18

Complications, Management,
and Avoidance
The endonasal sinus surgery developed by Halle1–4 and Hajek1,2,5–7 at the beginning of the 20th century was abandoned in the 1930s due to severe complications. Mosher,8 a pioneer of this surgical technique in the United States, wrote in 1929 “Theoretically the operation is easy. In practice, however, it was proved to be one of the easiest operations with which to kill a patient.” Because of this, the endonasal operation techniques were not found and described in surgical textbooks. Heermann9 was one of the few who never gave up the endonasal technique. He was also the first to revive the endonasal technique by introducing the surgical microscope for endonasal sinus surgery, in 1958. Without a doubt, however, the revitalization of the endonasal operation method is due solely to Messerklinger10–15 and his constant application of Hopkins’s optical devices in rhinological diagnosis, as well as the development of the later-named infundibulotomy. Wigand and colleagues,16,17 Kennedy,18 Stammberger,19–21 and Draf22 helped this idea become accepted at last.
Currently, extranasal operative techniques no longer play a major role in the surgical treatment of inflammatory sinus diseases. Complications peculiar to this technique, such as swelling of the cheek, scar abscess, and symptomatic trigeminal neuralgias, are no longer observed. But the expectations of endoscope- and microscope-assisted endonasal operative techniques reducing the number of orbital or endocranial complications have not been met. One reason is because the absolute number of paranasal sinus interventions has increased greatly since the reintroduction of the endonasal technique due to the significantly expanded number of indications for surgery. Between 1982 and 1985, 1204 patients were operated on at the ENT department of the University of Kiel, during which time the operations were in part performed extranasally. Between 1991 and 1994, 2178 patients underwent endonasal sinus surgery.23 Most complications are caused by beginning and inexperienced surgeons, because the supervision by an experienced surgeon is limited by a narrow surgical approach. There is no doubt that the individual number of complications is decreasing as the surgeons become more experienced.24 While performing more than 1000 endonasal surgeries, the author personally caused only one slight lesion of the dura, whereas the injury rate of the remaining surgeons at the ENT department at Kiel University was 0.85%.23 There are published papers, however, that maintain that the complication rate of surgeons with a great deal of experience can increase again.25–27
The increase of published intra- and postoperative complications may be caused by the increasing number of medicolegal conflicts during the last years. In the meantime, it seems that endonasal surgery constitutes the largest group of medicolegal conflicts in otorhinolaryngology.28
 Classification of Complications
Classification of Complications
The classification can be performed according to different aspects, for example, the location of the injury in the sinus system or according to the injured organs. Often complications are classified as major or minor.29 The category temporary, requires no treatment includes periorbital emphysema and chemosis as well as sensitivity disturbances of the teeth and lips. The category temporary, correctable with treatment includes adhesions, bleeding, broncho-spasm, and postoperative infections of the sinuses. The third group consists of any change that is permanent if it persists beyond 1 year. These are dental or lip pain or numbness, and anosmia. May et al29 divide the group of major complications into correctable with treatment and permanent. The category correctable with treatment consists of orbital hematoma (postseptal), loss of vision, diplopia, CSF leakage, meningitis, brain abscess, focal brain hemorrhage, hemorrhage requiring transfusion, carotid artery injury, and epiphora. Permanent major complications are blindness, diplopia, central nervous system deficit, and death.
The above-mentioned complications are usually early complications. Late complications are recurrences, mucoceles,30–33 and myospherulosis due to ointments and foreign bodies.34–36 Of course, also orbital and endocranial complications can occur as late complications.
Meanwhile, the literature about the complications of endonasal surgery is very extensive and is growing from month to month. A comparison of complication rates of single authors is complicated because of the lack of general standards. For this reason, no table showing such a comparison is given here. The relevant literature is published by May et al,29 Levine,37 Stammberger,38 Rudert,23 and Hosemann and colleagues.39,40 We will start with the orbital complications and the injuries of the lacrimal duct, including bleeding, then turn to the endocranial complications.
Lid Hematoma
Of the minor complications, the most frequent is bleeding into the anterior parts of the orbit. This manifests itself shortly after the operation as a lid hematoma (black eye). It is caused by unintentionally opening the orbit in its anterior part. It happens during either the removal of the uncinate process or the exploration of the maxillary ostium in the anterior rising part of the infundibulum. In most cases, the lid hematoma heals without residuum, if no orbital fat is removed.
THERAPY
Special therapy is not necessary. Repositioning or sealing a potential prolapsus of orbital fat into the nose is not necessary. We also refrain from doing this during the orbital decompression in case of endocrine orbitopathy. Because the opening of the orbit is in the very anterior parts of the orbit in the extraconal space and the medial rectal muscle is not injured, no functional disturbances are expected.
Injury of the Lacrimal Duct and Sac
Injuries of the lacrimal duct are much more frequent than actually diagnosed, because they rarely cause drainage problems. But of 195 patients sent to us for a dacryocystorhinostomy, 29 (14.8%) had a history of one or more sinus surgeries.23 Bolger et al41 performed a fluorescein rinse of the lacrimal duct in 46 patients directly after endonasal sinus surgery. In seven patients, they found lesions of the lacrimal duct by detecting fluorescein in the middle nasal duct. In three of the seven cases, a fluorescein drainage into the middle nasal duct was observed even some months after surgery. None of the patients showed a stenosis of the lacrimal duct.
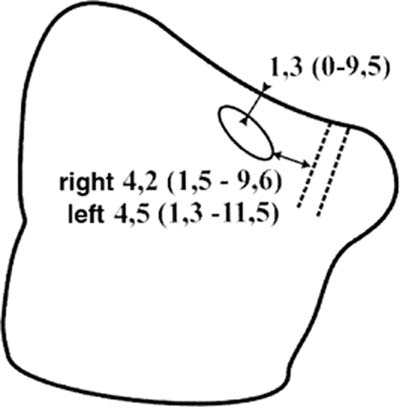
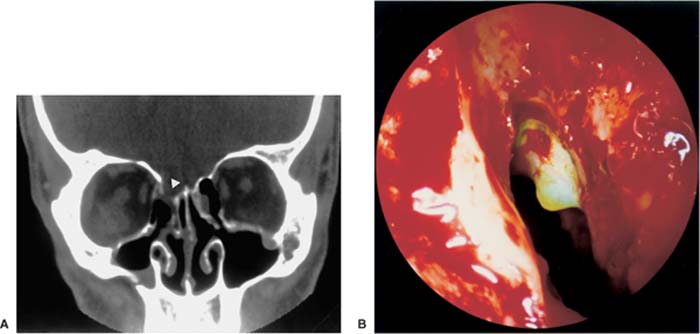
Injuries of the lacrimal sac occur during the removal of the uncinate process, whose insertion at the lateral nasal wall is just a few millimeters behind the lacrimal sac. They also occur during the enlargement of the maxillary ostium in the anterior direction. According to Lang and Papke,42 the distance from the lacrimal sac to the maxillary ostium measures ~4.35 mm (range 1.3 to 11.5 mm) (Fig. 18–1). In the case of a good pneumatization, 30% of the anterior ethmoid cells lie as lacrimal cells and 77% as agger cells above the lacrimal sac.43 Therefore, the sac can also be damaged during the exposition of the frontal recess.
THERAPY
A therapy in the form of an endonasal dacryocystorhinostomy is necessary only when a stenosis with epiphora occurs as a late result of the lacrimal duct injury.
Intraconal Injuries of the Orbit
Injuries of the eye muscles were seen more frequently during the days when predominantly extranasal operations were performed. This is definitely true for disturbances of the superior obliquus muscle after extranasal frontal sinus operations (Killian and Lothrop procedures). During endonasal surgery, only the medial rectal muscle is more endangered. The circumstance that mostly injuries to the right orbit are observed is explained by the fact that the surgeon is usually positioned on the right side of the patient.44–49
FIGURE 18–1 The maxillary ostium is located fairly high and anterior in the maxillary sinus (mean distance to the orbit: 1.3 mm, mean distance to the lacrimal sac: 4.3 mm). The lacrimal sac and the orbit are at high risk if the maxillary ostium is not probed carefully, as described in Figure 14–8.
Damage to the medial rectal muscles is more frequently caused by injuries to the lamina papyracea and the periorbit during surgery of the middle and posterior parts of the ethmoid sinuses than by injuries in the anterior parts of the ethmoid sinuses, because the extraconal layer of fatty tissue beyond the insertion of the medial rectal muscle at the bulb is relatively thin.50 The medial rectal muscle is injured either directly by a surgical instrument (e.g., ethmoid forceps) or indirectly by an injury of the blood or nerve supply. Not recognizing a fat hernia bulging into the ethmoid sinus is another form of damage, if it is interpreted as a nasal polyp. During the removal of this “polyp,” the eye muscle is caught and injured.51–53
Injury to the contents of the orbit occur in cases of anatomical variants of the ethmoid such as Onodi cells, a missing ethmoid bulla, or when the lamina papyracea is missing due to operations or a strong polyposis of the ethmoid. The optic nerve can be injured in the apex of the orbit or in the optic nerve canal if it is bulging medially into the posterior parts of the ethmoid sinuses or is without bony layers (Onodi cell).
THERAPY
The injury of the medial rectal muscle needs urgent therapy in cooperation with the ophthalmologist. Exploring the medial bony orbital wall and the injury of the periorbit through an extranasal approach (Killian procedure) and finding the medial rectal muscle under the surgical microscope is recommended, along with stitching the two remaining bodies of the muscle if necessary. Interventions later on are usually unsuccessful, because the muscles cannot be separated from the surrounding scar tissue.50
An injury of the optic nerve is always associated with an irreversible blindness. There is no therapy.
Retrobulbar Postseptal Hematoma
The occurrence of a retrobulbar hematoma with subsequent blindness is one of the most severe complications of endonasal surgery. According to Stankiewicz,54 there are two types: the slowly developing venous hematoma and the arterial retrobulbar hematoma that develops quickly even during the operation. The venous type develops through a direct injury of intraorbital veins, which means the lamina papyracea and the periorbit must already have been opened. The quickly developing arterial hematoma usually happens when the severed anterior ethmoid artery running through the anterior ethmoid is retracted back into the orbit without opening the lamina papyracea or the periorbit. The symptoms are a significant proptosis, mydriasis, edema of the lid, chemosis, massive increase of bulb pressure, and loss of vision. In case of a venous hematoma, the symptoms develop over a few hours. The sequelae are the same.
The anterior ethmoid artery runs anteriorly to the insertion of the ground lamella of the middle turbinate at the skull base either in the roof of the ethmoid or in a form of a bony mesenterium from the anterior ethmoid foramen obliquely from lateral in the back to medial in the front, where it runs into the olfactory fossa. A smooth cut of the artery leads to heavy bleeding into the anterior ethmoid, which usually stops spontaneously, or by coagulation. This is much easier to perform using the surgical microscope than by endoscope, because you can work bimanually under the surgical microscope. A retrobulbar hematoma develops if the anterior ethmoid artery is not cut sharply, but with dull ethmoid forceps (e.g., ripped out of the surrounding bone), and the remnants of the artery retract themselves into the orbit. The developing retrobulbar hematoma causes choking of the ophthalmic artery. We know from the ophthalmological literature that blindness will be irreversible after 90 minutes unless the pressure is relieved immediately. The loss of vision is caused by the compression of the ophthalmic artery and the posterior short ciliar arteries and by the additional compression of the retinal blood vessels if the inner eye pressure increases to above 80 mm. In a few publications, the incidence of a retrobulbar orbit hematoma is described as being between 0.05 and 0.5%. This was observed 3 times in our patient contingency of 1172 patients between 1986 and 1990, or 0.25% of the cases.23
THERAPY
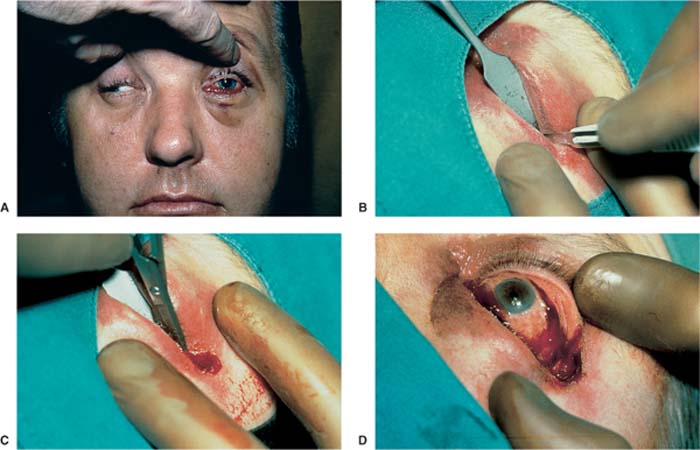
Because the neurosensoric retina can tolerate an ischemic phase for a maximum of 90 minutes without irreversible damage, the therapy must occur immediately. All therapeutic steps are geared to acutely decompressing the orbit. Therefore, massaging the bulb, as recommended by a few authors,54 is strictly rejected by our ophthalmologists. It is extremely unlikely that a hematoma can be moved outside the orbit, because the periorbita is not opened, and therefore the hematoma cannot be drained. The emergency treatment in the form of a lateral canthotomy should be performed immediately, especially because there are no cosmetic or functional disturbances remaining after this intervention. Therefore, the following steps are necessary (Fig. 18–2)55:
1. Extended lateral canthotomy. Because the bulb is encapsulated through the orbital septum like an hourglass and there is a risk of injuring it, the bulb should be protected by a spatula positioned in the outer corner of the eye. The cut is performed down to the bone under a complete horizontal dissection of the lateral lid, either with a scalpel or with straight pointed scissors.
FIGURE 18–2 (A) Patient with a left-sided retrobulbar hematoma. There is a massive protrusio of the left bulb and chemosis. The eyeball is fixed and does not move. The pupil is dilated and does not react to light. (B) Lateral canthotomy. The bulb is protected by a spatula. The cut is carried down to the bone under a complete horizontal dissection of the lateral corner of the eye. (C) The lateral canthal ligament is then cut vertically at the bone edge (upper and lower cantholysis). (D) The bulb is now soft, and the pupil reacts to light again.
2. The lateral tendon of the lid is then cut vertically with the scissors at the bone edge to the top and to the bottom (upper and lower cantholysis). It needs to be proven by palpation that the canthotomy goes down to the bone and that the lateral canthal ligament is cut. Canthotomy is sufficient if you can see the orbital fat in the depth when you separate the upper and lower lids with the fingers.
With these measures, sufficient decompression is usually achieved. Should this not be the case, the next step must follow: a wide transcutaneous cut in the area of the temporal lower lid in the region of the bony orbital limitation, with consecutive separation of the orbital septum with bowed dull scissors, until orbital fat tissue is bulging forward.
The attempt to try decompression by removing the lamina papyracea either endonasally or extranasally and separating the periorbita before lateral canthotomy is frequently not sufficient and leads to a delay of therapy. Of course, this intervention can be performed additionally.
The ligation or coagulation of the ethmoid artery causing the retrobulbar bleeding is not possible, because the vessel is retracted into the orbit. Subsequently, a high-dosage steroid treatment and dehydrating therapy (e.g., with mannitol), should be performed.
Injuries of the Ethmoid Roof and the Cribriform Plate
Iatrogenic lesions of the dura are observed despite the best optical and technical aids, and also despite the good education of the surgeons. The incidence reported in the literature is somewhere between 0.2 and 2.5%.23,25,38,47,49,56–60 In our own patient contingency, the risk, with a large number of surgeons, was 0.85% per patient and 0.5% per operated side. Between 1986 and 1996, 527 patients and 949 sides were operated on by the author himself, with only one CSF fistula. The risk therefore was 0.19% per patient and 0.1% per operated side, respectively.23
The danger to the skull base has several reasons:
1. In a prone patient, the skull base is turned completely toward the surgeon. Therefore, he or she is working perpendicularly to the skull base.
2. The basal lamella of the middle turbinate creates an angle with the roof of the ethmoid, an angle that is dull toward the front and peaked toward the back. The higher the basal lamella is perforated during the opening of the posterior ethmoid, the shorter the distance is to the roof of the ethmoid. The roof of the ethmoid can easily be mistaken for a bony septum of a posterior ethmoid cell.
3. During the attempt to remove a cell septum located close to the skull base with a dull instrument, a piece of the skull base could be removed and the thin dura torn.
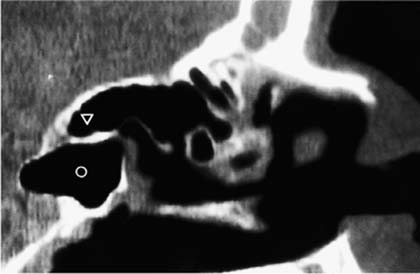
4. Depending on the pneumatization, the roof of the ethmoid might be situated higher than the roof of the sphenoid. In this case, the skull base could be injured during the attempt to open the sphenoid sinus transethmoidally (Fig. 18–3).
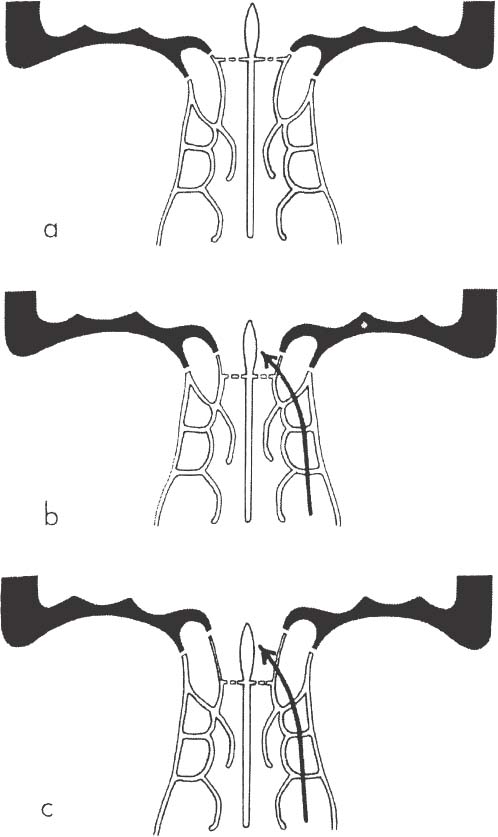
5. Another danger zone is the lateral lamella of the cribriform lamina plate in the case of a low rima olfactoria, which is nevertheless, according to Keros,61 4 to 16 mm in 89% of cases (Fig. 18–4). The lateral lamella of the cribriform plate is, at 0.05 mm, thinner than the neighboring roof of the ethmoid at 0.5 mm.62 The lateral lamella is injured if one is working too medial or if the middle turbinate is resected too close to the skull base or is fractured. According to Keros,61 the danger of an injury is extremely high in a Type III case, because the lateral lamella is very long (up to 16 mm). A low or high level of the lamina cribrosa can be identified on the preoperative coronal CT through the location of the anterior ethmoid arteries, because these and the fossa olfactoria have the same level. If the anterior ethmoid artery should be located in the ethmoid roof, the lamina cribrosa is also high (Type I, according to Keros). If the ethmoid artery runs in a bony mesenterium through the ethmoid, a low level of the cribriform plate must be expected (Type III, according to Keros). That means that the ethmoid roof is significantly higher than the cribriform plate and the lateral lamella and offers a much larger area of potential injury.
FIGURE 18–3 CT (sagittal reconstruction) of the paranasal sinuses. In this case, the roof of the ethmoid (triangle) stands higher than the roof of the sphenoid sinus (circle). Therefore, the sphenoid sinus should only be opened transnasally, never transethmoidally.
6. Another cause for potential harm to the dura can be gaps in the skull base. Ohnishi63 detected in human cadavers a significant percentage of bony gaps in the medial wall of the ethmoid sinus (lateral lamella of the lamina cribrosa), along the anterior and posterior ethmoid nerves, in the area of the filae olfactoriae, and at the medial wall of the middle turbinate, as well as in the anterolateral aspect of the ethmoid roof. Around these gaps, the nasal mucosa is fixed to the dura, so that the dura can be injured without perforation of the bony ethmoid roof.
FIGURE 18–4 The three different positions of the lamina cribrosa. (A) High level: no lateral lamella of the lamina cribrosa. (B,C) The lower the position of the lamina cribrosa, the longer the very thin lateral lamella and the higher the risk of injuring.
DIAGNOSTIC
Fresh injuries of the skull base during the course of an ethmoid sinus operation can usually be identified with the consequent application of optical aids (endoscope or microscope). Their prompt localization and treatment do not cause any problems. The diagnosis of old skull base defects is much more problematic. First of all, it must be clarified whether the liquid secretion is cerebrospinal fluid. Nowadays, this can be done with the determination of the β-2-transferrin in the nasal secretion.59,64,65 To locate the defect, a high-resolution CT scan (2 mm slices) of the frontal basis is performed in coronal levels. If it is not possible to detect the location of the injury with CT or a T2 weighted MRI, a fluorescein nasal endoscopy is performed, according to Messerklinger.12 Usually, 5% sodium fluorescein is employed, according to Stammberger.66 After lumbar puncture and injection of 0.25–0.50 mL of 5% sodium fluorescein solution specifically for intrathecal use, diluted with 10 mL of the patient’s CSF, the patient must lie face down, and the bottom end of the bed is lifted so that the patient’s head is lower than his or her feet. Because of the higher specific weight, the fluorescein sinks down the ventricle and runs through the dura leakage into the nose. After only a few minutes, the presence of an intensely yellow-green cerebrospinal fluid in the nose can be identified. The nose is examined intraoperatively with the 30-degree endoscope and under blue light. Slight marked CSF traces can be identified as bright white-green areas. The fluorescence can be detected in a dilution of 1:10 million. The marked cerebrospinal fluid shows the way to the lesion of the dura with a high degree of certainty (Fig. 18–5).
According to Stammberger et al,66 it is a safe method. The complications that occur are caused by
1. Applying too much of the fluorescein solution. According to body weight, only 0.5 to a maximum of 1 mL of the 5% fluorescein solution should be injected.
2. Wrong concentration. Only the application of a 5% sodium fluorescein solution produces the same osmolarity as the cerebrospinal fluid.
3. Fluorescein used for external application is not allowed. Nor should ampullas for IV application be used, due to preservatives in this material.
THERAPY
We are of the opinion that a lesion of the dura definitely needs a surgical closure, because the direct connection of the nasal mucosa contaminated with bacteria with the intracerebral space leads to the danger of lethal inflammatory complications such as meningitis and intracerebral abscess. The risk of experiencing such an infection is ~20 to 50% in untreated traumatic defects of the frontal base.67 The closure of the defect is usually performed through an endonasal approach. We usually take homologous tissue (fascia lata) or lyodura, if possible in two layers with the underlay and overlay technique. Transplants are fixed with fibrin glue. An additional covering with nasal or turbinate mucosa is not absolutely necessary. The transplant tissue is covered with Gelfoam, then the ethmoid is filled with oxidiced cellulose, which is supported in the case of a larger defect by cottonwool tamponade. The tamponade is removed in stages after the eighth day.
FIGURE 18–5 Lesion of the lateral lamella of the right lamina cribrosa in a patient who suffered from meningitis several weeks after endonasal surgery. (A) CT scan. The lesion is marked with a triangle. (B) Endoscopic view after lumbar puncture and intrathecal injection of 1.0 mL of 5% sodium fluorescein solution. The prolapse of the brain shows the greenish color of the fluorescein. The defect was closed transnasally with lyodura.
Bleeding
The number of cases of bleeding that need transfusions has been greatly reduced since the introduction of endonasal surgery.23,68 In 10 of 1172 cases in our patient contingency, blood transfusions were necessary. In all cases, it was late bleeding. Intraoperative bleeding that needed to be taken care of was usually observed in the area of the sphenopalatine artery and its branches, especially during the dissection in the area of the posterior ends of the middle turbinate (arteria nasalis posterior lateralis) and in the area of the frontal wall of the sphenoid sinus and floor (injuries of the posterior medial nasal arteries).
Intraoperative bleeding is controlled by the use of postoperative dressing of oxidized cellulose (Oxycel cotton) by creating small strips and inserting them into the sinus cavity. Use of saline spray and irrigation over the first postoperative week causes the material to become soft and can be suctioned from the sinus at the first postoperative visit.
Bleeding often stops spontaneously or can be coagulated with the bipolar forceps. The same is true for injuries of the anterior ethmoid artery. Late bleedings in the first days after the operation are usually easy to locate and can be stopped by coagulation or by packing. There are rare single bleedings from the lateral posterior parts of the nose that are resistant to therapy. In these cases, a coagulation of the sphenopalatine artery has proven useful.69 We like to use the surgical microscope for these, as well as the self-retaining nasal speculum,70 so that the surgeon can work bimanually, aspirating with one hand and coagulating with the other hand (Fig. 18–7).
Bleeding into the orbit was discussed in the section Retrobulbar Postseptal Hematoma.
Bleeding of the Internal Carotid Artery
Injuries of the internal carotid artery occur due to anomalies and aneurysms of the artery, and in the case of a missing bony covering of the artery in the sphenoid sinus, if the sphenoid sinus is not opened strictly transnasally and paraseptally. The internal carotid artery can be compressed with a tamponade of the sphenoid sinus or with a balloon occlusion of the internal carotid artery, but frequently, hemiplegia or death is the result.
Intracerebral Bleeding of the Anterior Ethmoid Artery and Branches of the Anterior Cerebral Artery
These bleedings are very rare but difficult to control. Neurosurgical decompression of the brain by applying a trephination in the skull can avoid a fatal ending. We had to give a testimonium in one such case in which an injury of the skull happened between the roof of the ethmoid and the sphenoid sinus. The patient died despite neurosurgical decompression of the brain.
Different Complications
MYOSPHERULOSIS
A myospherulosis with formation of granulomas in the orbit or the eyelids develops through a foreign body reaction in the orbit, probably caused by ointment tamponades or nasal emulsions containing paraffin.34–36,71,72 These unpleasant, difficult-to-treat alterations in the area of the eyelids are probably explainable by the transportation of ointment into the orbit. Also, recurrences of chronic sinusitits are sometimes caused by foreign body reactions. After operating on recurrences, we were able to detect light-breaking foreign bodies in our pathologic specimens. Therefore, we do not use any kind of ointment, either in the form of tamponades or for postoperative care.
CONTROVERSY
This author has used mupirocin (Bactroban) ointment as a water-soluble antibiotic preparation to cover dressings placed in the sinus cavity. No evidence of foreign body reaction has been seen.