Fig. 1
(a) An useful simple framework anticipating level of anesthesia and possible tools for safe airway management. (b) Potentially difficult or critical airway with Goldenhars Syndrome. (Courtesy of John McCloskey, The Children’s Hospital of Philadelphia, [CHOP]). (c) Conditions associated with difficult airway management in neonates
Neonates are also more prone to develop hypoxia during the process of securing the airway due to differences in lung volumes and metabolic rate as compared to children and adults. In neonates, the functional residual capacity is approximately 27 mL/kg versus 43 mL/kg in the adult. Oxygen consumption is approximately 6–8 mL/kg/min in neonates versus 3–4 mL/kg/min. Thus, when a neonate becomes apneic, hypoxia occurs much more rapidly due to the increased metabolism depleting oxygen from the relatively smaller reserve. Attention to the airway management strategy is a central component of the preanesthetic assessment of the neonate. Underlying diagnosis, coexisting diseases, and diagnoses should be reviewed with attention to airway management. If tracheal intubation has previously been performed, the involved providers can be questioned and the medical records reviewed to determine if there were unexpected difficulties. Conditions such as omphalocele and congenital diaphragmatic hernia may indirectly impact airway management causing rapid decompensation during anesthesia. Oxyhemoglobin desaturation follows apnea secondary to pulmonary hypoplasia and reduced functional residual capacity. Special attention to peak inspiratory pressures during anesthetic induction and intubation in particular lung malformations can prevent competing intestinal insufflation prior to intubation while also avoiding pneumothorax following intubation.
Rapid Sequence Intubation in Neonates
The goal of rapid sequence induction is to decrease the risk of pulmonary aspiration of gastric contents. Rapid sequence induction of anesthesia is a technique to minimize the time from anesthetic induction to tracheal intubation and can be challenging in infants. Due to the higher oxygen consumption rates, reduced FRC, and elevated closing volumes compared to older children and adult’s desaturations occur much more rapidly following apnea. Consequently, when a classic rapid sequence induction technique is performed and 45–60 s of apnea are allowed to occur before tracheal intubation is attempted, significant Oxyhemoglobin desaturation commonly occurs. Therefore many pediatric anesthesiologists perform a “modified” rapid sequence induction in this population [53–56]. This technique uses facemask ventilation performed under low pressures (<10–15 cm H2O) while cricoid pressure is applied until enough time has elapsed for complete neuromuscular blockade to occur. In support of this practice, appropriately applied cricoid pressure has been shown to be effective in preventing gastric inflation during gentle bag-mask ventilation in anesthetized infants and children [54–56]. If the initial attempt at intubation fails, gentle facemask ventilation through cricoid pressure should be performed. If ventilation is difficult with cricoid pressure despite the use of noninvasive devices (oral or nasopharyngeal airway, laryngeal mask airway) cricoid pressure should be lessened [54–56].
Abnormal Neonatal Anatomy and Physiology
There are numerous genetic abnormalities that can cause difficulties with airway management, but the most commonly encountered craniofacial syndromes that can cause difficulty right from birth include Treacher–Collins syndrome, the Pierre Robin sequence, and Goldenhar syndrome. Each of these syndromes involves some element of mandibular dysplasia, micrognathia, and retrognathia that can make intubation with a standard laryngoscopy extremely difficult. Goldenhar syndrome is especially challenging since these patients typically have asymmetrical hypoplasia of the mandible which makes it extremely difficult to get a good mask fit for bag-mask ventilation (Fig. 1b, c). In a busy neonatal practice, these syndromes can be seen quite often since the incidence of Treacher–Collins syndrome, Pierre Robin sequence, and Goldenhar syndrome is 1 in 10,000–50,000, 1 in 8,500–10,000, and 1 in 3,000–5,000 live births, respectively. One of the most dreaded situations that can be faced in a neonate with a difficult airway is the neonate who presents with CHAOS—Congenital High Airway Obstruction Syndrome. This syndrome consists of patients whose upper airways are distorted by either teratomas, hemangiomas, or lymphatic malformations. Due to the high probability of needing a surgical airway, these patients are usually delivered via an EXIT, or ex utero intra partum, procedure. The placental bypass allows for oxygenation of the neonate while securing the airway. In many instances, endotracheal intubation is performed via rigid bronchoscopy or immediate placement of a tracheostomy.
Other examples of abnormal airway anatomy related in neonates potentially include the former premature infant with a past history of prolonged intubation in the neonatal intensive care unit. Although a number of these patients are successfully extubated and no obvious problems with their airways, careful attention should be made to ascertain any history of stridor/croup; these patients can occasionally present at induction with difficulty to ventilate and intubate due to mild micrognathia or occult subglottic stenosis (Fig. 2a–d). The patient with a history of prolonged intubation while in the NICU can also present months later with stridor secondary to cyst formation. Patients with airway hemangiomas can also present months after birth with stridor since the natural course of these lesions is for them to enlarge before involuting (Fig. 3).
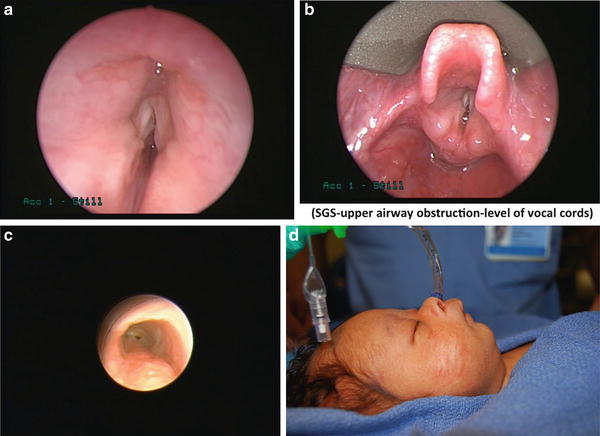
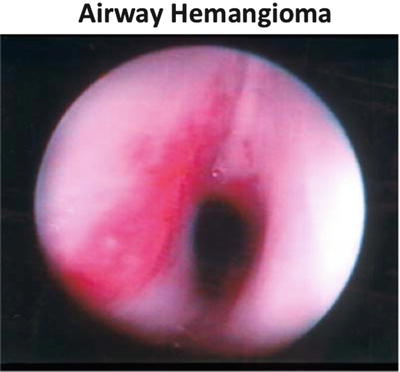

Fig. 2
2 (a), (b), (c) Bronchoscopy photos showing various forms of glottic and subglottic abnormalities such as subglottic stenosis, vocal cord paralysis and vocal cord atresia posing anesthesia challenges. (Courtesy of Ian Jacobs and Steve Sobol, CHOP). (d) Neonate with micrognathia posing anesthesia challenges. (Courtesy of Jesse Taylor, CHOP)

Fig. 3
Airway hemangioma, (Courtesy of Ian Jacobs, CHOP).
In terms of abnormal physiology, patients with cyanotic cardiac disease not uncommon can have issues with craniofacial syndromes causing airway difficulties due to these lesions being midline defects. These patients are truly challenging when trying to secure their airways since they are hypoxic to begin with and have very little reserve. The patients with an abnormal airway and chronic lung disease secondary to prematurity are also complex to manage due to a limited respiratory reserve. This difficulty can be further compounded by the presence of pulmonary hypertension, which can accompany the chronic lung disease.
Intraoperative Management
Approach to Securing the Airway in Neonate with a Difficult Airway
What is a Difficult Airway?
According to the report by the American Society of Anesthesiologists (ASA) Task Force on Management of the Difficult Airway, a standardized definition of the difficult airway cannot be identified in the literature [3]. The definition suggested by this task force is “The clinical situation in which a conventionally trained anesthesiologist experiences difficulty with face mask ventilation of the upper airway, difficulty with tracheal intubation, or both [3].” As such, the difficult airway is not a single entity or disease state, but a clinical endpoint associated with a wide variety of diseases and conditions. The extant literature on difficult airway management is replete with case series and case reports describing various techniques that have been effective in specific scenarios by specific providers. However, owing to the myriad of clinical conditions associated with the difficult airway in children and the relatively low incidence, systematic studies to determine the optimal management are lacking (Fig. 2b).
Principles of Difficult Airway Management: The ASA Difficult Airway Algorithm
Evaluating some prospective randomized clinical trials that evaluate various management strategies can be problematic, and the approach to the airway must be tailored to the specific airway problem of the available resources. In 1993, the American Society of Anesthesiologists (ASA) Task Force on Management of the Difficult Airway published a set of practice guidelines for difficult airway management in adults which have since been updated again in 2013 [1]. Due in large parts to the paucity of high-level evidence in the literature, these guidelines were developed based on the available information and expert opinion/consensus to provide rational guidance to practitioners. The algorithm included in these guidelines (Fig. 4) has become the gold standard for most adults when managing difficult airway clinical decision-making. As a framework, this algorithm provides an appropriate decision tree for individual patient management. Furthermore, the algorithm represents guidelines that, while widely adopted, do not necessarily represent the standard of care, especially in neonatology. In fact, one of the initial assessments proposed by the ASA guidelines that can affect the decision tree/pathway in the algorithm is whether there is difficulty in patient cooperation or consent. The neonate is similar to the uncooperative adult patient; a two-way dialogue cannot be held with the neonate if one is considering an awake intubation. However, it is common and thought to be a safe practice to intubate a neonate awake. In many pediatric institutions, the ASA Difficult Airway Algorithm has been modified to their specific patients and utilized to create emergency airway algorithms such as we use in our institution (Fig. 5).
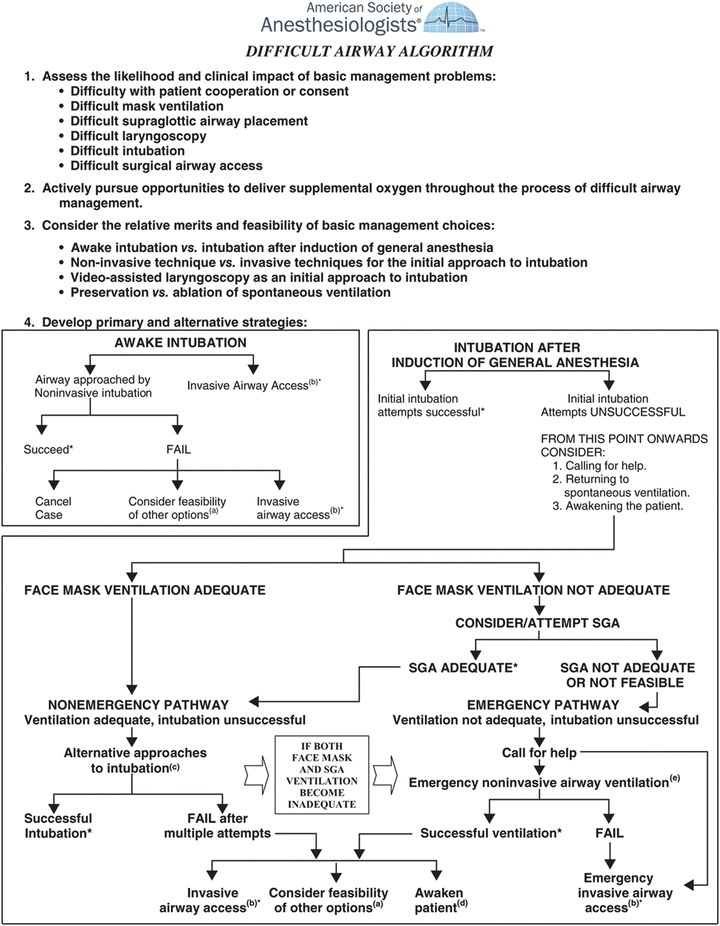
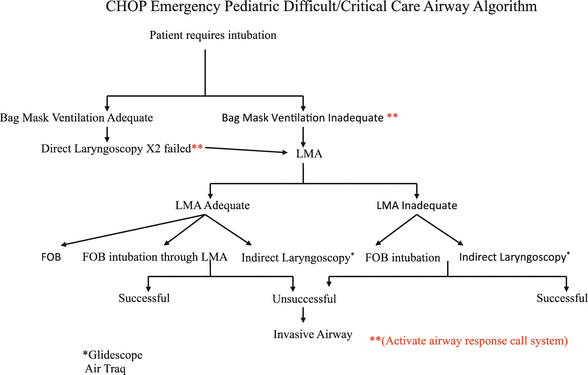

Fig. 4
American Society of Anesthesiologist difficult airway algorithm (Reprinted from [3]. With permission from Wolter Kluwers Health

Fig. 5
CHOP difficult/critical airway algorithm
In reviewing the ASA algorithm, two basic questions need to be answered when approaching the patient with a potentially difficult airway; can I ventilate? can I intubate? In most situations, where there is a high likelihood that one cannot ventilate then there should be a consideration to either intubate the neonate awake or keep the patient breathing spontaneously with light sedation or through the use of an inhalational anesthetic. Difficult mask fits are typically seen in many craniofacial syndromes. For neonates with these problems, these difficulties can be circumvented with a Laryngeal Mask Airway (LMA) (Fig. 6).
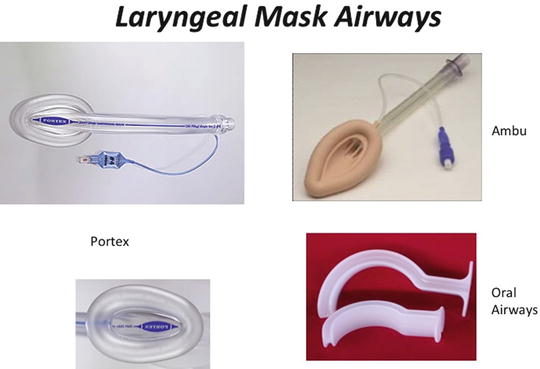

Fig. 6
Variety of laryngeal mask airways, (LMA’s), and oral airways
The ASA difficult airway algorithm highlights the use of the laryngeal mask airway (LMA) as a rescue device. First introduced in 1983, the LMA has revolutionized the approach to the patient with a difficult airway not only as a rescue device, but also as a conduit for placement of an endotracheal tube into the trachea [4]. Its use as a rescue device is described in the International Neonatal Resuscitations Guidelines [5, 6]. This device allows for the ability to oxygenate and ventilate a patient while using a fiberoptic bronchoscope. The LMA has also been proven to be useful as a way to perform an inhalation induction in a neonate when it is difficult to get a mask fit such as in patients with Goldenhar syndrome [7]. A topical local anesthetic can be applied to the oropharynx of the patient to reduce airway activation when the LMA is placed awake; however, this is not necessary in most neonates.
In the situation where there is a potential for a difficult intubation, multiple plans for securing the airway with the use of different techniques and standard and advanced devices should be developed prior to attempting to secure the airway.
Preparation for managing the difficult airway should take several factors into consideration such as anatomy, physiology, pharmacology, and equipment choices. As outlined above, congenital anomalies of the face along with masses obstructing the airway are the most common causes of difficulties with the neonatal airway. The unique differences in cardiorespiratory function exponentially increase the difficulty, whether the laryngoscopist is a neonatologist or anesthesiologist sedative, and induction agents have been used successfully during intubations for neonates [8]. For most anesthesiologists, the approach is usually an inhalation induction since the patient can be slowly anesthetized, continue to breath spontaneously, and maintain their airway since muscle tone is still present. The use of large doses of intravenous induction agents and muscle relaxants leads to apnea and can be detrimental if the patient cannot be bag-mask ventilated. In situations where an inhalation induction may still be risky in patients with airway masses or severe anomalies, these patients can be “sedated” with small doses of agents such as ketamine, midazolam, or dexmedetomidine [9] (Table 1). Muscle relaxants, such as succinylcholine, vecuronium, or rocuronium, should only be considered if a patient could be satisfactorily bag-mask ventilated and further use of sedative or induction agents will potentially lead to cardiovascular instability (Table 2). Neuromuscular blockade can be advantageous in some scenarios. The advantages include a motionless patient, jaw relaxation, and removal of the risk of laryngospasm. Some of the potential disadvantages of neuromuscular blockade are essentially the opposites of the advantages for spontaneous ventilation—namely, a greater potential for upper airway soft tissue collapse with loss of the ability to ventilate, and the risk of arterial desaturation rapidly due to apnea during prolonged intubation attempts. Another disadvantage is gastric distension from manual positive pressure ventilation resulting in compromised ventilation (through abdominal competition) and/or regurgitation of gastric contents. This can be particularly problematic in infants and smaller children where an inflated stomach can significantly reduce functional residual capacity
Table 1
Comparison of three sedation regimens for airway management
Midazolam/Fentanyl | High-dose dexmedetomidine | Ketamine | |
|---|---|---|---|
Sedation | ++ | + | ++ |
Analgesia | ++ | + | +++ |
Amnesia | ++ | +/– | +++ |
Reversible | Yes | No | No |
Clinician familiarity | +++ | +/– | ++ |
Pt responsive to commands | + | + | No |
Respiratory drive | – | ++ | + |
Adjuncts required | No | No | No |
Table 2
Drugs used for intubation
Drugs | Doses |
|---|---|
Sedatives | |
Midazolam | Starting dose: 0.05 mg/kg IV—bolus to a total max of 0.2 mg/kg |
Fentanyl | Starting dose: 1 mcg/kg IV—bolus to a total max of 2–3 mcg/kg |
Ketamine | Starting dose: 0.5 mg/kg IV—bolus to a total max of 1.5–2.0 mg/kg |
Dexmedetomidine | Starting bolus: 1 mcg/kg IV—bolus to a total max of 3 mcg/kg |
Infusion: 0.5 mcg/kg/h IV | |
Paralytic agents | |
Succinylcholine | 1–2 mg/kg IV |
Vecuronium | 0.1 mg/kg IV |
Rocuronium | 1 mg/kg IV |
Basic Equipment
With any approach to securing a difficult airway, a primary plan and backup plans need to be formulated. Each approach/plan should define the equipment necessary to secure the airway, whether it is a supraglottic airway such as an LMA, an endotracheal tube, or a tracheostomy tube. Inherent to any approach is a progression from basic to more complex equipment.
The basic equipment required for any approach to any airway includes airway adjuncts such as oral and nasal airways, laryngoscopes with blades, a bag-mask ventilation system, and an LMA as a rescue device [10] (Fig. 7). For most neonates, there should be two laryngoscopes available with straight blades, Miller #0 and #1. As described above, the anatomical differences allow for the straight blade to be used like a curved blade: the tip of the blade is placed in the vallecula to raise the epiglottis. The Rendell–Baker mask, a hard black rubber mask with little dead space, was the standard mask utilized with a manual bag-mask ventilator system; however, this has been supplanted by the modern clear mask with an inflatable rim. The inflatable rim allows for a tight seal by conforming to almost any contours of the face. It is imperative this rim be checked for function prior to using it; poor inflation or leak of the rim can lead to difficulties in bag-mask ventilation. The next important basic device to have is the LMA. As mentioned before, the LMA serves both as a rescue device and a conduit for placing an endotracheal tube [11]. Its basic design as a mask that sits over the larynx allows for easy identification of the vocal cords and displacement of soft tissues of entire airway allowing for easier entry into the trachea with a fiberoptic bronchoscope. An endotracheal tube preloaded on the bronchoscope can then be easily passed into the trachea. Difficulties with earlier versions of the LMA were encountered due to the flexible grill over the aperture of the LMA, which made passing the tube difficult. Furthermore the length of endotracheal tubes was similar to the length of the airway tube of the LMA making it difficult to insert the tube completely through the mask without adjuncts. Also, the pilot balloon on cuff endotracheal tubes would not easily pass through the tube of the LMA. A newer version of a laryngeal mask, the AirQ® (Fig. 8) is designed specifically to allow as a conduit for fiberoptic intubation. It has no grill over the aperture, the airway tube has a relatively greater diameter and shorter length, and the airway connector is easily disconnected. It also comes in a variety of sizes for a variety of sized patients. The #1 size has been used as an adjunct to fiberoptically intubate neonates [12].
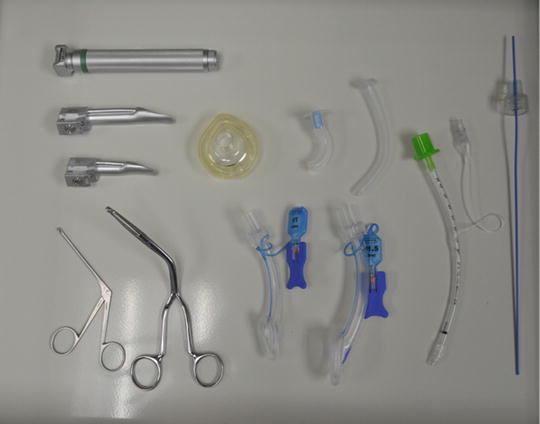
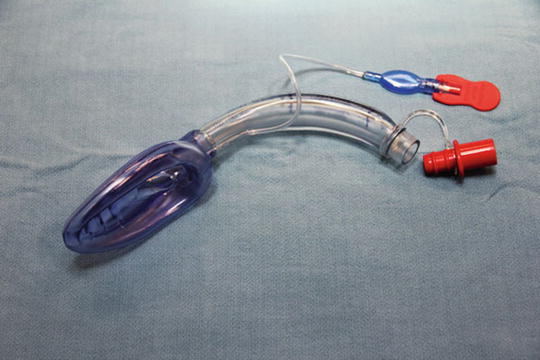

Fig. 7
Neonatal intubating equipment kept handy in an emergency airway cart. (Courtesy of Janet Lioy, neonatal airway program-CHOP)

Fig. 8
Air Q LMA for indirect intubation through tube lumen
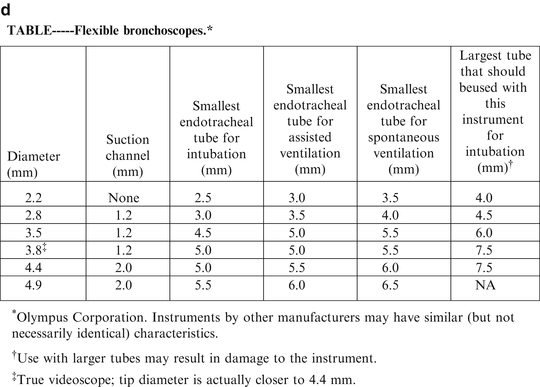
Although not usually thought of as a basic device, the fiberoptic bronchoscope is routinely used in the next step to secure a difficult airway, whether either as a stand-alone device or in conjunction with a laryngeal mask. Its ability for the tip to be manipulated makes it an essential piece of equipment. For neonates, there are two sizes of scopes that can be used, the 2.2 or 2.8 mm scope. The measurement describes the outside diameter of the scope, thus, the 2.2 scope can accommodate a 2.5 mm inside diameter endotracheal tube and the 2.8 can accommodate an endotracheal tube 3.0 mm or greater. The 2.8 scope has a suction channel to remove secretions which can obscure the view of the airway, but the 2.2 scope does not. Due to the small size of the visual fields it can sometimes be difficult to ascertain anatomic structures by viewing through the eyepiece on the scope. Attaching a camera to the scope will transmit the image to a larger monitor that can enhance the view of the airway. This also allows other individuals at the bedside to help assist in identifying structures. Newer models of scopes are now incorporating a video “chip” at the tip which essentially builds integrates a camera into the scope and allows for a higher resolution.
Typically, in most operating room suites and critical care areas, most of the equipment described above to secure a difficult area are stored together in a difficult airway cart. The cart, as is our institution will include other equipment such as oral airways to facilitate an oral fiberoptic intubation, Magill forceps to allow manipulation of the tip of the endotracheal tube in the posterior pharynx, airway exchangers, tracheostomy dilators, and needle cricothyroidotomy kits. In our own institution, our carts also have an Airtraq camera with screen mounted to the top of the cart and are stored in the same area with fiberoptic bronchoscopes and GlideScopes so they can be easily obtained at the same time (Fig. 9).


Fig. 9
Emergency airway neonatal cart. (Courtesy of Janet Lioy, neonatal airway program-CHOP)
The rest of this section will now focus on advanced equipment and techniques, specifically indirect laryngoscopic devices that are used to secure difficult airways. Not one device is better than the next device; however, anatomic conditions will dictate the choice of one device versus another. Comfort and skill of the operator will also direct decisions on a device. There are many different devices on the market but the ones presented have been used successfully for securing the airway in a neonate. It is recommended that practice should be obtained with simulation and normal airways prior to incorporating them to secure a difficult airway.
Advanced Equipment and Techniques
Video and Optical Laryngoscopes
Video and optical laryngoscopes have transformed adult airway management. They incorporate a camera into the tip of a curved or straight blade. Their use in neonates has been limited because the earlier designed models were ineffective. Newer designs are now smaller and more appropriate for neonates. Although video and optical laryngoscopy is easier to learn than direct laryngoscopy, a learning curve still remains. Deliberate practice and kinesthetic skill is necessary for successful intubation. Because a camera is placed in the vicinity of the larynx, video-laryngoscopes provide a better view of the glottis and improve intubation success in simulated difficult intubations [13, 14].
GlideScope™
The GlideScope™ is the most popular and most studied video-laryngoscope.
The original GlideScope™ design (GVL 2) was too large for infants and neonates and was associated with repeated failures when used in neonates [15–17]. The analog GlideScope Cobal™t and digital GlideScope AVL™ videolaryngoscopes (GCV; Verathon Medical, Bothell, WA) are newer designs made strictly for pediatric use (Fig. 10a–c). The blades are narrower (10 mm compared to 14.5 mm) and are available in a full complement of pediatric sizes. The Cobalt model consists of a flexible video baton which provides a high-resolution image on a video monitor. This baton is inserted into different-sized disposable plastic blades called Stats. The size 1–2 video baton is most suitable for infants and neonates. Specifically, the manufacturer recommends the following weight-based guidelines for the various blades; size 0 blade (patients <1.5 kg), size 1 blade (patients 1.5–3.6 kg), and size 2 blade (patients 1.8–10 kg). The size 0 blade too is ideal for preterm neonates, size 1 for full-term neonates, and size 2 for infants.