective, clinical trials often randomized patients with “small peripheral lesions” to the new treatment or to observation for a brief period of time until there was retinitis progression. Formalization of this methodology included the definition of retinal zones: zone 1 encompassed an area one disc diameter from the edge of the optic nerve or two disc diameters from the center of the fovea (these lesions are considered immediately vision-threatening); zone 2 extended from the edge of zone 1 approximately to the equator as marked by a circle identified by the vortex vein ampullae, and zone 3 extended anteriorly from the edge of zone 2 to the ora serrata. Clinical trials of “small peripheral lesions” enrolled patients with lesions only in zones 2 or 3, which occupied less than 25% of the retinal area. A long-term follow-up study of patients enrolled in one such trial demonstrated no evident long-term adverse outcomes in terms of mortality, vision loss, or structural complications from the brief period of observation.
The second major type of trial was a comparative trial between two drugs. All of the systemically administered drugs appear to have similar efficacy. Comparative trials of intravenous ganciclovir and intravenous foscarnet and of intravenous ganciclovir and oral valganciclovir have demonstrated similar efficacy. Because of the ease of administration and absence of catheter-related complications, valganciclovir now is nearly always used for systemic therapy.
The ganciclovir implant produces intraocular levels of ganciclovir five times that of systemically administered ganciclovir and generally controls new cases of CMV retinitis until it runs out of drug, on average in seven months. Comparative studies have shown that the implant is associated with longer times to retinitis progression than is systemic ganciclovir therapy. The implant can be replaced, and studies showed that the same site could be used twice relatively easily, but that the third surgery in a quadrant was associated with a higher rate of surgical complications, suggesting that eight implants could be placed in sequence (two in each quadrant) over a period of four years with a reasonably low level of surgical complications. However, because CMV retinitis is part of a systemic infection, and local intraocular therapy does not control the systemic part of the disease, local-only therapy was associated with high rates of contralateral or “second eye” disease (in those presenting with unilateral disease) and visceral disease, substantially higher than those seen with systemic therapy. Given the benefit of systemic therapy on mortality (see below) and on dissemination of disease, most patients treated with an implant also receive systemic therapy with valganciclovir.
Intravitreal injections of ganciclovir and foscarnet have to be given two to three times weekly as induction therapy and weekly as maintenance therapy and have relapse rates similar to those of systemic therapy. Therefore, in the industrialized world, injections generally are used as initial therapy until an implant can be placed or to determine if relapsed retinitis can be controlled by higher doses of the drug (eg, an implant in a patient previously treated systemically) or the alternative drug (eg, in a patient with presumed resistant CMV). In resource-poor countries, such as sub-Saharan Africa, for economic reasons, repetitive intravitreal injections often are the primary mode of therapy.
Because immune recovery can result in control of the retinitis without concomitant anti-CMV therapy, the United States Department of Health and Human Services recommendations for managing patients with CMV retinitis recommend that patients with immune recovery to >100 cells/μL for at least three to six months have anti-CMV therapy discontinued. However, because not all patients with immune recovery will control the retinitis without anti-CMV therapy, and because the risk of relapse continues for at least five years ( Table 3 , Figure 4 ), all such patients require regular ophthalmologic follow-up, which has been recommended to occur at three-month intervals.
| Outcome | Pre-HAART | HAART Era | |
|---|---|---|---|
| 2- to 3-Year Rate | 5-Year Rate | ||
| Mortality (/PY) | 0.12 | 0.10 | |
| Retinitis progression (/PY) | |||
| Overall | ∼3.0 | 0.10 | 0.07 |
| CD4+ T cells <50 cells/μL | 0.27 | 0.23 | |
| CD4+ T cells >100 cells/μL | 0.03 | 0.02 | |
| Retinal detachment (/EY) | |||
| Overall | 0.33 | ∼0.04 | 0.02 |
| CD4+ T cells <50 cells/μL | ∼0.10 | 0.04 | |
| CD4+ T cells >100 cells/μL | ∼0.03 | 0.01 | |
| 2nd eye involvement (/PY) | |||
| Overall | 0.40 | 0.07 | 0.03 |
| CD4+ T cells <50 cells/μL | 0.16 | 0.07 | |
| CD4+ T cells >100 cells/μL | 0.02 | 0.01 | |
| Visual acuity loss to worse than 20/40 (/EY) | |||
| Overall | ∼0.96 | 0.10 | 0.08 |
| CD4+ T cells <50 cells/μL | 0.19 | 0.09 | |
| CD4+ T cells >100 cells/μL | 0.10 | 0.05 | |
| Visual acuity loss to 20/200 or worse (/EY) | |||
| Overall | ∼0.48 | 0.06 | 0.03 |
| CD4+ T cells <50 cells/μL | 0.07 | 0.05 | |
| CD4+ T cells >100 cells/μL | 0.04 | 0.025 | |
Treatment for CMV retinitis is dependent on the location of the retinitis and the likelihood of immune recovery ( Table 4 ), as well as treatment adherence considerations. Because there is a three- to six-month lag between initiation of HAART and recovery of specific immunity to CMV, and because the likelihood of complications of CMV (eg, retinal detachment, IRU) is related to the size of the CMV lesion, nearly all patients with CMV retinitis, even those expected to experience immune recovery, should be treated with anti-CMV therapy for at least six months. Patients without immune recovery will require lifelong therapy. Because of the improvement in survival and decrease in the incidence of second eye and visceral disease with systemic therapy, most patients should receive systemic anti-CMV therapy. Because the implant controls retinitis progression better than systemic anti-CMV therapy, it generally is the preferred treatment for zone 1 lesions. Small peripheral lesions in a HAART-naïve patient often are treated with systemic therapy alone, as immune recovery is expected.



