Correction of Blepharoptosis in Children
Forrest James Ellis
The term blepharoptosis refers to an eyelid with a lower than normal position of the eyelid margin relative to the eye. The term is shortened to the less specific (but widely used) term ptosis in this text.
EYELID DEVELOPMENT
The development of the eyelid is intimately associated with the development of the eye.1 The optical vesicle forms as a projection from the side of the forebrain during the fourth week and invaginates forming the optic cup. The ectoderm overlying the optic vesicle forms the lens placode, which forms the lens of the eye. In addition, ectoderm also develops into the cornea. During the sixth week, two small folds of the surface ectoderm with underlying mesenchyme appear. These become the upper and lower eyelids. The folds grow toward each other and ultimately fuse between weeks eight and ten of gestation. The eyelids remain fused until they separate during the sixth month of gestation.
UPPER EYELID ANATOMY
The margin of the upper eyelid forms a curved arch from the medial canthus to the lateral canthus and overlies the superior 1 to 2 mm of the cornea. The peak of this curve is approximately 1 mm nasal to the center of the cornea. Attachments of strands of the external levator aponeurosis to the overlying skin form the upper eyelid crease.2 Congenital ptosis due to abnormal levator development often results in reduced or absent upper eyelid crease. However, the normal Asian eyelid has a low-set and less developed eyelid crease.3 Dehiscence of the levator aponeurosis may result in an abnormally elevated eyelid crease.
The upper eyelid tarsus is approximately 10 mm in its vertical height in the adult and proportionately shorter in children. Both medially and laterally the tarsus is attached to the orbital rims by the canthal tendons.
The upper eyelid overlying the tarsal plate consists of eyelid skin covering the orbicularis oculi muscle. This pretarsal orbicularis is adherent to the underlying tarsal plate. Conjunctiva is also firmly adhered to the tarsus posteriorly. Superior to the tarsus the skin covers the preseptal orbicularis muscle. Beneath preseptal muscle is the orbital septum. The orbital septum defines the anterior boundary of the orbit and overlies orbital fat. In most upper eyelids, the orbital septum and levator aponeurosis fuse at approximately the superior tarsal border 10 mm above the lid margin. However, in Asian eyelids the septum inserts much lower into the levator aponeurosis resulting in inferior displacement of orbital fat and a lower eyelid crease.3 Posterior to the levator aponeurosis is the underlying Müller muscle.
LEVATOR PALPEBRAE SUPERIORIS AND MÜLLER MUSCLE
The levator muscle has its origin at the orbital apex on the lesser wing of the sphenoid. The muscle is positioned posterior to anterior in the superior aspect of the orbit. Just posterior to the superior orbital rim, the levator muscle crosses and fuses with the Whitnall ligament. The Whitnall ligament provides vertical support to the levator muscle and its aponeurosis. Anterior to the Whitnall ligament the levator muscle becomes more fibrous, forming the more vertically oriented levator aponeurosis. The levator aponeurosis spreads out horizontally to form a fan-shaped structure with attachments medially and laterally:the medial and lateral horns, which insert in the periosteum. The levator aponeurosis inserts broadly across the anterior surface of the upper eyelid tarsus. Small strands of the levator aponeurosis also project anteriorly, inserting into the eyelid skin and forming the eyelid crease.
Cranial nerve VII (the facial nerve) innervates the facial musculature, and the temporal branch of the facial nerve provides innervation to the orbicularis oculi muscle, frontalis muscle, pars ciliaris, and corrugator muscles. The superior division of cranial nerve III innervates the levator palpebrae superioris muscle. The Müller muscle is innervated by the sympathetic nervous system. Interruption of ocular sympathetic supply causes ptosis, miosis, and anhydrosis. This triad of signs is known as Horner syndrome.4
Recent work has demonstrated that normal extraocular muscle development is dependent on neurologic innervation.5,6,7,8 Congenital fibrosis of the extraocular muscles has been shown to result from primary genetically identified neurodevelopmental defects. The defects may involve the cranial nerve nuclei or the nerve axon guidance. Congenital ptosis is seen as part of the congenital fibrosis syndromes as well as several other congenital cranial nerve dysinnervation syndromes. An extraocular muscle will not develop normally in the absence of neural innervation. Such a muscle is typically atrophic with markedly reduced contractility. In addition, the muscle is less elastic. The antagonist extraocular muscle(s) develops unopposed and is typically contractured.
With regard to congenital ptosis, the levator palpebrae superioris muscle is typically inelastic and atrophic with reduced contractility.9 Not only will the eyelid not elevate normally, but the eyelid may not close completely either (lagophthalmos).
In the absence of “normal” innervation, an extraocular muscle may be congenitally misinnervated by a branch of another cranial nerve. The affected muscle develops a more normal structure and contractility. However, the muscle will contract at “the wrong time”. Duane syndrome is the most well known of these misinnervation syndromes.10 In Duane syndrome the lateral rectus muscle typically contracts during attempted adduction of the eye. Since the medial rectus muscle is also contracting at the same time, the eye retracts into the orbit during attempted adduction. A secondary ptosis may be observed due to the relative enophthalmos that occurs during this co-contracture.
With regard to the eyelid, Marcus Gunn jaw wink ptosis is a common form of misinnervation. In this condition, the levator palpebrae superioris has reduced innervation by the superior branch of the third cranial nerve but is misinnervated by a branch of the fifth cranial nerve that normally innervates the lateral pterygoid muscle. Affected individuals typically have mild ptosis in primary position, but with contralateral jaw movement the ptotic eye elevates. This is often noticed in infancy during feeding as the child seems to wink when nursing or taking a bottle.
BLEPHAROPTOSIS
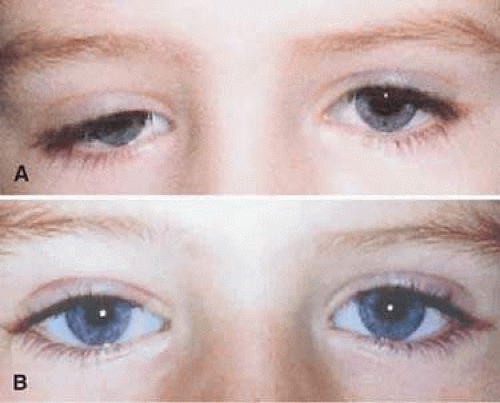
Congenital ptosis of the upper eyelid is typically seen in association with a maldevelopment of the levator palpebral complex. Superior rectus muscle weakness may occur in association with congenital ptosis. The levator muscle and the aponeurosis appear fatty and fibrous. However, blepharoptosis is not related to other forms of muscular dystrophy.9 Congenital ptosis occurs either unilaterally or bilaterally. Although typically sporadic, familial ptosis has been linked to chromosome 1p.11
Congenital ptosis may be associated with broader neurodystrophic abnormalities in development such as congenital fibrosis of the extraocular muscles. In addition, congenital and acquired ptosis in children may be due to congenital or acquired myasthenia gravis, or to loss of innervation to the Müller muscle or the levator muscle. Mechanical factors may also contribute to ptosis such as relative enophthalmos following an orbital fracture, or ptosis may be due to eyelid tumors such as capillary hemangiomas.
Astigmatism and amblyopia occur as a result of blepharoptosis.12,13,14,15 Surgical repair of blepharoptosis does not mitigate the need to treat amblyopia. Indeed, amblyopia treatment in young patients may become more necessary after surgical treatment of ptosis.12 Any noted increased astigmatism after surgical repair of ptosis may be transient.16 Severe ptosis has been shown to produce axial myopia in experimental animals.17 Surgical correction of blepharoptosis can be undertaken at any age. Intervention may be required by the discovery of significant amblyopia, anisometropia, or strabismus. Correction of ptosis will not alleviate any of these conditions, which, once developed, must be treated directly. Severe bilateral ptosis in an infant may delay walking because of the balance problems produced by an extreme chin-up head position.
Blepharoptosis has other physical, functional, and psychological consequences, especially for the school-age child.
Although surgical correction of ptosis may be undertaken at any age, surgery is frequently delayed until a child is 3 to 5 years old unless associated conditions dictate earlier intervention. At age 3 to 5 years, more accurate preoperative measurements can be obtained and postoperative cooperation of the child can be expected. Earlier intervention, however, can lead to equally acceptable results and is recommended by many surgeons. Parents often are anxious for early surgical intervention but can be reassured that it is not mandatory in most cases.
Many methods of blepharoptosis repair have been described in detail. The procedure chosen depends on treatment goals, the underlying diagnosis, degree of levator function, and the skill and experience of the surgeon. Surgical treatment results can be pleasing to the patient and to the family (Fig. 104-1).
DIAGNOSTIC EVALUATION OF THE CHILD WITH PTOSIS
A thorough patient history, family history, review of systems, drug or allergic history, and general physical examination should be done in all cases. Historical emphasis should be placed on aspirin and ibuprofen intake, anticoagulant use, or bleeding tendencies. The examiner should inquire into the possible association of childhood blepharoptosis with malignant hyperthermia and cardiac disorders18,19,20 A review of family members’ experiences with anesthesia can be helpful in that regard. Any previous adverse anesthetic event in a patient or family member should be researched in detail. When the clinical history suggests an increased risk for malignant hyperthermia, susceptibility testing may be initiated.21 The anesthesia team may select anesthetic agents felt less likely to trigger malignant hyperthermia.22
Although most patients with congenital ptosis will have a history of slight worsening with fatigue, a history of significant ptosis variability or alternating ptosis necessitates diagnostic studies for myasthenia gravis. Alternating blepharoptosis is caused by myasthenia gravis in virtually all cases. Review of family photographs assists in the determination of onset or variability of ptosis. Tests that can be performed to establish the diagnosis of myasthenia gravis include the ice test, rest test, Tensilon test, and Neostigmine tests. Tests for acetylcholine receptor antibodies are rarely positive in isolated ocular myasthenia gravis, especially in childhood, but if positive are strongly indicative of the presence of myasthenia gravis.23 A positive Tensilon test, abnormal single-fiber electromyography (EMG) recordings, and therapeutic responses to anticholinesterase medicines or corticosteroids establish this diagnosis. If myasthenia is strongly suspected, a trial of Mestinon or corticosteroids is indicated.
A history of progressive, otherwise unexplained, bilateral ptosis suggests that an electrocardiogram and perhaps an electroretinogram should be obtained to rule out a mitochondrial disorder such as Kearns-Sayre syndrome.
Visual acuity and refractive error should be recorded. A thorough eye examination, particularly for strabismus, corneal sensitivity, and retinal dysfunction, should be done. Tear function is measured if any doubt as to the adequacy of tear production exists. However, reliable Schirmer testing is difficult in a younger child. Examination of the tear film and careful evaluation of the cornea for any signs of exposure both preoperatively and postoperatively is necessary. Patients with diminished corneal sensitivity due to innervational abnormalities are at increased risk of exposure keratopathy following surgery for correction of congenital ptosis. Corneal sensation can easily be determined using a simple wisp of cotton at the tip of a cotton-tip applicator applied to the corneal surface. Abnormal corneal sensitivity cautions the surgeon to avoid surgery or reduce the amount of ptosis correction.
A full examination of the extraocular movements is necessary. In addition to specific examination of the function of the superior rectus muscle, one should also check the Bell phenomenon (upward deviation of the eye during forced lid closure). A normal (present) Bell phenomenon is important because increased lagophthalmos is common after surgical repair of congenital ptosis. A normal Bell phenomenon and normal superior rectus muscle function allow for protection of the cornea postoperatively.24 With reduced superior rectus muscle function, the surgeon should be more conservative in the amount of surgery performed to correct the ptosis.
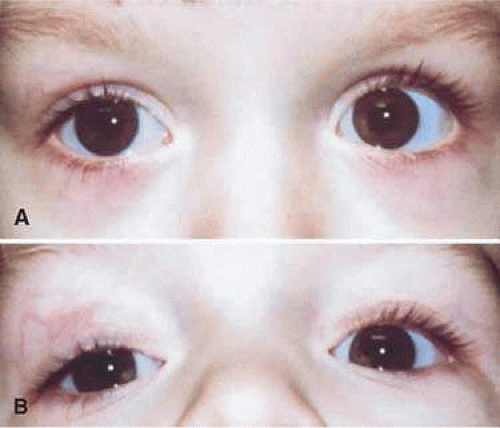
Occasionally, the examiner must determine whether apparent ptosis is in fact pseudoptosis or whether lid retraction of the contralateral upper lid exists. To detect pseudoptosis, lid height should be observed and measured with each eye fixing on a distant target. Lid position in downgaze should be noted. If ptosis is congenital, the ptotic lid may appear higher than the contralateral upper eyelid in downgaze (lagophthalmos). However, a ptotic lid does not appear higher than the contralateral eyelid in downgaze when ptosis is acquired. If lid retraction is present, the higher lid remains higher in downgaze (Fig. 104-2).
Lid height, contour, crease, lash position, and any special facial characteristics should be noted and recorded. The lid margin-reflex distance (MRD) should be measured. The MRD is the distance from a corneal light reflex to the upper eyelid margin with the patient’s eyes in primary gaze. The amount of levator excursion also should be measured. This can be difficult in younger children and infants. In congenital ptosis the degree of ptosis inversely correlates with the amount of levator function. Serial examination at separate visits assists the surgeon obtaining reliable measurements of the true amount of ptosis and levator function. The patient is asked to look downward, as a ruler is positioned with a mark adjacent to the upper lid margin. With the examiner’s hand eliminating brow movement by the patient, the patient is looks upward as far as possible without changing head position. The amount of eyelid excursion is measured directly from the ruler and is recorded in millimeters of levator function.
The amount of surgical correction desired is determined and the procedure decided. Although refinements often are made during the surgical procedure, the preoperative plan remains the foundation for the overall procedure. If a patient has an anomaly of the globe, orbit, or lower lid, the corneal light reflex–lid margin measurements can be helpful in positioning the ptotic lid (Fig. 104-3).
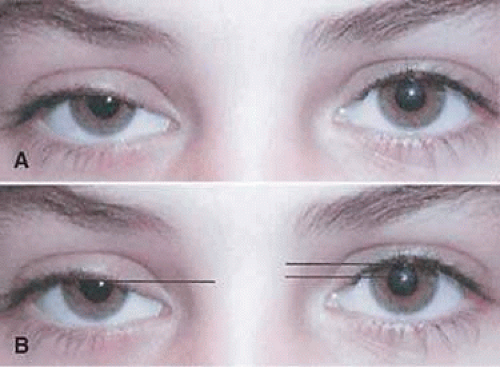 Figure 3. A. Congenital blepharoptosis, right upper lid, level gaze. B. Congenital blepharoptosis, right upper lid. Illustration of lid margin–corneal light reflex relationships. |
In patients with Marcus Gunn jaw winking ptosis, the amount of eyelid retraction with movement of the jaw should be evaluated. In those patients with mild retraction ptosis repair should be undertaken using standard amounts of surgery dependent on the degree of ptosis. An external levator resection is the usual procedure. If significant retraction is present, extirpation of the involved levator muscle combined with a frontalis suspension should be performed. Failure to extirpate the involved levator muscle will result in persistent wink.
In addition to measuring levator function, the eyelid should be assessed with its response to phenylephrine. One drop of 2.5% is instilled into the lower cul-de-sac in younger children and infants. A second measurement of the MRD is made after approximately 5 minutes. If the eyelid elevates to a near normal position, tightening or resection of the Müller muscle is considered for ptosis repair.
All patients with congenital ptosis require repeated visual acuity testing and repeated determinations of refractive error. Amblyopia frequently occurs secondary to strabismus, induced astigmatism, and less commonly due to occlusion of the line of sight.25 The presence of a chin elevation may allow for peripheral fusion but does not exclude the presence of amblyopia.26
TIMING OF SURGICAL INTERVENTION
In most situations congenital ptosis is repaired between the ages of 3 and 5 years. Severe ptosis causing occlusion amblyopia may be surgically repaired when recognized. Nevertheless, most ptosis-associated amblyopia is caused by induced astigmatism. If a significant astigmatism develops, spectacle correction and amblyopia therapy should be instituted.
SURGICAL PROCEDURES
There are many options for the surgical correction of congenital ptosis. The two main types of treatment involve levator resection procedures and frontalis suspension procedures. The Müller muscle procedures (Fasanella Servat and Müllerectomy) may be used for the correction in mild ptosis, particularly neurogenic ptosis associated with Horner syndrome.
Levator Muscle Procedures
Indications
Levator aponeurosis/muscle shortening procedures are performed in cases of mild to moderate ptosis. Shortening of the levator-aponeurosis complex through a lid-crease incision with minimal disturbance of the other lid tissues offers the best possible physiologic and cosmetically desirable result of ptosis repair in children. The skin incision is hidden either in the existing lid fold or, if necessary, in a new lid fold created to match that of the fellow lid. The resultant scar in the lid crease is not visible when the eyes are open and is recognized only by close inspection of the closed lid.
The amount of levator-aponeurosis complex resected significantly differs in children with congenital blepharoptosis compared with adults with acquired blepharoptosis. Although a 1-mm resection generally elevates the eyelid 1 mm in adults, such surgical numbers do not apply to most pediatric ptosis patients. A more generous resection is required in children and is dependent on the amount of levator function measured (Table 104-1).
Table 104-1. Ptosis Repair Based on Levator Muscle Function and Amount of Ptosis | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Although classic levator dehiscence can be encountered in the pediatric population, more commonly decreased levator excursion and levator muscle dysgenesis is encountered. Adequate levator function must be present to offer the ptosis patient a reasonable chance for correction with a levator resection. Most authors are in general agreement as to how much levator function is required to justify this procedure. A measurable lid excursion from downgaze to upgaze of 4 mm or less (measured with the ipsilateral brow stabilized) is too poor to permit lasting results from levator resection. If levator function is more than 4 mm but less than 6 mm, a “large” levator resection is recommended. If levator function is between 6 and 8 mm, a “moderate” levator resection is indicated. If levator function exceeds 8 mm, a smaller resection of the levator is indicated. A large levator resection is defined as 22 mm or more, moderate as 16 to 18 mm or more, and small as 13 to 15 mm.
Other clinicians prefer to measure the height of the lid margin relative to the corneal light reflex or corneoscleral limbus or the width of the palpebral fissures and to use these measurements as guides to the amount of surgery to be performed.27 Lid height relative to the cornea can be measured toward completion of the surgical procedure using either Berke recommendations or the lid margin to corneal reflex distances, thus allowing further refinement in the amount of levator aponeurosis to be resected.
Contraindications
The external levator resection surgical technique is applicable to a patient of any age, but it is best used when levator function can be assessed accurately. As a practical guide, correction of congenital blepharoptosis with this approach is often done in the preschool age group, when the patient is 3 to 5 years old. At these ages, accurate visual acuities have been obtained, amblyopia has been identified and treated, and reliable measurements of levator function have been recorded. However, amblyopia or parental concerns often dictate earlier intervention.
An external levator resection is not indicated when levator function is less than 4 mm. In such cases, immediate postoperative results might be acceptable after a large levator resection. but experience has shown that the lid height will not be maintained over time.
Any ptosis procedure performed in a patient with a poor Bell phenomenon, limited elevation of the eye, reduced corneal sensitivity, or poor tear production risks development of postoperative corneal complications.
Surgical Technique for External Levator Resection
Preoperative photographs are helpful, in that such photographs can become part of the patient’s permanent record and can be referenced during the surgical procedure as desired. Used properly, these photographs can help prevent inadvertent surgery on the wrong lid and can assist the surgeon with the artistic portions of the procedure, such as lid contour and lash position.
General anesthesia is required for children.
When ptosis is bilateral, bilateral symmetric or asymmetric levator resection usually is done in one surgical procedure. If ptosis is severe but still amenable to levator resection, the surgeon may prefer to operate first on the more severely involved side. After healing has occurred and final lid position is established, surgery can then be performed on what was the less ptotic eyelid. Symmetry of eyelid height is more easily achieved by proceeding in this order.
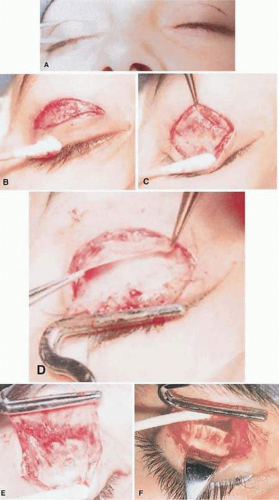
Existing lid creases are outlined on both lids with a fine-tipped surgical marking pen (Fig. 104-4A). If the existing lid crease does not match the desirable lid crease in the fellow eyelid, a new position is marked. The marked areas are compared visually and are measured with a ruler or calipers for accuracy. As a general rule, it is easier to raise the level of an eyelid crease than to lower an eyelid crease. In children without ptosis, the height of the center of the lid crease averages 6.5 to 7 mm from the lid margin. The nasal and temporal ends of the lid crease measure about 4.5 to 5 mm above the lid margin in children. The height of the lid crease increases with age and is about 10 mm centrally in adults.
After the proposed incision site has been marked and verified, the lid to be incised is further isolated from the fellow eye with appropriate drapes. A corneal protector is inserted between the eyelids. A solution of 1% lidocaine containing epinephrine in a concentration of 1:100,000 is injected underneath the skin and orbicularis muscle at the eyelid mark. A 30-gauge needle held parallel to the lid is used for the injection. Only one or two skin punctures are required to traverse the length of the incision with a short needle. No more than 0.5 mL of solution is required, because larger volumes of fluid only distort the anatomy of the lid. If the needle tip is just beneath the skin, it is in the orbicularis layer because no subcutaneous fat exists in the lids. The lidocaine-epinephrine solution facilitates dissection and reduces bleeding. Some surgeons omit this step if the patient is under general anesthesia, whereas others use it in virtually all cases. Epinephrine is omitted from the injection if any risk factors regarding its use exist.
An incision is made with a curved scalpel blade from one end of the eyelid mark to the other without raising the blade from the lid. The incision is made more precisely if it is made from a nasal to temporal direction; in this instance, the surgeon places slight tension on the temporal side of the lid as the blade is drawn from nasal to temporal. This prevents the loose skin of the upper lid from gathering beneath the knife and forcing the blade away from the mark. The depth of the incision should extend through the skin and into the orbicularis layer. (Fig. 104-4B). If the incision is only skin deep, further dissection through orbicularis is completed with sharp dissection. A fine-tip monopolar cautery is often used during dissection through orbicularis as this reduces bleeding. Caution is required to avoid too deep a dissection. Skin hooks help the assistant to provide adequate exposure. After dissection extends beneath the orbicularis layer, the aponeurosis-orbital septum is encountered. The surgeon must be aware that the orbital septum may extend farther than usual toward the lid margin, particularly in Asian patients and in patients who have blepharophimosis.3
When the depth of the dissection extends beneath the orbicularis, the orbicularis is undermined superiorly until the orbital septum is identified (Fig. 104-4C). The septum is then elevated at its inferior margin from the anterior surface of the levator aponeurosis, usually without disruption of retroseptal fat (Fig. 104-4D). Orbicularis is undermined inferiorly to near the lid margin. As undermining continues, lash follicles may become visible inferiorly, which indicates that dissection is adequate. Lash follicles should not be incised or cauterized.
With the orbicularis exposed from its posterior surface, the remaining attachments of the aponeurosis to the anterior surface of the tarsal plate can be elevated. Holding blunt-tipped scissors vertically with the tips parallel to the skin incision and making a series of small cuts across the length of the tarsus best accomplishes aponeurosis elevation. If this incision is started at about the vertical center of the tarsus, little aponeurosis tissue is encountered with the first series of snips. Aponeurosis becomes quite thin as it terminates on the anterior tarsal surface. The tarsal plate should not be incised. Incised aponeurosis tissue is gently elevated and undermined, further separating it from the tarsus. Meibomian glands become visible within the tarsal plate as the dissection proceeds. With each millimeter of vertical progress, aponeurosis tissues become thicker.
Here the surgeon has a choice of dissection of the levator tendon off of the Müller muscle or dissecting posterior to the Müller muscle. Bleeding is encountered at the superior tarsal border as the Müller muscle is exposed (Fig. 104-4E). Bleeding is minimal if dissection has been made in the correct tissue plane. If the Müller muscle is incorporated in the dissection, then it is undermined and elevated from the only remaining deeper layer, the palpebral conjunctiva. At this point, dissection usually can be accomplished with a moistened cotton-tipped applicator. Blunt dissection is facilitated by clamping the distal free end of the dissected levator aponeurosis with a clamp of the type devised by Berke for this purpose. An Ehrhardt clamp placed on the lid further facilitates this part of the dissection (the corneal protector can be removed while this clamp remains in place; Fig. 104-4F). Alternatively, the levator aponeurosis is separated from the tarsal plate and dissection in the plane between the Müller muscle and levator aponeurosis is carried superiorly. Superiorly the origin of the Müller muscle is separated from the levator tendon and dissection continued to separate the tendon from the underlying conjunctiva. The origin of the Müller muscle is sometimes reattached to the levator tendon a few millimeters superior to the planned level of resection of the levator tendon.
Vertical parallel incisions are made at the temporal and nasal borders of the levator-aponeurosis tongue of tissue held in the ptosis clamp. These incisions are made under direct view and control approximately at the nasal and temporal limits of the horizontal skin incision. Attention is paid to the location of the superior oblique tendon nasally and the lacrimal gland temporally. Additional dissection is carried out beneath the levator, and the vertical incisions are extended until the desired length of levator muscle is free from surrounding attachments. It is important to free only the amount of levator desired because excessive dissection in the area of the Whitnall ligament (superior suspensory ligament) may diminish the results. Moreover, if the superior conjunctival fornix is totally freed from surrounding attachments, the probability of postoperative conjunctival prolapse increases.
After dissection of levator aponeurosis and muscle, the Ehrhardt clamp is exchanged for the corneal protector. Three double-armed (spatula needles) 5-0 or 6-0 synthetic absorbable sutures are passed across the upper one third of the tarsus parallel to the lid margin. About 3 mm of tarsus is included in each bite. The corneal surface of the tarsus is inspected to ensure that the sutures were placed within but not through the tarsus. Sutures placed too deep will be exposed on the ocular surface of tarsal conjunctiva where they may produce corneal erosion.
Needles on the sutures are brought through the tongue of the levator-aponeurosis complex at a measured height, emerging on the external levator surface (Figs. 104-5A and B). A single overhand knot is tied, opposing the aponeurosis and the tarsal plate. Clamps and corneal protector are removed, and the lid is inspected for height and contour. The single-throw knot may be untied and the sutures repositioned in the levator if modifications in height or contour are required. It is better to reposition the sutures in the levator-aponeurosis complex and not in the tarsus because the tarsal plate will not accept repeated passes of a needle without becoming friable and unreliable in retaining a suture. When the surgeon is satisfied with the lid position, a second throw is added to the knot, forming a square knot on the external levator-aponeurosis surface (Fig. 104-5C). Redundant levator muscle and aponeurosis distal to the sutures can then be excised (Fig. 104-5D).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree