Chronic Rhinosinusitis with Nasal Polyposis
Matthew W. Ryan
Nasal polyps (NPs) are among the oldest recorded ear nose and throat disorders, with historical references that date back to the ancient Egyptians of approximately 2000 bc. NP disease was later the subject of work by Hippocrates who gave these nasal masses their name “polypus.” Today we recognize NPs to be benign nasal growths that usually arise as a result of paranasal sinus inflammation, that is, chronic rhinosinusitis (CRS). For this reason, NP disease is considered a subtype of CRS and is labeled chronic rhinosinusitis with nasal polyposis (CRSNP) (1). A variety of other terms have been applied to inflammatory sinonasal conditions with bilateral or diffuse polypoid inflammation: chronic hyperplastic sinusitis, nasal polyposis, or simply NP disease. However, NPs may also arise as isolated lesions in otherwise healthy paranasal sinuses; these solitary polyps are pathophysiologically distinct, and are not considered under the umbrella of CRSNP. Despite the fact that NP disease has been a matter of scientific interest for thousands of years, we do not yet have a widely accepted classification system for NP disorders, the diverse pathophysiologic mechanisms for NP formation are unsettled, and the ideal treatment approach continues to evolve. This chapter reviews the pathophysiology, diagnosis, and classification of NP disease. Treatment for NP disease is covered in Chapters 39 and 40.
The pathophysiology of CRSNPs may differ significantly from other forms of chronic rhinosinusitis without polyps. Thus chronic rhinosinusitis is generally subclassified as “polypoid” or : “nonpolypoid” (based upon the presence of polyps). Nevertheless, the clinical and pathologic presentation of these two entities often overlaps, resulting in a significant spectrum of inflammatory disease in the paranasal sinuses that defies distinct classification (2). Histopathologically, most NPs are noted to have pronounced eosinophilic inflammation. For this reason, allergic mechanisms were assumed to play a causative role in polyp formation. In contrast, chronic rhinosinusitis without nasal polyposis is more commonly found to have a neutrophilic inflammatory infiltrate, and bacterial infection was assumed to play a causative role. We now know that neither of these explanations is consistent or accurate. For example, CRSNP may arise as a result of disease processes that lead to chronic infection (e.g., ciliary dyskinesia, cystic fibrosis [CF]) and the inflammatory infiltrate may be either neutrophilic or eosinophilic. There are some cases in which nasal polyposis evolves out of an underlying CRS without polyps. While the distinction of polypoid and nonpolypoid CRS may be useful clinically, many studies consider them together. So our current classification system should be regarded as tentative, awaiting further clarification of the pathophysiology and appropriate subclassification of chronic rhinosinusitis (Table 34.1).
EPIDEMIOLOGY OF NASAL POLYP DISEASE
The epidemiology of NP disease is difficult to characterize because the diagnosis incorporates multiple syndromes and possible etiologies for NP formation. Questionnaire and survey studies are likely to be inaccurate, and nasal endoscopy is usually required for definitive diagnosis. Despite these limitations, data from a few well-designed studies reveal the prevalence of NP disease to be 1% to 4% (3). Autopsy studies report a higher prevalence but it is not clear that these polyps are clinically significant. NP disease appears to be more common in males and the incidence peaks in the fourth to sixth decades of life (3).
Ethnic and geographic differences may impact the incidence of the various forms of NP disease. Allergic fungal rhinosinusitis (AFRS), which may represent 5% to 10% of CRSNP cases taken to surgery, appears to be more common in warm or humid climates, and is predominantly a disease of teens and young adults. Noneosinophilic NP disease appears to be more common in Asian populations (4).
Whether there is a difference in incidence of NP disease in Asian versus European peoples is unclear.
Whether there is a difference in incidence of NP disease in Asian versus European peoples is unclear.
TABLE 34.1 CLASSIFICATION AND FEATURES OF VARIOUS FORMS OF NP DISEASE | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Age is an important factor in the incidence of various forms of NP disease. Nasal polyposis in childhood is very rare with an estimated incidence of 0.1% to 0.2%. For this reason, the discovery of an NP in a child should prompt an evaluation to secure an accurate diagnosis and rule out causative conditions such as CF. Antrochoanal polyps account for approximately 5% of polyp cases and present more commonly in young adults (3). AFRS, an important subtype of CRSNP (discussed further in Chapter 37), is also predominantly a disease of teenagers and young adults. Patients with other (non-AFRS) eosinophilic mucin rhinosinusitis tend to be middle aged (average age 48) (5).
Genetic predisposition quite plausibly accounts for at least some cases of NP disease, given the heritability of other eosinophilic inflammatory diseases such as allergic rhinitis and asthma. A genetic basis is also clearly established for diseases that are known to cause NPs, such as CF and Kartagener syndrome. While this topic has not been extensively investigated, one study found that 25% of NP patients had a first degree relative with NP disease (6). However, the majority of NP disease is not clearly heritable via Mendelian genetics.
CLINICAL PRESENTATION
NPs cause symptoms that are typical of other forms of chronic rhinosinusitis. Most patients will have months or years of progressively worsening sinonasal symptoms before they are diagnosed. In some cases, patients without a history of significant sinonasal symptomatology will present with continuous symptoms that can be traced back to an initial inciting event—usually a severe upper respiratory infection or flulike illness. Nasal obstruction, hyposmia, and rhinorrhea are common symptomatic complaints, but nasal congestion, facial pressure or headache, and postnasal drainage are also common. Symptoms alone are unreliable for the diagnosis of polypoid CRS, but smell dysfunction seems to be one feature that is more commonly encountered in CRSNP. This smell dysfunction may be a result of obstruction of airflow into the olfactory cleft or direct inflammation of the mucosa of the olfactory cleft.
TABLE 34.2 DIFFERENTIAL DIAGNOSIS OF NP DISEASE | |||||
|---|---|---|---|---|---|
|
The definitive diagnosis of NP disease requires histopathologic examination. A wide variety of tumors and other lesions can mimic NP disease (Table 34.2). These polyp mimics tend to be solitary and unilateral. A suspected polyp that is isolated or unilateral should be further evaluated with computed tomography (CT) imaging followed by biopsy. In most cases of CRSNP the polyps are present bilaterally, owing to their inflammatory etiology, and a clinical diagnosis can be made with information from the history and examination.
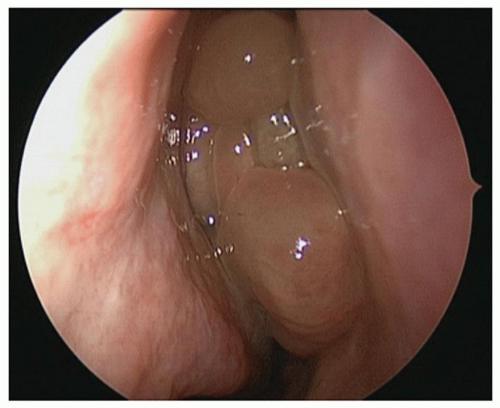
Large NPs may be visible on anterior rhinoscopy. Polyps appear as smooth rounded masses with a pale to yellow coloration, usually occurring in clusters (Fig. 34.1) The polyps may be almost translucent. However, there is a great variability in NP appearance with some having an erythematous or irregular mucosal covering. It must be noted that depending upon the degree and type of inflammation,
certain NPs may be indistinguishable from tumors. Likewise, some neoplastic lesions or encephaloceles may have an appearance very similar to an NP (Fig. 34.2). Thus in cases of suspected NP disease, imaging, biopsy, or close follow-up are indicated. Clinically significant NPs may not be visible on anterior rhinoscopy, however.
certain NPs may be indistinguishable from tumors. Likewise, some neoplastic lesions or encephaloceles may have an appearance very similar to an NP (Fig. 34.2). Thus in cases of suspected NP disease, imaging, biopsy, or close follow-up are indicated. Clinically significant NPs may not be visible on anterior rhinoscopy, however.
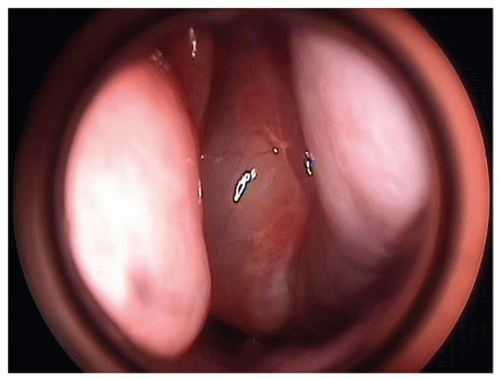 Figure 34.2 Endoscopic view of a smooth, rounded, pale, polypoid mass filling the left nasal cavity and extending almost to the nasal floor. Imaging revealed this mass to be a meningoencephalocele. |
In most cases, NPs are only revealed with thorough nasal endoscopy. The development of office endoscopy has greatly improved our ability to diagnose and monitor the activity of NP disease. With the magnification and illumination of endoscopy it is possible to identify smaller polyps. Polyps usually arise from the lateral nasal wall in the middle meatus or sphenoethmoidal recess. Those polyps that originate lateral to the middle turbinate usually take their origin from the frontal, anterior ethmoid, or maxillary sinuses. Polyps medial to the middle turbinate are usually arising from the posterior ethmoid or sphenoid sinuses. In rare cases, polyps may originate on the nasal septum or from the olfactory cleft. In general, however, a nasal mass that appears to arise or take its origin from the nasal mucosa is less likely to be a benign polyp. NP disease may also be seen in conjunction with abnormal mucus secretions or purulent exudates. Eosinophilic mucin, which is a feature of eosinophilic mucin rhinosinusitis and AFRS, may sometimes be seen on endoscopy, especially in operated patients with an exacerbation of their inflammatory disease. Grossly, eosinophilic mucin is dense, gelatinous to clay-like with color that ranges from yellow to fluorescent green to brown (Fig. 34.3). The finding of eosinophilic mucin may point to one of these diagnoses but is not otherwise diagnostically useful.
As a clinical tool, nasal endoscopy is extremely helpful to monitor the disease burden in patients with CRSNP. Subjective symptoms are a notoriously poor predictor of polyp size or inflammatory disease burden. The treatment approach for CRSNP is discussed in Chapters 39 and 40. In general, while a patient with CRSNP is undergoing medical treatment or being followed up after sinus surgery, regular endoscopic examinations are warranted to evaluate the degree of sinus inflammation and polyp burden. Symptoms alone may not provide adequate warning of significant polyp recurrence until nasal airway obstruction or hyposmia recur. Accurate assessment of disease activity is best performed with surveillance endoscopic evaluations. Medical therapy can then be adjusted based upon the degree of sinonasal inflammation as well as the patient’s symptom burden.
 Figure 34.3 Endoscopic view of dense golden eosinophilic mucin in the right middle meatus with generalized nasal edema. |
Imaging studies are an important component of the diagnostic workup of suspected NP disease. For sinonasal imaging, noncontrast CT is the study of choice. Imaging serves a variety of purposes in this setting. Nonpolyp lesions such as meningoencephalocele or nasal glioma can be distinguished from polyps based upon their location and associated skull base defects. Some tumors such as inverted papilloma and juvenile angiofibroma have characteristic imaging features. In CRSNP, CT enables a delineation of the involved sinuses, can reveal the presence of eosinophilic mucin, and can identify mucoceles, lamina disruption, or skull base erosion that may alter the treatment approach (Fig. 34.4). This imaging provides important anatomic information if surgery is performed. CRSNP causes a significant quality of life (QOL) burden, but is not usually a threat to life or bodily function. Imaging can identify those cases in which a conservative treatment approach is reasonable as well as cases in which more immediate surgical intervention is advisable (e.g., mucocele formation, intraorbital or intracranial expansion of disease). Magnetic resonance (MR) imaging is not usually necessary for the evaluation of NP disease. While MR can delineate polyp from retained secretions, establish a diagnosis of meningoencephalocele, and determine the extent of sinonasal neoplasms, the bony detail of the paranasal
sinuses is lacking. Therefore MR imaging is reserved for special circumstances.
sinuses is lacking. Therefore MR imaging is reserved for special circumstances.
SEVERITY CLASSIFICATION IN NASAL POLYP DISEASE
The severity of NP disease can be measured along several different axes. Like many other chronic medical conditions, the symptom burden of CRSNP is only loosely correlated with objective measures of disease severity. From a patient’s standpoint the symptom burden and QOL impairment are paramount. However, the treating physician must also use information from objective measures to formulate an appropriate treatment approach.
Symptomatic burden in CRSNP can be assessed with a variety of tools including nonvalidated symptom scales and validated disease-specific QOL instruments such as the sinonasal outcome test and rhinosinusitis outcome measure (7). These tools may be used during follow-up visits to assess patient symptom burden over time, and this information should be integrated with objective information to make appropriate adjustments in treatment.
Nasal endoscopy is the most useful objective tool for assessing the burden of inflammatory polyps in the nose and paranasal sinuses. Reliable estimation of polyp size is facilitated by the use of a grading system. One such method described by Lildholdt et al. (8) estimates the bulk of NPs in relation to fixed anatomical landmarks within the nose. A 0-3 grading system is used where 0, no polyps; 1, small polyps not reaching the upper edge of the inferior turbinate; 2, medium sized polyps reaching between the upper and lower edges of the inferior turbinate; and 3, large polyps reaching below the lower edge of the inferior turbinate. However, other similar grading systems have been devised with different scales (e.g., 1-4); therefore, it is important to describe the grading system used when reporting findings (8,9).
NATURAL HISTORY OF NASAL POLYP DISEASE
The clinical course of CRSNP is highly variable with some patients bothered only by nasal obstruction and others with a pattern of recurrent acute exacerbations that may represent episodes of obstructive bacterial sinusitis. In general, however, many patients with CRSNP will have functionally obstructed sinuses without associated bacterial sinusitis episodes. The natural history of most forms of NP disease is uncertain. Some NP diseases like antrochoanal polyp may be “cured” with a single surgery. Other individuals who develop NP disease in youth appear to “grow out of” their problem over a number of years. Yet others may develop the disease in middle age or later and appear to have an inexorable chronic disease that requires multiple surgeries and repeated courses of systemic steroids. Most CRSNP requires a disease management approach that incorporates current and future burdens of disease. The goals of treatment are to minimize the symptomatic burden over time, prevent rare complications, and minimize the impact of treatment—usually this means minimizing the number of surgeries required over time and minimizing the use of systemic steroid bursts.