CLASSIFICATION
There have been many classifications proposed for uveitis. The most recent and widely employed is the one set forth by the Standardization of Uveitis Nomenclature (SUN) Working Group, who classified uveitic terminology in regard to clinical presentation, inflammatory grading, clinical complications, and clinical outcomes.
2 Standardization of terminology not only helps clinicians discuss individual cases, but allows for establishment of better diagnostic criteria for uveitic entities, as well as comparison of clinical research from around the world, enabling meta-analyses for rare conditions. Uveitis should be classified according to the anatomic location of the inflammation (anterior, intermediate, posterior, or panuveitis), disease onset (sudden or insidious), duration (limited or persistent), clinical course (acute, recurrent, or chronic), and clinical activity (worsening, improving, or in remission).
Anatomically, uveitis is divided into anterior, intermediate, posterior, and panuveitis, as set forth previously by the International Uveitis Study Group and endorsed by the SUN working group (
Table 32.1).
2,3 The classification of the anatomic location of the uveitis should be based on the site(s) of inflammation and not on the presence of structural complications. For example, a patient with dense vitreitis would be classified as having intermediate uveitis, even in the presence of vascular sheathing and/or macular edema. The term panuveitis should be reserved for situations where inflammation is present in the anterior chamber, vitreous, and retina or choroid, without a predominating site.
Presentation of uveitis should be described as sudden, typically presenting with acute symptoms of pain, redness or photophobia, or as insidious, usually presenting over an indeterminate period with symptoms such as cloudy vision or floaters (
Table 32.2). Duration of disease is considered limited if it resolves in less than 3 months versus persistent if it lasts beyond 3 months. Together these terms can characterize the clinical course, such as acute uveitis, which is of sudden onset with limited duration. This classification helps to narrow the differential diagnoses and diagnostic work-up. Recurrent disease is characterized by repeated episodes, typically sudden in onset, with quiescence off all therapy for at least 3 months between exacerbations. Chronic uveitis is persistent disease with relapse within 3 months if therapy is stopped. Describing the clinical course of disease not only focuses the clinician’s differential, but also has important treatment implications.
The SUN Working Group has also clarified the use of various ambiguous terms. The term pars planitis should be used only for that subset of intermediate uveitis where there is formation of snowbanks or snowballs, occurring in the absence of an associated systemic disease, that is, it is idiopathic. The use of the term retinal vasculitis should be limited to those cases in which there is evidence of ocular inflammation and retinal vascular changes, such as vascular sheathing or leakage. The presence of occlusive retinal vasculopathy, for example, in the absence of visible inflammation (e.g., antiphospholipid antibody syndrome) should not be considered retinal vasculitis.
When attempting to categorize uveitis, it is also important to note whether the inflammation is unilateral or bilateral. Certain conditions, such as HLA-B27-associated iridocyclitis, tend to be recurrent, involving one eye at a time, often alternating between eyes. Other conditions, such as toxocariasis and Fuchs iridocyclitis, tend to remain unilateral. Still other entities, such as Vogt-Koyanagi-Harada (VKH) syndrome or tubulointerstitial nephritis and uveitis (TINU) syndrome, tend to present with simultaneous or nearly simultaneous bilateral involvement.
Causation, if known, is used to modify the previously described classification. Patients such as those with sarcoidosis, Behçet disease, or HLA-B27-associated disease have this modifier added to their diagnosis. For example, using this schema, we may describe a patient as having bilateral chronic iridocyclitis associated with sarcoidosis or recurrent acute iridocyclitis associated with HLA-B27-positive ankylosing spondylitis.
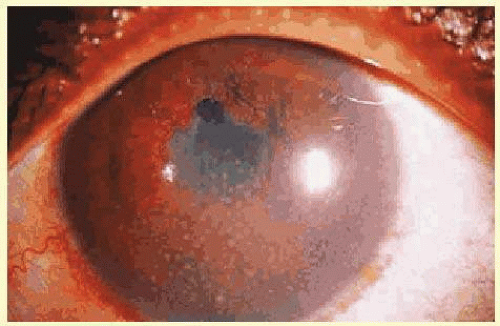
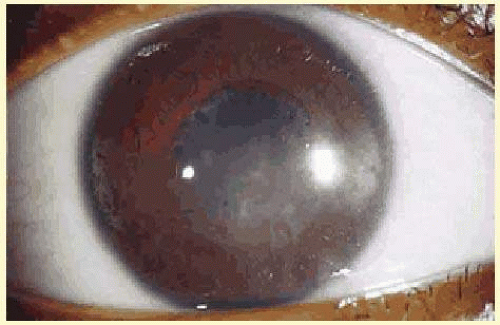
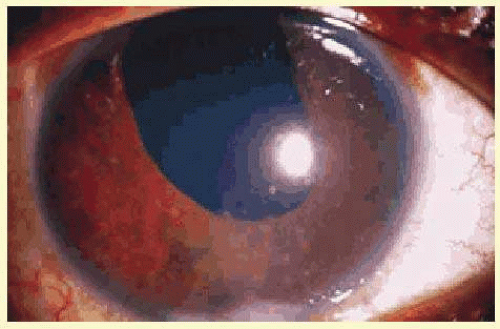
Another classification divides uveitis into granulomatous or nongranulomatous disease. This is a descriptive classification based on clinical examination, not histology, but it may help narrow the differential diagnosis. Uveitis is considered granulomatous if there are Busacca or Berlin nodules in the iris stroma, large greasy “mutton-fat” keratic precipitates, or choroidal granulomas. The presence of small Koeppe nodules at the pupillary margin does not imply that the disease process is granulomatous. Common causes of granulomatous inflammation include sarcoidosis, VKH, sympathetic ophthalmia, tuberculosis, and syphilis, although all of these entities may also present with nongranulomatous disease.