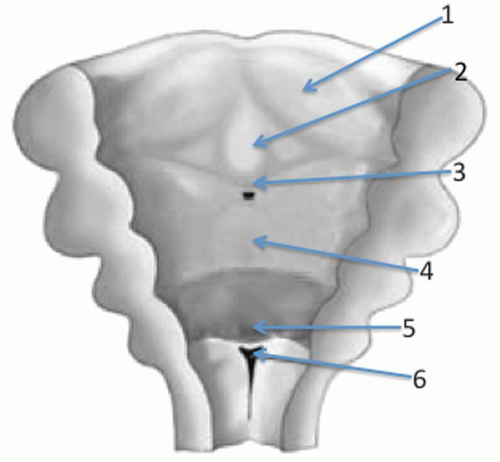
is complex, and the numerous steps involved are detailed elsewhere in this text.
TABLE 87.1 TYPES OF CONGENITAL AERODIGESTIVE TRACT ANOMALIES BY ANATOMIC ZONE | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
within the esophagus results in a muscular layer of striated muscle in the upper one-third and smooth muscle in the lower two-thirds of the tube (16).
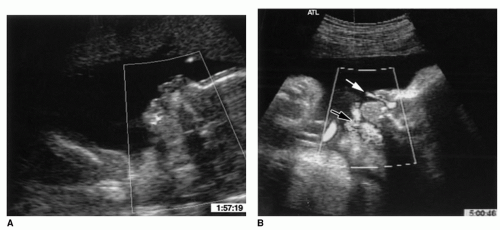 Figure 87.3 Parasagittal fetal ultrasound around 19 months depicting the fetus (A). Power Doppler imaging demonstrates amniotic fluid flowing through the mouth and nose (B) during swallows (black arrow) and respiration (white arrow). From Miller JL, Sonies BC, Macedonia C. Emergence of oropharyngeal, laryngeal and swallowing activity in the developing fetal upper aerodigestive tract: an ultrasound evaluation. Early Hum Dev 2003;71(1):61-87, with permission. |
imaging. In an unstable patient, diagnosis and management often occur at the same time in the operating room in the process of securing a safe airway.
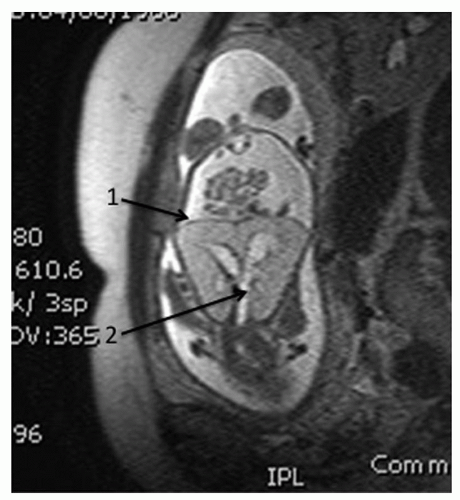 Figure 87.4 Fetal T2 FSE coronal MRI demonstrating (1) flattened diaphragms and (2) a dilated distal tracheobronchial tree. The 22-week fetus is inverted in this image. |
may also guide airway management when the patient undergoes treatment in the operating room.
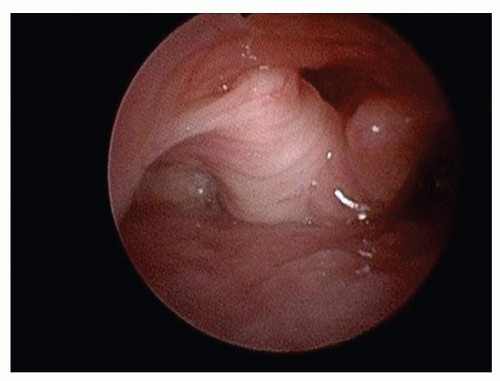 Figure 87.5 Flexible endoscopic view of the larynx in a supine infant demonstrating laryngomalacia secondary to bulky, obstructing supra-arytenoid tissue. |
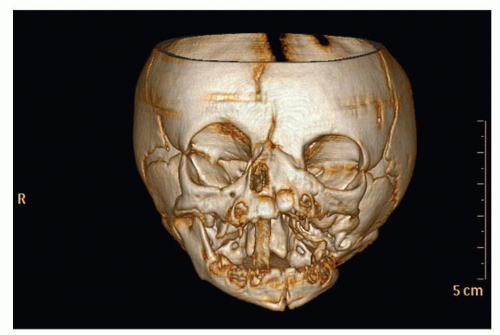
nasal passageway secondary to choanal stenosis or atresia. A high-resolution maxillofacial CT scan with axial, coronal, and sagittal reconstructions and/or three-dimensional (3-D) reconstruction is the single most useful radiographic study in the evaluation of congenital nasal anomalies because of its superior bony/soft tissue resolution (33,34). Rigid nasal endoscopy adds little to the diagnosis, but it is a platform from which both diagnosis and treatment can be instituted.
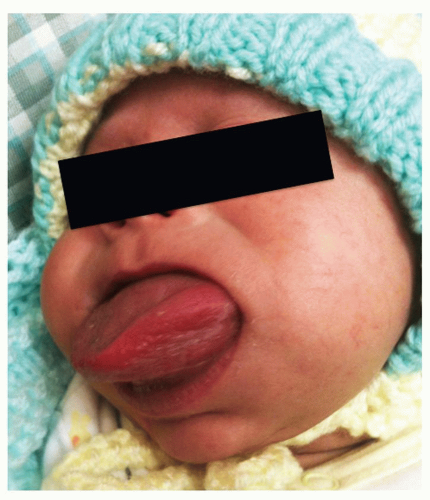
(37,38). The symptoms and signs of minor anomalous bands largely relate to feeding, as the inability to evert the upper lip or protrude the tongue may preclude effective breastfeeding. Airway and voicing are unaffected. Similarly, cleft palate (without micrognathia and/or relative macroglossia) is mostly a feeding problem at birth. In contrast, microstomia, intergnathic bands, micrognathia with relative macroglossia, absolute macroglossia and intraoral tumors produce drooling, gasping during feeding, muffled cry, and obstructed breathing patterns. When no oropharyngeal obstruction is present, quiet breathing through the nose may be unaffected. Nasopharyngoscopy rules out oropharyngeal extension, but the majority of the salient physical exam can be accomplished through direct observation with a tongue depressor and a headlamp. Consistent with the heterogeneous nature of congenital oral anomalies, there is no single imaging modality that is required or preferred for diagnosis. When the physical examination alone is insufficient, mandibular or dentigerous anomalies may be evaluated best with a panorex. Relationships between the upper and lower jaws and the encompassed soft tissue are best shown on a maxillofacial CT scan, while intrinsic soft tissue lesions (such as lingual masses) achieve best delineation on MRI (39). For vascular malformations, MRI adds the benefit of vascular phases that provide additional diagnostic sensitivity (MR angiography [MRA] and MR venography (40)).
larynx include dysphagia and aspiration with or without coughing and recurrent aspiration pneumonias. Stridor is present and is classically described as inspiratory when the lesion is supraglottic or glottic and biphasic when the lesion is subglottic. Stridor may progress to retractions and cyanosis. When stridor is absent at birth but appears in the first few weeks or months of life, laryngomalacia is the most likely cause, although subglottic hemangiomas also usually present with some delay. Vocal dysfunction due to laryngeal disease may be muffled or coarse if there is a large supraglottic lesion, hoarse, breathy, or aphonic if the disease is glottic, and hoarse and breathy if the disease is subglottic.
 Get Clinical Tree app for offline access 
|