Fig. 18.1
Melanoma of the peri-limbal bulbar conjunctiva with “feeder vessels” entering the tumor
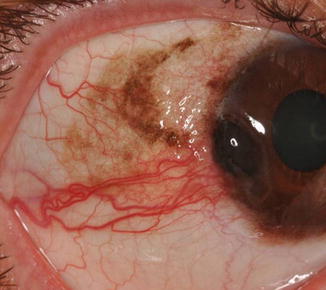
Fig. 18.2
Conjunctival PAM with atypia, which has progressed to corneo-limbal melanoma
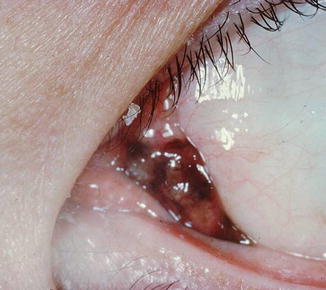
Fig. 18.3
Melanoma of the plica semilunaris
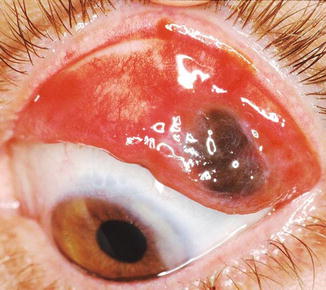
Fig. 18.4
Melanoma of the upper palpebral conjunctiva
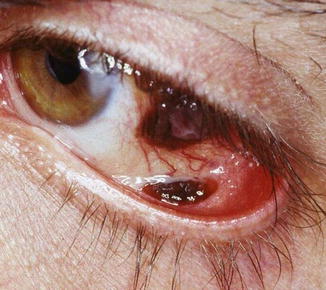
Fig. 18.5
Multifocal melanoma arising from PAM with atypia, with a tumor in the bulbar conjunctiva and a tumor in the lower fornix
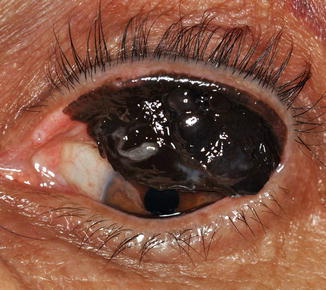
Fig. 18.6
Extensive conjunctival melanoma with involvement of the entire upper palpebral conjunctiva, which could be seen only upon inversion of the upper eyelid. No involvement of the bulbar conjunctiva was noted, leading to a delay in diagnosis
18.5 Variants
18.5.1 Conjunctival Melanoma Associated with PAM
Microinvasive melanoma arising in PAM with atypia may be difficult to identify clinically; therefore, one should search carefully for subtle placoid thickening within the area of PAM. The more dramatic clinical evidence for development of melanoma from PAM is the sudden emergence of one or more nodules in the otherwise flat lesion [15, 16]. Multifocal melanomas usually are associated with PAM with atypia and may appear simultaneously or sequentially in different parts of the conjunctiva [15, 16]. Melanoma with PAM may also involve the adjacent eyelid skin [11].
18.5.2 Conjunctival Melanoma Associated with Nevus
Conjunctival melanoma that arises from a nevus or de novo appears clinically as a solitary pigmented or nonpigmented smooth vascularized nodule, commonly in the limbal area; rarely are these lesions pedunculated [1].
18.5.3 Primary Melanoma of the Cornea
Primary melanoma of the cornea is very rare, although several cases have been reported [1]. Many of these cases represent examples of corneal invasion from a limbal melanoma. It is important to remember that Bowman’s layer is a barrier to the penetration of surface malignancy into the corneal stroma. Therefore, pigmented neoplasms affecting the cornea are usually superficial to Bowman’s layer unless this tissue has been violated by previous surgery.
18.6 Differential Diagnosis
Any conjunctival pigmented lesion may simulate conjunctival melanoma.
18.6.1 Conjunctival Nevus
Conjunctival nevi may be elevated and dark, and in the absence of cysts (typical of compound conjunctival nevi), it may be difficult to differentiate the nevus from melanoma by clinical examination alone [16]. Most conjunctival nevi are noticed in childhood and adolescence. Therefore, any newly-elevated pigmented conjunctival lesion that develops in adulthood should be viewed with suspicion. Conjunctival nevi almost always develop in the bulbar conjunctiva and caruncle. Therefore, any pigmented lesion presenting in the palpebral conjunctiva or fornix should be considered suspicious for melanoma [16].
18.6.2 Epithelial Tumors (Nonmelanocytic)
Nonmelanocytic epithelial tumors such as squamous papilloma, conjunctival intraepithelial neoplasia, and invasive squamous cell carcinoma may acquire melanin in darkly-complexioned individuals [17].
18.6.3 Intraocular Tumors
Epibulbar extension of uveal melanoma or melanocytoma should also be considered in the differential diagnosis of conjunctival melanoma [18]. In these cases, the trans-scleral nature of the lesion can be identified by high-frequency ocular ultrasound.
18.7 Histopathologic Features
18.7.1 Light Microscopy
The definitive diagnosis of conjunctival melanoma is made by histopathologic examination. Most cases can be diagnosed with confidence by light microscopic features (Fig. 18.7). Four types of atypical melanocytes have been described in conjunctival melanoma: small polyhedral, spindle, balloon, and round epithelioid cells with eosinophilic cytoplasm [16]. The invasive melanoma is often accompanied by the intraepithelial PAM with atypia, which may be the precursor to the melanoma (Fig. 18.8). Any breeching of the basement membrane by atypical melanocytes in PAM with atypia should be considered as invasive melanoma. The presence of atypical melanocytes within the epithelium of subepithelial cysts is considered to represent PAM with atypia and not invasive melanoma. The common presence of cystic and solid epithelial inclusions in compound conjunctival nevi may confuse a pathologist without experience in ophthalmic pathology, who may consider these findings as signs of malignancy.
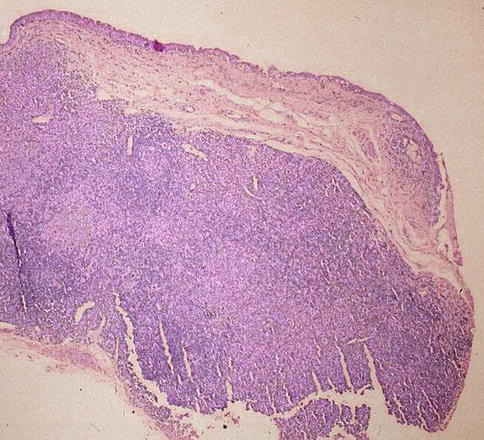
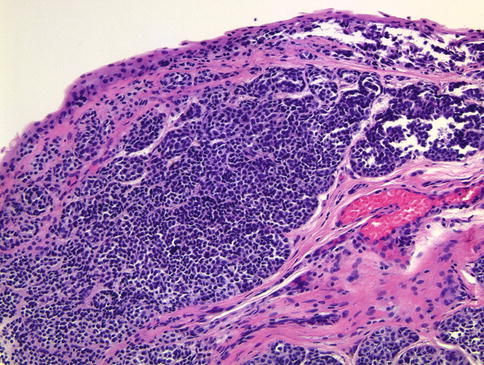

Fig. 18.7
Low-magnification histological picture showing thick conjunctival melanoma involving the specimen’s margin (hematoxylin and eosin; original magnification ×4)

Fig. 18.8
This photomicrograph illustrates conjunctival melanoma arising in the context of PAM with atypia. Note the lack of maturation or architectural organization from top to bottom and the presence of atypical intraepithelial melanocytes that do not appear to be cohesive or to have any architectural relationship with the epithelial cells (hematoxylin and eosin; original magnification ×20)
18.7.2 Immunohistochemistry
If in doubt, immunohistochemical stains such as HMB-45 or MELAN-A, either individually or in a cocktail, can be used to demonstrate the presence of melanocytes [20–22], and Ki-67 proliferation index may help to separate melanoma from nevi [23]. BRAF mutations were found in about half of conjunctival melanoma cases, while BRAF mutations are seldom encountered in uveal melanomas [24, 25]. Conversely, GNAQ mutations that are detected in uveal melanomas are not encountered in conjunctival melanomas [26].
18.7.3 Histopathologic Prognostic Factors
There are several histopathologic features that predict adverse prognosis of conjunctival melanoma. The major prognostic factor predictive of metastasis is the depth of invasion, measured with an ocular micrometer from the top of the epithelium to the deepest point of invasion. The other important histologic prognostic factors are presence of pagetoid spread, mixed cell tumors versus spindle cell tumors, histologic evidence of lymphatic invasion, high number of mitotic figures, and high cell proliferation index using immunohistochemical stains such as PCNA [1, 11]. Ulceration in conjunctival melanoma also was found to be an important histopathologic predictor for more aggressive tumors [27]. The presence of PAM does not appear to be a prognostic indicator [11].
18.8 Clinical Course
18.8.1 Local Recurrence
Local recurrence of conjunctival melanoma has been reported in 56–65 % of the patients and nearly half of these patients develop more than one recurrence [1, 3, 4, 28]. The mean interval between the first treatment and the first recurrence is 2.5 years. Some authorities assert that patients treated by surgical excision alone have more recurrences than those receiving adjuvant treatment such as cryotherapy or brachytherapy, but it is not known if this assertion applies to surgical resection with clear margins. For example, patients with multifocal disease, usually originating in PAM with atypia, are prone to develop recurrences because it may be difficult to ensure a clear resection margin. Other risk factors for developing recurrences are melanoma located other than at the limbus, and involvement of surgical margins [28]. Therefore, the pathologist’s report should specifically comment on the involvement of the margins. Local recurrences are managed as the primary melanoma.
18.8.2 Local Spread
Conjunctival melanoma may spread locally in the conjunctiva before the onset of regional and systemic metastases [29]. “In-transit” metastases of conjunctival melanoma, which are believed to represent local lymphatic spread within the conjunctiva, have been described [30]. Dissemination of melanoma cells at the time of tumor excision has also been reported. The spread of conjunctival melanoma through the nasolacrimal duct to the nasal cavity and paranasal sinuses has been attributed to shedding of exfoliated melanoma cells in the tear film, by direct extension, or as regional hematogenous metastases [1]. Epistaxis or epiphora may serve as a sign of extension and recurrence of the conjunctival melanoma in the nasolacrimal system [31]. Rarely, conjunctival melanoma invades the eyeball or extends directly into the orbit [1, 32].
18.8.3 Regional and Distant Metastasis
Conjunctival melanoma can metastasize to any organ in the body. In about half of the patients with metastases, regional lymph node metastases are detected before systemic ones [28, 33]. Other common locations are the brain, liver, and lung [28]. It is important, however, to emphasize that in conjunctival melanoma, the most common primary locations of the metastases are the regional parotid (preauricular) and submandibular lymph nodes. This reflects an important difference between conjunctival and uveal melanoma, which tends to disseminate almost exclusively to the liver as a first site of dissemination. Therefore, ophthalmologists should be specific when referring patients with conjunctival melanoma to oncologists and avoid applying the general term “ocular melanoma” lest the oncologist assume incorrectly that the first target for metastasis is likely to be the liver as is the case with uveal melanoma.
18.9 Treatment
18.9.1 Surgery
The primary treatment of conjunctival melanoma is surgical excision of the entire tumor with surgical margins that are as wide as possible; some authorities recommend an excision margin of 3–5 mm, and when this is not feasible, adjunctive treatment will be necessary. Most surgeons will add an adjuvant treatment to the primary excision as a matter of routine. The most common supplemental treatment is cryotherapy to the surgical margins and/or to the surgical bed [15]. When deep limbal and scleral involvement are suspected, scleroconjunctivectomy should be considered [34].
Some surgeons advocate supplemental brachytherapy, usually using beta irradiation [35], and some surgeons use absolute alcohol to devitalize corneal epithelial cells adjacent to a limbal melanoma before excision [34]. Areas of PAM with atypia, either around the excised melanoma or distant from it, must be treated because these lesions may be the source of recurrent melanomas. The PAM can be treated by surgery, cryotherapy, or brachytherapy, and in recent years topical chemotherapy using mitomycin C has been advocated. (See Chap. 17).
18.9.2 Cryotherapy
Cryotherapy, using a double freeze-thaw cycle, should not be used as a primary treatment modality.
18.9.3 Radiotherapy
Conjunctival melanoma is not radiosensitive. Therefore, brachytherapy should not be used as the sole treatment modality [35]. In recent years brachytherapy, usually using beta radiation, was found to be superior to cryotherapy as adjuvant therapy to surgical excision [36–38]. In addition, proton beam radiotherapy has been advocated in treating extensive conjunctival melanoma as an alternative to exenteration [39].
18.9.4 Topical Chemotherapy and Immunotherapy
In recent years topical mitomycin C [40–42] and topical interferon alpha-2b [43, 44] have been used successfully as adjuvant treatment to surgical excision in treating conjunctival malignant melanoma.
< div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


