(1)
Department of Ophthalmology and Visual Sciences, University of Iowa Hospitals and Clinics, Iowa City, IA, USA
The cilioretinal artery was first described by Müller [1] in 1856. Its existence is a congenital anomaly. It belongs to the posterior ciliary artery system, so it usually arises from the peripapillary choroid or directly from one of the short posterior ciliary arteries; this origin has important clinical implications, as discussed below. Various anatomical aspects of cilioretinal arteries are discussed in detail in Chap. 3. Following is a brief summary.
The cilioretinal artery is generally small, but may be as large as one of the main divisions of the central retinal artery. It is usually situated on the temporal side of the disc, but not always. It has a characteristic sharp, diagnostic hooklike appearance at its site of entry into the retina at the optic disc margin. The incidence of cilioretinal arteries in an eye had been described variably by different studies on ophthalmic evaluation (see Chap. 3); it seems that ophthalmoscopic examination is not a true guide to the presence, absence, or number of cilioretinal arteries but may be quite misleading. The most reliable way to ascertain their true incidence in vivo is by fluorescein fundus angiography. This is because the cilioretinal artery fills synchronously with the filling of the choroid, since it belongs to the posterior ciliary artery system. The choroid usually starts to fill shortly before the start of filling of the central retinal artery. There are two large studies, using fluorescein fundus angiography, providing the incidence of the cilioretinal artery. As discussed in Chap. 3, Justice and Lehmann [2], using stereoscopic color fundus photographs and fluorescein angiograms, found one or more cilioretinal arteries in 49.5 % of all patients or in 32 % of the eyes. The cilioretinal arteries were present bilaterally in 14.6 %. Great variation in the size, number, and distribution of cilioretinal arteries was observed. Wang [3], in a fluorescein fundus angiographic study of 2,050 eyes, found a cilioretinal artery in 40.2 %. He found that in 129 eyes, there appeared to be a cilioretinal artery on ophthalmoscopy, which was not confirmed by fluorescein angiography. The size and location of the area of retina supplied by a cilioretinal artery vary widely from a minute artery supplying a tiny area of the peripapillary retina to about half and rarely even the entire retina [4–8]. Usually there is one cilioretinal artery, but there may be more than one in some eyes. In my anatomical study in one rhesus monkey, there were eight cilioretinal arteries in the right eye and three in the left [9]. (It may be worth mentioning here that the basic vascular pattern of the eye and optic nerve in the rhesus monkey is closely similar to that in man [10], so that the findings in one are applicable to the other for all practical purposes.)
In some eyes a branch of the central retinal artery, arising in the optic nerve, may travel all the way to the optic disc and emerge as a separate artery distinct from the rest of the central retinal artery. This artery could mistakenly be identified as a cilioretinal artery. In one specimen in my anatomical study [11] of 100 central retinal arteries, a branch arose from the middle of the horizontal section of the intraneural part of the artery and ran forward in the center of the nerve to enter the eyeball separately from the central retinal artery at the optic disc, to supply the superior part of the retina (Fig. 16.1).
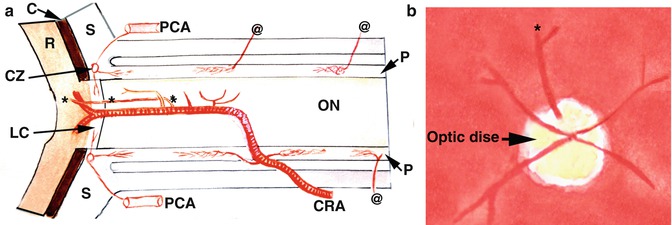

Fig. 16.1
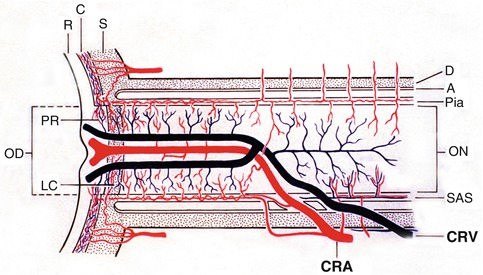
Diagrammatic representation of an intraneural branch of the central retinal artery emerging at the optic disc to supply the superior part of the retina (indicated by asterisk) (Reproduced from Hayreh [11]). (a) Diagram showing entire course of the central retinal artery and its branches from various parts. Intraneural branch indicated by asterisk emerges at the optic disc to supply the superior part, as shown in (b). C choroid, CRA central retinal artery, CZ circle of Haller and Zinn, LC lamina cribrosa, ON optic nerve, P pia mater, PCA posterior ciliary artery, R retina, S sclera, Asterisk intraneural branch emerging at the optic disc supplies the superior retina, @ pial arterial branches arising from the ophthalmic artery and its various branches
Cilioretinal Artery Occlusion
Cilioretinal artery occlusion is a distinct clinical entity, because the cilioretinal arteries arise from the posterior ciliary artery circulation, instead of the central retinal artery. Cilioretinal artery occlusion is not as common as branch retinal arteriolar occlusion or central retinal artery occlusion.
Classification of Cilioretinal Artery Occlusion
Based on my studies [12, 13], from the point of view of etiology, clinical features, and visual outcome, cilioretinal artery occlusion can be classified into the following three types: (1) non-arteritic cilioretinal artery occlusion alone, (2) arteritic cilioretinal artery occlusion associated with giant cell arteritis, and (3) non-arteritic cilioretinal artery occlusion associated with central retinal vein occlusion or hemi-central retinal vein occlusion. Of the three types of cilioretinal artery occlusion, those associated with giant cell arteritis and those with central retinal vein occlusion or hemi-central retinal vein occlusion require detailed discussion about their pathogeneses, which are controversial.
Non-arteritic Cilioretinal Artery Occlusion Alone
In my study [12] of 212 eyes with branch retinal arteriolar occlusion, there were 11 eyes with non-arteritic cilioretinal artery occlusion. It was located superiorly in 5, inferiorly in 2, and in the macular region in 4. It was almost always due to embolism.
Arteritic Cilioretinal Artery Occlusion Associated with Giant Cell Arteritis
In giant cell arteritis, arteritic cilioretinal artery occlusion is almost always associated with arteritic anterior ischemic optic neuropathy. This clinical entity was first described in 1974 [14]. I have described this condition in my various publications on giant cell arteritis [14–20], and there are a few more anecdotal reports in the literature. In my study [12] of 212 eyes with branch retinal arteriolar occlusion, there were 12 eyes with arteritic cilioretinal artery occlusion – in 11 of them it was associated with arteritic anterior ischemic optic neuropathy. In the one where cilioretinal artery occlusion was not associated with arteritic anterior ischemic optic neuropathy, the occluded posterior ciliary artery did not supply the optic nerve head.
Pathogenesis of Arteritic Cilioretinal Artery Occlusion Associated with Giant Cell Arteritis
This is an extremely important type of cilioretinal artery occlusion. As Kearns [21] rightly stressed, giant cell arteritis “ranks as the prime medical emergency in ophthalmology, there being no other disease in which the prevention of blindness depends so much on prompt recognition and early treatment.” It can result in massive bilateral visual loss if not detected and treated urgently – the sooner high-dose steroid therapy is instituted, the smaller is the risk of any further visual loss [20, 22].
The posterior ciliary artery supplies the optic nerve head [23–25] as well as the cilioretinal artery (Fig. 16.2). Giant cell arteritis has a selective tendency to involve the posterior ciliary artery [14, 17, 20], resulting in its occlusion, which in turn results in simultaneous development of both arteritic anterior ischemic optic neuropathy and cilioretinal artery occlusion (Figs. 16.3, 16.4, 16.5, and 16.6) [14, 15, 17, 20]. The arteritic anterior ischemic optic neuropathy causes massive visual loss in these eyes.
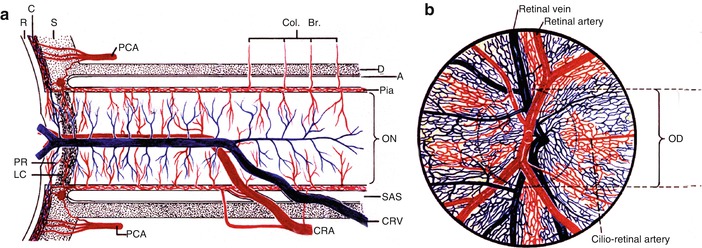
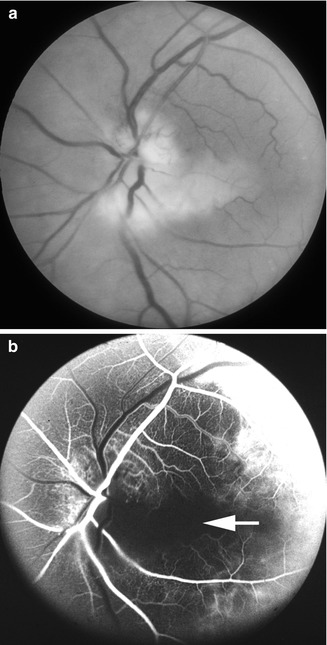
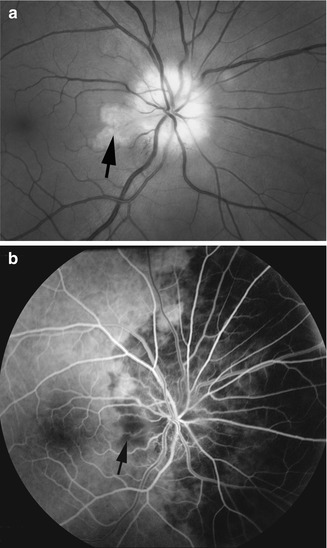
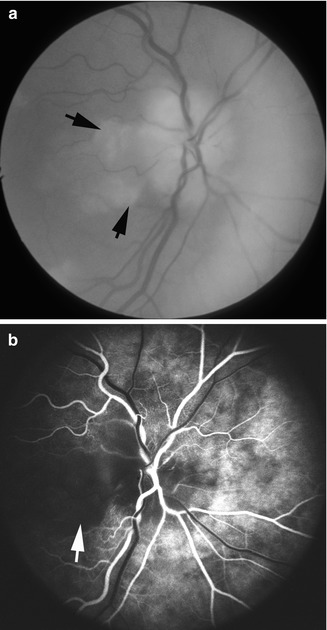
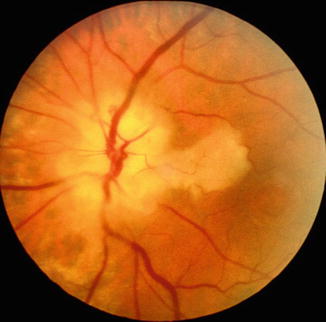

Fig. 16.2
Schematic representation, showing: (a) the course of central retinal artery and its branches and anastomoses, central retinal vein and its tributaries, and blood supply of the optic nerve. (b) Blood vessels on the optic disc and in the retina (Modified from Hayreh [26]). Abbreviations: A arachnoid, C choroid, CRA central retinal artery, Col. Br collateral branches, CRV central retinal vein, D dura, LC lamina cribrosa, OD optic disc, ON optic nerve, PCA posterior ciliary artery, PR prelaminar region, R retina, S sclera, SAS subarachnoid space

Fig. 16.3
Fundus photograph (a) and fluorescein angiogram (b) of the left eye with arteritic anterior ischemic optic neuropathy and associated cilioretinal artery occlusion (arrow), in a patient with giant cell arteritis. Fluorescein angiogram (b) shows normal filling of the area supplied by the lateral posterior ciliary artery, but no filling of the choroid and entire optic disc supplied by the medial posterior ciliary artery or of the retinal vessels in the area supplied by the occluded cilioretinal artery (arrow) (Reproduced from Hayreh [27])

Fig. 16.4
Fundus photograph (a) and fluorescein fundus angiogram (b) of the right eye with arteritic anterior ischemic optic neuropathy and cilioretinal artery occlusion. (a) Fundus photograph shows chalky white optic disc edema, with retinal infarct in the distribution of occluded cilioretinal artery (arrow) and small inferior temporal peripapillary hemorrhage. (b) Fluorescein fundus angiogram shows evidence of occlusion of the medial posterior ciliary artery and no filling of the retinal vessels in the region of the cilioretinal artery (arrow) (Reproduced from Hayreh [28])

Fig. 16.5
Fundus photograph (a) and fluorescein fundus angiogram (b) of the right eye with arteritic anterior ischemic optic neuropathy and cilioretinal artery occlusion. (a) Fundus photograph shows chalky white optic disc edema with retinal infarct in the distribution of occluded cilioretinal artery (arrows). (b) Fluorescein fundus angiogram shows evidence of occlusion of the lateral posterior ciliary artery and no retinal vascular filling in the region of the cilioretinal artery (arrow)

Fig. 16.6
Fundus photograph of the left eye with arteritic anterior ischemic optic neuropathy and cilioretinal artery occlusion. It shows chalky white optic disc edema, with retinal infarct in the distribution of occluded cilioretinal artery
Cilioretinal artery occlusion in giant cell arteritis is sometimes erroneously diagnosed as “branch retinal artery occlusion” [29], but the so-called branch retinal arteries are in fact arterioles, and giant cell arteritis is a disease of the medium-sized and large arteries and not of the arterioles [17, 20]. I have seen patients with cilioretinal artery occlusion diagnosed by ophthalmologists as ordinary branch retinal arteriolar occlusion and left untreated, resulting in catastrophic and unnecessary visual loss in both eyes. These eyes present with the following classical, diagnostic clinical picture. A combination of chalky white optic disc edema, retinal infarct in the region of the occluded cilioretinal artery, and posterior ciliary artery occlusion on fluorescein angiography is diagnostic of giant cell arteritis as the cause (Figs. 16.3, 16.4, and 16.5) [14, 15, 17, 20]. In all patients ≥50 years who have cilioretinal artery occlusion, it is essential to rule out giant cell arteritis and associated posterior ciliary artery occlusion by fluorescein angiography.
Cilioretinal Artery Occlusion Associated with Central Retinal Vein Occlusion or Hemi-central Retinal Vein Occlusion
This clinical entity was first described by Oosterhuis [30] in 1968 and later by me [31] and McLeod and Ring [32]. Since then, a large number of reports have been published, mostly anecdotal in nature or based on small series, except for one recent report based on 38 eyes [33]. In this condition, there is cilioretinal artery occlusion secondary to central retinal vein occlusion or hemi-central retinal vein occlusion.
I investigated this in 38 eyes in which cilioretinal artery occlusion was associated with central retinal vein occlusion or hemi-central retinal vein occlusion [33]. A full account of the results of this group of cilioretinal artery occlusion eyes is given elsewhere [33]. In summary, of the 38 eyes with cilioretinal artery occlusion in this study, 30 had nonischemic central retinal vein occlusion, 5 had ischemic central retinal vein occlusion, and 3 had nonischemic hemi-central retinal vein occlusion. Initially the ophthalmoscopic and fluorescein angiographic findings were similar to those seen in central retinal vein occlusion and hemi-central retinal vein occlusion, except that all these eyes had retinal infarct in the distribution of the cilioretinal artery. The size and site of the occluded cilioretinal artery varied widely, as shown in Figs. 16.7, 16.8, 16.9, 16.10, 16.11, 16.12, 16.13, 16.14, and 16.15. The occluded cilioretinal arteries are usually temporal in location (Figs. 16.6, 16.7, 16.8, 16.9, 16.10, 16.11, and 16.12) and sometimes nasal (Figs. 16.14 and 16.15). In Fig. 16.14 there were three cilioretinal arteries, and all of them were occluded.
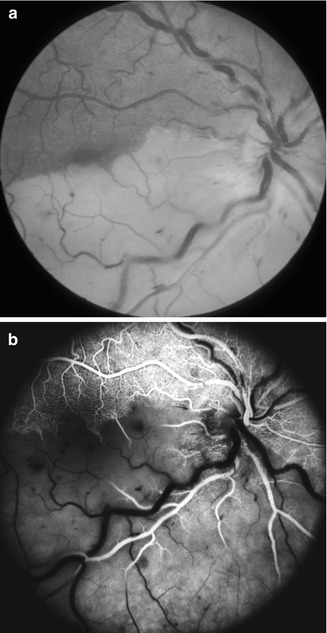
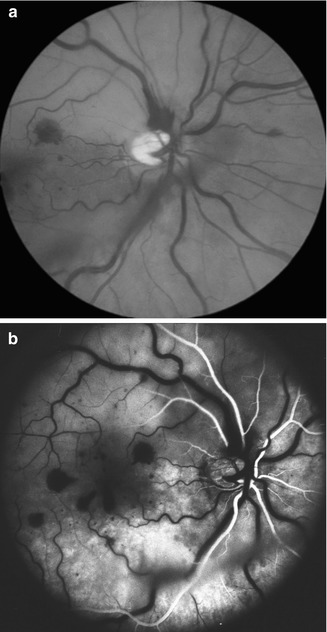
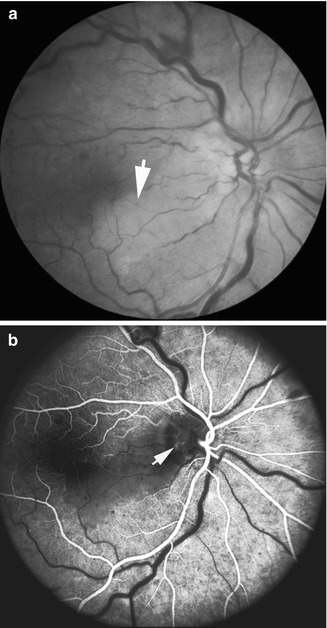
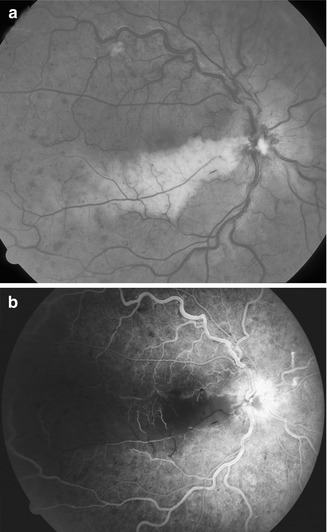
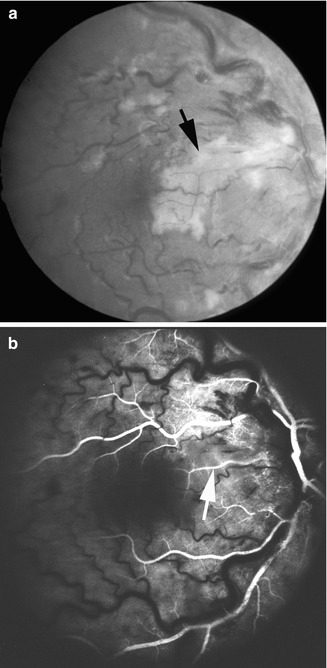
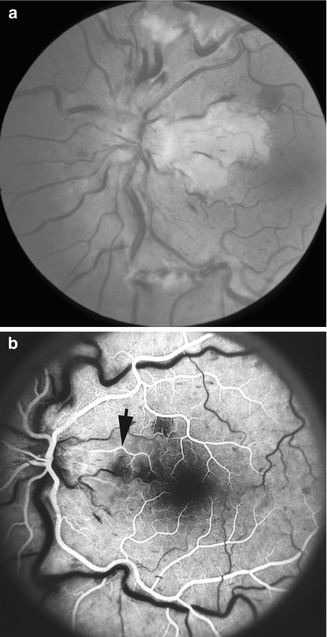
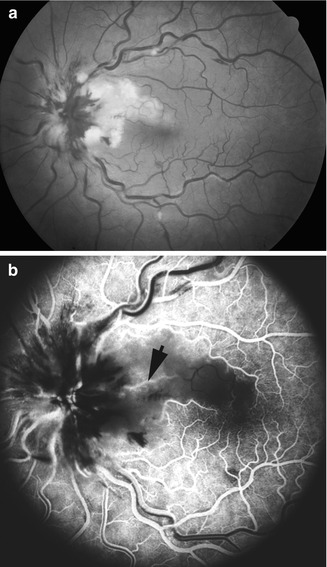
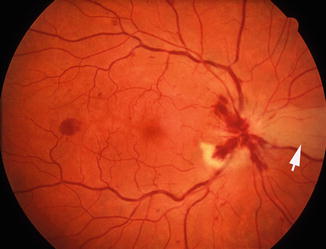
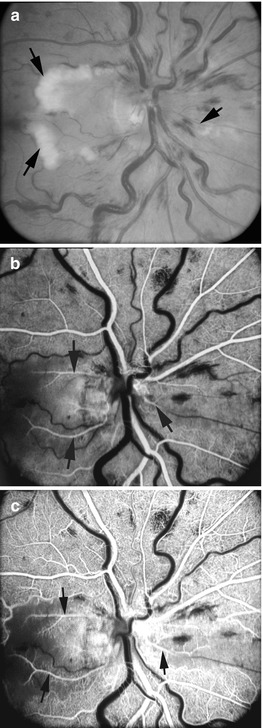

Fig. 16.7
I saw this 20-year-old woman, 1 day after development of cilioretinal artery occlusion with nonischemic CRVO. At initial visit, the visual acuity was 20/200 with superior visual field defect corresponding to the distribution of the occluded cilioretinal artery. (a) Fundus shows engorged retinal veins, a few retinal hemorrhages, and a large area of inferior retinal infarct in the region of the occluded cilioretinal artery. (b) Fluorescein fundus angiogram shows complete filling of the retinal circulation supplied by the central retinal artery, but it took 17 s for cilioretinal artery to fill completely. Final visual acuity was 20/20 (a, Reproduced from Hayreh et al. [33])

Fig. 16.8
I saw this 37-year-old man, 2 days after he developed cilioretinal artery occlusion with nonischemic CRVO in his right eye. His visual acuity was 20/400 with absolute inferior nasal sector visual loss. (a) Fundus photograph shows engorged retinal veins, a few retinal hemorrhages, and retinal infarct involving the superior temporal retina. (b) Fluorescein angiogram shows slow filling of the cilioretinal artery and it took 16 s to fill completely. Visual field finally showed permanent inferior nasal sector loss, with visual acuity 20/20

Fig. 16.9
A 40-year-old woman developed cilioretinal artery occlusion with nonischemic CRVO. I saw her 6 days later, when the visual acuity was counting fingers, with a centrocecal scotoma. (a) Fundus shows engorged retinal veins, a few retinal hemorrhages, and retinal infarct in the distribution of the occluded cilioretinal artery (arrow). (b) Fluorescein fundus angiogram during retinal arterial phase shows filling of the cilioretinal artery at the optic disc (arrow), but no filling of the retinal capillary bed in the area of cilioretinal artery occlusion. Three months after onset, visual acuity was 20/25

Fig. 16.10
I saw this 32-year-old man with cilioretinal artery occlusion with nonischemic CRVO, counting fingers visual acuity, and visual field defect corresponding to occluded artery. (a) Fundus photograph shows engorged retinal veins, few retinal hemorrhages, optic disc and macular edema, and area of retinal infarct below the fovea and optic disc edema. (b) Fluorescein fundus angiogram during retinal venous phase shows no filling of the cilioretinal artery. Final vision in this eye after 3 months was 20/40 (a, Reproduced from Hayreh et al. [33])

Fig. 16.11
I saw this 19-year-old woman 12 days after development of cilioretinal artery occlusion with nonischemic CRVO onset. Visual acuity was 20/200 and there was centrocecal visual field defect. (a) Fundus shows engorged retinal veins, optic disc and macular edema, few retinal hemorrhages, and retinal infarct in the region supplied by the occluded cilioretinal artery (arrow). (b) Fluorescein fundus angiogram during retinal arterial phase shows slow filling of the cilioretinal artery (arrow). Visual acuity improved to 20/50 in 2 months, 20/30 3 months after that, and finally 20/20

Fig. 16.12
This 40-year-old woman was seen one day after the development of cilioretinal artery occlusion with nonischemic CRVO. Initially visual acuity was 20/30, with centrocecal visual field defect. (a) Fundus shows engorged retinal veins, optic disc and macular edema, few retinal hemorrhages and cotton-wool spots, and retinal infarct in the region supplied by the occluded cilioretinal artery. (b) Fluorescein fundus angiogram during retinal arterial phase shows early filling of the cilioretinal artery (arrow) and no filling of the capillary bed in the area of cilioretinal artery occlusion. Visual acuity had improved to 20/20 when seen 3 months later

Fig. 16.13
This 33-year-old woman 11 days earlier had developed cilioretinal artery occlusion with nonischemic CRVO with visual acuity of 20/80 and centrocecal scotoma. (a) Fundus shows engorged retinal veins with some perivenous exudates, hemorrhages mainly on the optic disc, a small area of retinal infarct temporal to the optic disc, and two cotton-wool spots. (b) Fluorescein fundus angiogram during retinal arterial phase shows slow filling of the cilioretinal artery (arrow) and no filling of the capillary bed in the area of cilioretinal artery occlusion

Fig. 16.14
A 42-year-old man developed nasal cilioretinal artery occlusion with nonischemic CRVO 1 week before I saw him. The visual acuity was 20/15 with a large temporal scotoma continuous with blind spot and a superior arcuate defect. Fundus photograph shows occlusion of a nasal cilioretinal artery (arrow) with engorged retinal veins and hemorrhages mainly on the optic disc

Fig. 16.15
I saw this 52-year-old woman 1 week after she suddenly noticed a purple colored patch in the eye. Her visual acuity was 20/20. She had developed cilioretinal artery occlusion with nonischemic CRVO. (a) Fundus photograph shows markedly engorged retinal veins, an area of retinal infarct supplied by 2 cilioretinal arteries temporal to the optic disc with white axoplasmic flow accumulation at the margin of the infarct (arrows), and also a third cilioretinal artery occlusion nasally (arrow). (b, c) Fluorescein fundus angiogram shows delayed filling of 3 cilioretinal arteries (arrows). There was a centrocecal scotoma extending 5° from the fixation spot (Dr. D. McLeod in his article in Br J Ophthalmol. 2005;89;229–37. Wrongly cited this photograph as belonging to Dr. J.F. Cullen, when in fact it was mine)
Fluorescein angiography typically showed only a transient hemodynamic block in the cilioretinal artery [33] and not the typical organic occlusion seen in other types of cilioretinal artery occlusion. The fluorescein angiograms in Figs. 16.7, 16.13, 16.15, and 16.16 show a variable degree of filling of the cilioretinal arteries.
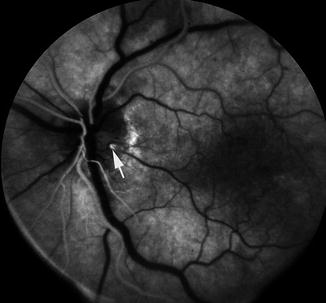

Fig. 16.16
Fluorescein fundus angiogram, during retinal arterial phase, of the left eye with nonischemic central retinal vein occlusion and cilioretinal artery occlusion. It shows filling of the cilioretinal artery where it just emerges at the optic disc (arrow)
Pathogenesis of Cilioretinal Artery Occlusion Associated with Central Retinal Vein Occlusion/Hemi-central Retinal Vein Occlusion
Several hypotheses have been put forward to explain the simultaneous development of cilioretinal artery occlusion and central retinal vein occlusion; that has resulted in a controversy on the subject, which requires detailed discussion. McLeod and Ring [32], based on a study of 11 eyes, initially postulated that in these patients, “partial obstruction of their posterior ciliary arteries may be the explanation for at least some of our cases.” They went on: “This series may represent a spectrum of ocular vascular lesions intermediate between acute central retinal vein occlusion and acute ischaemic optic neuropathy.” McLeod [34], based on a series of presumptions and speculations, has recently postulated the following somewhat confusing hypothesis to explain the development of cilioretinal artery occlusion with central retinal vein occlusion. He concluded: “In eyes with a cilioretinal supply, the probability that cilioretinal infarction will complicate retinal vein occlusion increases with increasing severity of venous obstruction and the more distally the cilioretinal artery arises from the posterior ciliary arterial tree. A distal branch point also facilitates observation of dye front reciprocation within the artery. Indicators of the degree of venous obstruction that may be necessary to instigate cilioretinal infarction include very prolonged dye transit times in the central retinal circulation, exaggerated venous cyanosis and tortuosity, perivenous cotton-wool sentinels, and macular perivenular whitening.” He goes on to state: “In eyes with central retinal vein occlusion and a cilioretinal supply, the degree of venous outflow obstruction that will be sufficient to trigger branch flow exclusion and diversion depends primarily on the site of cilioretinal branching from the posterior ciliary artery circulation. Cilioretinal territories supplied by arteries that arise from the proximal part of the posterior ciliary artery tree are unlikely to be selectively hypoperfused after central retinal vein occlusion, especially if they are hemodynamically isolated from the choroid. Finally, in theory, choroidal arterial steal might also operate by diverting blood from the central retinal artery after central retinal vein occlusion in some of the 60 % of individuals in whom the central retinal artery arises from the ophthalmic artery in company with one or more posterior ciliary arteries.”
I have myself performed fluorescein angiography in many such cases, and looking into the fundus camera while the dye is injected, I observed the actual dynamics of blood flow in the retinal vasculature and cilioretinal arteries. This provided information that is not always available from the pictures taken during angiography. I discuss below the fluorescein angiographic findings in such eyes, based on my observations. From these dynamic angiographic studies and my extensive basic, experimental, and clinical studies on the posterior ciliary artery circulation, central retinal vein occlusion, and central retinal artery occlusion, I find the hypothesis postulated by McLeod inadequate to explain the pathogenesis of development of cilioretinal artery occlusion with central retinal vein occlusion and hemi-central retinal vein occlusion. There is no hemodynamic logic in the speculation by McLeod that “choroidal arterial steal might also operate by diverting blood from the central retinal artery after central retinal vein occlusion in some of the 60 % of individuals in whom the central retinal artery arises from the ophthalmic artery in company with one or more posterior ciliary arteries.”
Glacet-Bernard et al. [35] stated that the possibility of primary occlusion of the cilioretinal artery must be considered in these eyes. It has also been postulated that optic disc edema caused by central retinal vein occlusion can cause cilioretinal artery occlusion, but this hypothesis is not valid because optic disc edema cannot cause cilioretinal artery occlusion. Some have even attributed cilioretinal artery occlusion with central retinal vein occlusion to embolism. Schatz et al. [36], based on a study of ten cases, discussed the various possible mechanisms.
To comprehend the pathogenesis of this clinical entity logically, one has to understand the factors that influence the ocular blood flow. Blood flow in general depends upon the intraluminal perfusion pressure (perfusion pressure = arterial pressure − venous pressure). Therefore, any of the factors that either reduce the arterial pressure or increase the venous pressure, or a combination of both, results in reduced perfusion pressure and consequently decreased blood flow or even no circulation.
Eyes with an additional cilioretinal artery differ from eyes with the central retinal artery as the only source of blood supply in their retinal arterial and venous drainage systems. The venous drainage from the entire retina is by the central retinal vein, irrespective of the numbers and sources of the arteries that supply the retina. In eyes with a cilioretinal artery, the arterial supply to the retina is obviously from two independent sources – the central retinal artery is the major source, and the cilioretinal artery usually supplies only a small part of the retina, though its distribution can vary widely (Figs. 16.7, 16.8, 16.9, 16.10, 16.11, 16.12, 16.13, 16.14, and 16.15) [9]. The central retinal artery and cilioretinal artery belong to two different types of arterial systems with different physiological properties. The central retinal artery has efficient blood flow autoregulation, so that when there is a fall in perfusion pressure in the retinal arterial bed caused by a rise in the retinal venous pressure, the autoregulatory mechanism in the retinal arterial bed causes a rise in its pressure, to try to maintain retinal circulation. By contrast, since the cilioretinal artery usually belongs to the choroidal vascular system, the following two entirely different mechanisms are working in the cilioretinal artery circulation in eyes with central retinal vein occlusion: (a) the choroidal vascular bed has no or poor autoregulation, and (b) in eyes with central retinal vein occlusion, there is no vortex venous obstruction. Therefore, a sudden onset of central retinal vein occlusion increases the retinal capillary intraluminal pressure, and that in turn decreases the perfusion pressure, and that then triggers the autoregulatory mechanism in the central retinal artery to maintain its blood flow; by contrast, no such compensatory mechanism exists in the choroid or the short posterior arteries supplying the choroid. Moreover, studies have shown that the perfusion pressure in the choroidal vascular bed is normally lower than that in the central retinal artery [37–39]. Thus, the theory proposed by McLeod and Ring [32] that in these eyes there is partial obstruction of their posterior ciliary arteries and that this represents “a spectrum of ocular vascular lesions intermediate between acute central retinal vein occlusion and acute ischaemic optic neuropathy” is not valid; moreover, contrary to what they stated, there is no evidence of anterior ischemic optic neuropathy in these eyes.
In the light of the above facts, then, let us consider the hemodynamic situation in eyes that have a cilioretinal artery and develop central retinal vein occlusion. Sudden occlusion of the central retinal vein results in a marked rise of intraluminal pressure in the entire retinal capillary bed; when that intraluminal pressure rises above that in the cilioretinal artery, the result is a hemodynamic stasis in the cilioretinal artery. In many of these eyes, angiography performed during the early acute phase provides information about the in vivo dynamics of blood flow in the eye. During the early stages of the transit of the dye, the cilioretinal artery usually fills up to the optic disc (Figs. 16.9b, 16.10b, and 16.16), since the optic nerve head is supplied mainly by the posterior ciliary artery circulation (Fig. 16.2) [23–25]. During systole, the cilioretinal artery often fills for a variable length from the optic disc into the retina (Figs. 16.7b, 16.8b, 16.11b, 16.12b,16.13b, and 16.15b, c), but during diastole, the filling retracts to the optic disc, resulting in an oscillating blood column in the cilioretinal artery, moving back and forth from the optic disc for a variable distance into the retina; I have often seen this when observing the fundus while doing fluorescein fundus angiography in these eyes – it presents a fascinating phenomenon. Thus, the cilioretinal artery occlusion in these eyes is simply a hemodynamic stasis and is not due to embolism or thrombosis. The hemodynamic stasis is invariably transient, lasting from a few hours to several days, depending upon how severe the retinal venous stasis is and how rapidly the collateral circulation is established by the central retinal vein through its multiple tributaries in the optic nerve (Fig. 16.2) [40]. Therefore, in the optic nerve, the further anterior the site of occlusion in the central retinal vein, the fewer tributaries are available (Fig. 16.2), and the longer it takes to re-establish the circulation – and vice versa. As soon as those tributaries establish collateral circulation, there is a fall of intraluminal pressure in the retinal capillary bed to below that of the blood pressure in the cilioretinal artery, resulting in restoration of retinal circulation in the distribution of the cilioretinal artery. Hence, if a patient with central retinal vein occlusion is seen for the first time many days after the onset of cilioretinal artery occlusion, a retinal infarct is present ophthalmoscopically, but on angiography the cilioretinal artery fills normally; that can result in confusion and mistaken diagnosis of the cause of retinal infarct. This may be the reason why occlusion of the cilioretinal artery has mistakenly been attributed to embolism. In hemi-central retinal vein occlusion, when one of the two trunks of the central retinal vein is occluded [41] (Fig. 16.17), the above mechanism applies only to the segment of the retina drained by the occluded trunk.