(1)
Department of Ophthalmology and Visual Sciences, University of Iowa Hospitals and Clinics, Iowa City, IA, USA
Central retinal artery occlusion (CRAO) has been known as a clinical entity since 1859, when von Graefe [1] first described CRAO due to embolism. After that, Schweigger [2] in 1984 described it on ophthalmoscopy. A voluminous literature has accumulated on its various aspects. It presents with classical clinical findings: a dramatic, marked visual loss in the involved eye, with typical fundus findings.
My experimental [3–14] and clinical [6, 15–18] studies on CRAO, together with a critical review of all this voluminous literature, have revealed that our understanding of CRAO is riddled with serious misconceptions, particularly about its management. These have resulted in major controversies about its various aspects, particularly concerning the usefulness and rationale of proposed treatments.
Classification of CRAO
CRAO has always been considered to be one disease. However, my studies [19, 20] showed that in fact it consists of the following four distinct clinical entities, with different clinical findings, as discussed below:
1.
Non–arteritic (NA) CRAO: This includes eyes with the classical clinical picture of permanent CRAO with retinal infarction, cherry-red spot (Fig. 13.1), absent or poor residual retinal circulation on fluorescein angiography, and no evidence of giant cell arteritis.
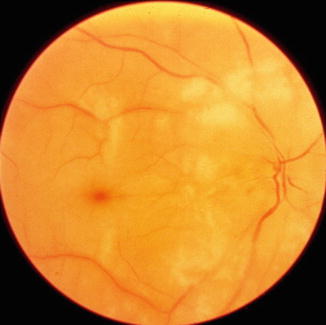

Fig. 13.1
Fundus photograph of the right eye with non-arteritic CRAO shows cherry-red spot and retinal opacity of posterior fundus
2.
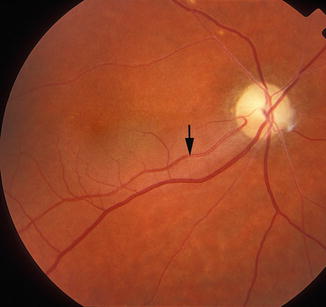
NA–CRAO with cilioretinal artery sparing (Figs. 13.2, 13.3, 13.4, and 13.5): Since the presence and size of a patent cilioretinal artery in permanent NA-CRAO can have a marked influence on the visual outcome and retinal circulation, it is imperative not to lump these eyes together with the type 1 above.
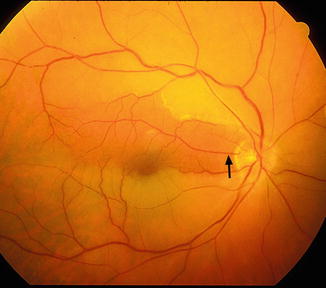
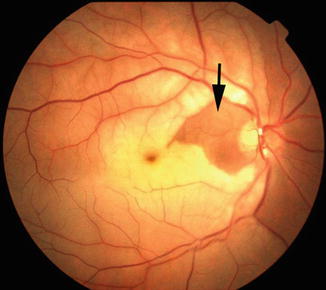
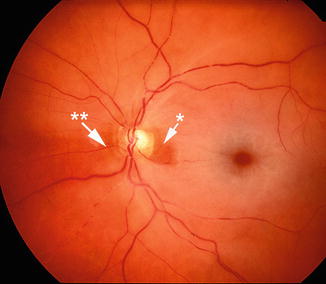
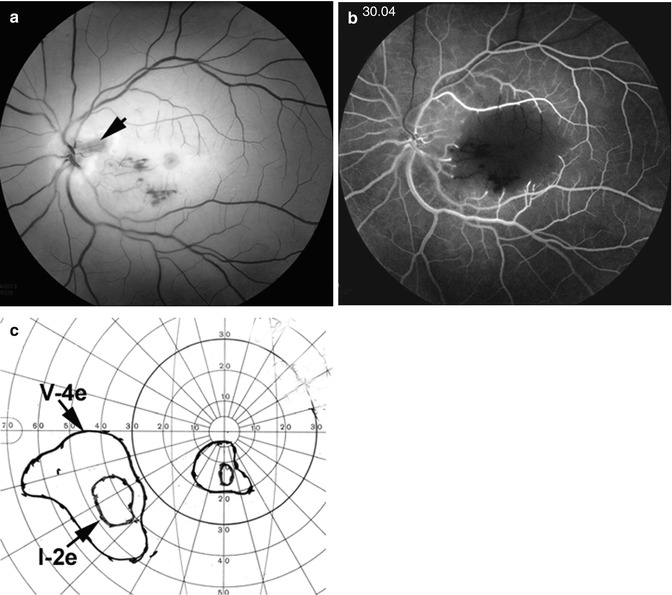

Fig. 13.2
Fundus photograph of the right eye with non-arteritic CRAO and patent cilioretinal artery (arrow)

Fig. 13.3
Fundus photograph of the right eye with non-arteritic CRAO and patent cilioretinal artery (arrow). It shows cherry-red spot; retinal opacity of posterior fundus, most marked in the macular region; and two emboli – one in the superior and the other one in the inferior branch of the central retinal artery

Fig. 13.4
Ophthalmoscopic appearance of the left eye with non-arteritic CRAO, shows cherry-red spot, retinal opacity of posterior fundus – most marked in the macular region, boxcarring the retinal vessels, and associated with two patent cilioretinal arteries – a tiny one in the temporal peripapillary region (one asterisk) and a small one nasal to the optic disc (two asterisks) (Reproduced from Hayreh [21])

Fig. 13.5
Fundus photograph (a) and fluorescein fundus angiogram during retinal arteriovenous phase (b) 4 days after the onset of CRAO and visual field defects (c) 3 months after that. (a) Fundus photograph shows cherry-red spot, a small patent cilioretinal artery (arrow), and a few retinal hemorrhages. (b) Fluorescein angiogram, during the retinal arteriovenous phase slightly delayed filling of the retinal arteries and veins, with central macular region showing no capillary filling. (c) Visual fields plotted with a Goldmann perimeter show two small island fields only – close to the center due to the patent cilioretinal artery
3.
Arteritic CRAO: In this type, giant cell arteritis is the cause of development of permanent CRAO. My studies on giant cell arteritis have shown that these eyes almost invariably have associated arteritic anterior ischemic optic neuropathy [22, 23]. Therefore, the visual loss is the result of acute ischemia, not only of the retina but also of the optic nerve head. Clinically, these eyes have the classical fundus findings of CRAO with or without optic disc edema, but, most importantly, on fluorescein angiography there is evidence of a posterior ciliary artery occlusion in addition to CRAO (Fig. 13.6). Without fluorescein fundus angiography, the presence of a posterior ciliary artery occlusion and the diagnosis of giant cell arteritis may be missed, resulting in the tragedy of bilateral blindness that could have been prevented by early institution of high-dose steroid therapy.
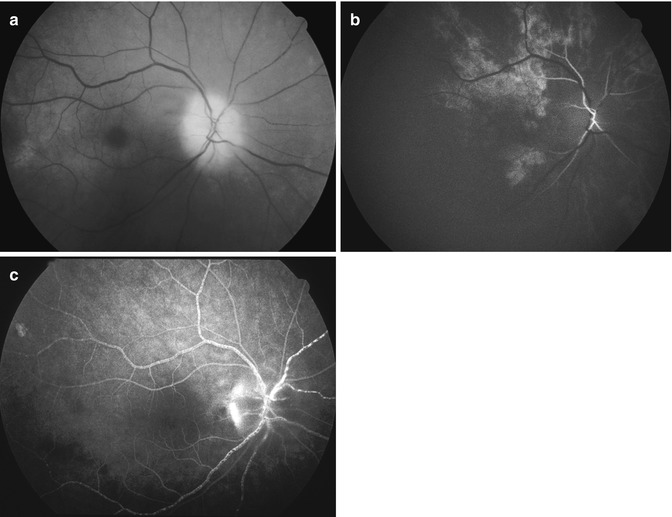

Fig. 13.6
The right eye with arteritic CRAO and arteritic anterior ischemic optic neuropathy. (a) Fundus photograph showing chalky-white optic disc edema and some mild boxcarring (cattle-trucking) in the retinal vessels and cherry-red spot with perifoveolar retinal opacity. (b) Fluorescein angiogram, during the early phase, showing a few small patches of choroidal filling and early filling of the central retinal artery. (c) Fluorescein angiogram during the late phase showing boxcarring (cattle-trucking) in the retinal vessels, with almost no disc staining
4.
Transient NA–CRAO (Figs. 13.7, 13.8, 13.9, and 13.10): The visual outcome in this type of CRAO can be totally different from any of the other types, depending upon the duration of transient CRAO, which may vary from several minutes to many hours. Its diagnosis is based on a history of marked, sudden visual loss (not amaurosis fugax) and classical fundus findings of CRAO but normal retinal circulation on initial fluorescein angiography.
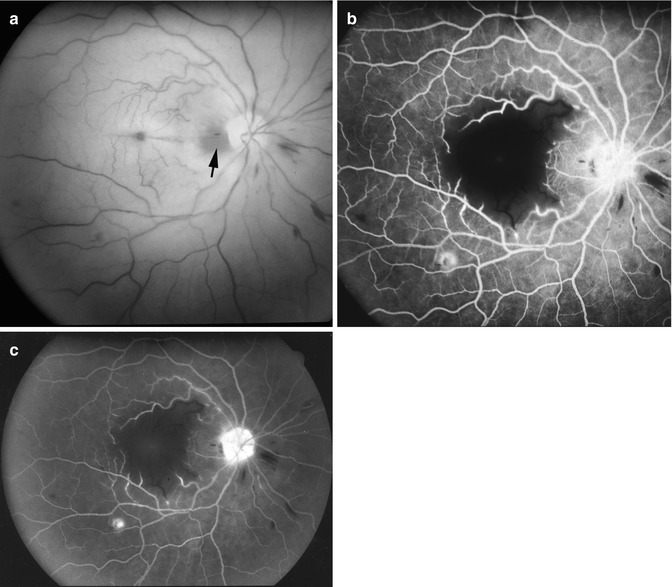
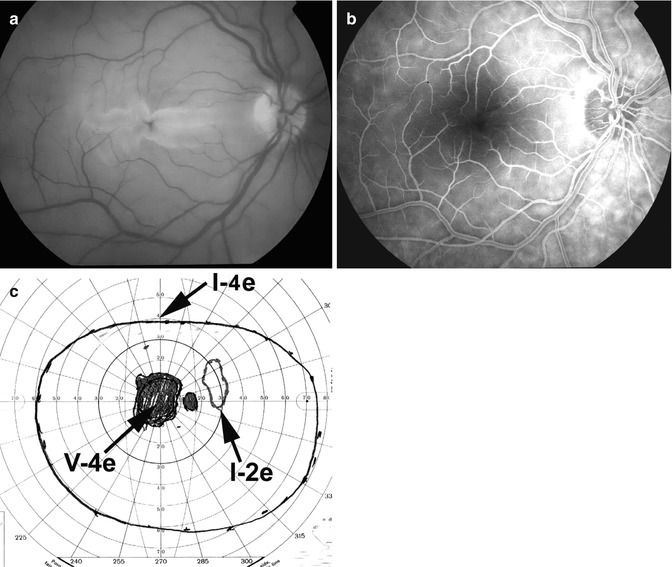
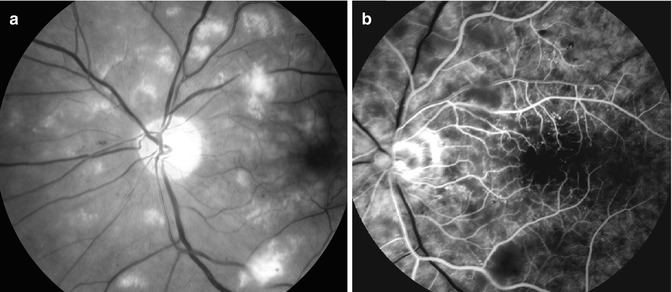
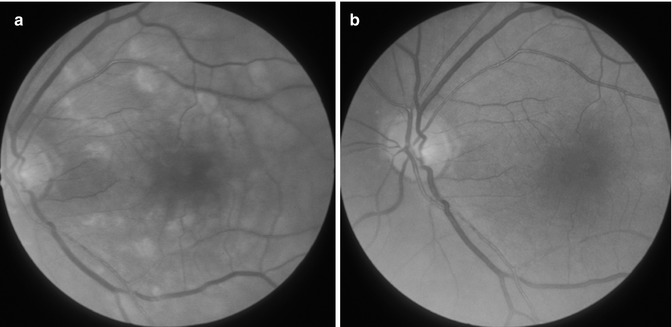

Fig. 13.7
Fundus photograph (a) and fluorescein angiogram (b, c) of the right eye at initial visit in an eye with transient non-arteritic CRAO 10 days earlier. (a) Fundus photograph shows cherry-red spot, retinal opacity of the posterior fundus – most marked in the macular region – and a small area of normal retina temporal to the optic disc corresponding to a patent cilioretinal retinal artery (arrow). (b) Angiogram during the retinal arteriovenous phase shows normal filling of the retinal vascular bed with complete absence of filling in the macular region, corresponding to the area with most marked retinal swelling. (c). Angiogram during the late phase shows persistence of the complete absence of filling in the macular region (Reproduced from Hayreh and Zimmerman [19])

Fig. 13.8
Fundus photograph (a) and fluorescein angiogram (b) of the right eye 1 day after development of transient non-arteritic CRAO and visual fields (c) 3 months after that. (a) Fundus photograph shows marked central macular retinal opacity and swelling and cherry-red spot, and the rest of the retina looks normal. (b) Fluorescein angiogram during the retinal arteriovenous phase shows normal filling of the retinal vascular bed, with complete absence of filling in the central macular region, corresponding to the area with most marked retinal swelling. (c) Visual field plotted with a Goldmann perimeter shows a large absolute central scotoma, and normal peripheral visual fields with I–4e isopter

Fig. 13.9
Fundus photograph (a) and fluorescein fundus angiogram during retinal arterial phase (b) of an eye with transient CRAO. Fundus photograph shows a large number of cotton-wool spots, maximum in the macular region. Fluorescein angiogram shows almost normal but slightly sluggish retinal circulation except for the absence of filling in the foveal region, and cotton-wool spots at places mask the background fluorescence (Reproduced from Hayreh [21])

Fig. 13.10
Fundus photographs of an eye with transient CRAO: (a) During the early stages shows large number of cotton-wool spots. (b) Later on when the retinal opacities have resolved, the fundus looks normal
Mechanism of Development of Transient NA-CRAO
This may be due to two mechanisms:
(a)
An embolus may get impacted in the central retinal artery and dislodge itself after some time.
(b)
Due to a transient fall of perfusion pressure below the critical level in the retinal vascular bed. To understand the mechanism of this, it is essential to know the factors that can cause a marked fall of the perfusion pressure. The perfusion pressure in the central retinal artery is equal to the mean blood pressure in the artery minus the intraocular pressure (IOP). Therefore, a fall of perfusion pressure can be due to the following two factors:
(i)
Marked fall in mean arterial blood pressure: This can occur for several reasons, including nocturnal arterial hypotension (Fig. 13.11) – particularly in patients who take blood pressure-lowering medicines in the evening or at bedtime [25, 26] – severe shock, during hemodialysis, spasm of the central retinal artery, marked stenosis or occlusion of the internal carotid or ophthalmic artery, or ocular ischemia [17, 18]. My experimental study in atherosclerotic monkeys showed that serotonin (5-hydroxytryptamine) released by platelet aggregation on atherosclerotic plaques, in the presence of atherosclerotic lesions, triggers transient, severe vasospasm of the central retinal artery, resulting in transient CRAO [27] (Fig. 13.12).
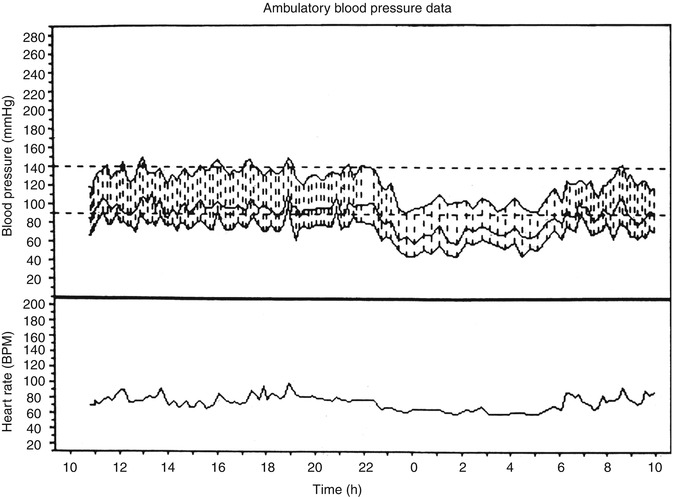
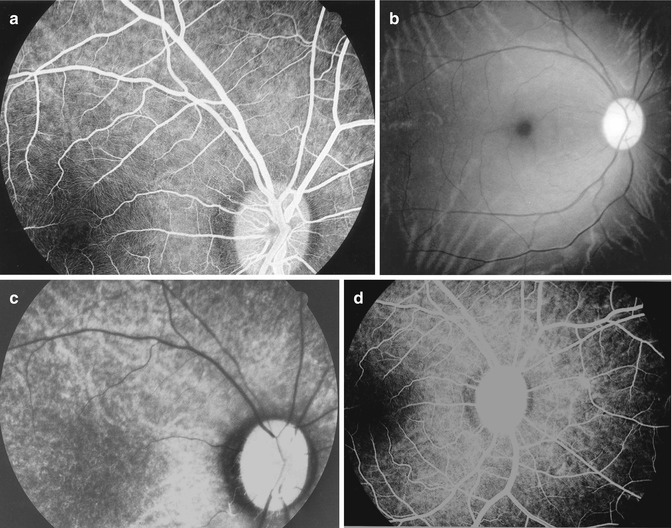

Fig. 13.11
A 24-h ambulatory blood pressure recording, starting at about 11 AM and ending at about 10 AM the next day. Note that during the waking hours the blood pressure is perfectly normal but shows a marked drop during the sleeping hours (nocturnal arterial hypotension) (Reproduced from Hayreh et al. [24])

Fig. 13.12
Fluorescein fundus angiograms (a, b, d) and a fundus photograph (b) of the right eye of an atherosclerotic monkey before and after infusion of serotonin. (a) Baseline angiogram during atherosclerotic phase, showing normal filling of the central retinal artery and the posterior ciliary artery circulations, 1 week before (b, c). (b, c) During infusion of serotonin. (b) A fundus photograph about 2 min after the start of serotonin infusion, showing evidence of CRAO, including pale optic disc, retinal opacity, and so-called cherry-red spot at the fovea. (c) An angiogram about 4 min after the start of serotonin infusion, showing normal filling of the posterior ciliary artery circulation but complete occlusion of the central retinal artery. (d) An angiogram during regression phase, showing once again normal filling of the central retinal artery and posterior ciliary artery circulations (Reproduced from Hayreh et al. [27])
When the IOP goes above the mean blood pressure or the blood pressure falls below the IOP, there is no retinal blood flow. Eyes with already poor perfusion pressure in the central retinal artery are especially vulnerable to CRAO with the fall in perfusion pressure, particularly with a fall of mean blood pressure, which acts as “the straw that breaks the camel’s back” [17, 18]. Since these patients suffer from transient CRAO lasting for several hours, usually during sleep, many of them typically give a history of discovering visual loss on waking in the morning or after hemodialysis or surgery. Fluorescein angiography performed during waking hours, when the perfusion pressure has returned to normal, then reveals normal retinal blood flow, in spite of severe visual loss and all the classical signs of CRAO. I have found that this normal angiographic finding has frequently misled ophthalmologists about the reason for the development of CRAO.
Thus, transient CRAO can be produced not only by transient impaction of an embolus in the central retinal artery but also by all these other factors. In transient CRAO, the visual outcome naturally depends upon the duration of CRAO. Thus, transient CRAO can be caused by a whole host of factors, other than embolism.
Conditions Associated with CRAO
There is a voluminous literature describing systemic conditions associated with CRAO. It is possible that some of them may not actually reflect a cause-and-effect relationship. Following is a summary of the conditions described in the literature.
Hyperlipidemia and Atherosclerosis
High cholesterol leads to the buildup of plaque on the walls of arteries, which can block arteries. Hyperlipidemia causes atherosclerosis. Atherosclerosis is the most common cause of cardiovascular disease, which is responsible for substantial mortality and morbidity. Hodgson [28] published the first chemical analysis of atherosclerotic plaque in 1815, and Lobstein [29] introduced the term “atherosclerosis” in 1829.
Historically, most interestingly, a recent study of atherosclerosis across 4,000 years of preagricultural human history investigated mummies from ancient Egypt, ancient Peru, Ancestral Puebloans of Southwest America, and the Unangan of the Aleutian Islands by CT scan [30]. Atherosclerosis was found in 38 % of ancient Egyptians, 25 % of ancient Peruvians, 40 % of Ancestral Puebloans of Southwest America, and 60 % of the Unangan hunter-gatherers. It was present in the aorta in 20 % and carotid arteries in 12 %. Age at the time of death was positively correlated with atherosclerosis. Atherosclerosis has been found in autopsies of ancient Egyptian mummies, including that of Amenhotep II, King of Egypt from 1450 to 1425 BC [31]. A study by Allison et al. [32] in modern-day human beings found atherosclerosis by the age of 50 years in the carotid, coronary, and aorta in 82 % of men, and 68 % of women. Atheromatous plaques have been demonstrated in the arteries of 50 % of people in Europe and North America who died before the age of 50 and 90 % of those who died before the age of 75 [33].
Atherosclerosis is wrongly assumed to be a disease of the elderly. An autopsy study [34] of a large multiethnic United States of America sample of all those aged 15–19 years showed early stage aortic atheroma and more than 50 % had coronary lesions. Among young American soldiers killed in the Korean war 40 %, averaging 22 years of age, had atherosclerotic plaques in the coronary arteries [35]. Studies have further shown that the disease process of atherosclerosis begins in childhood, with fatty streaks found in the aortas of children older than 3 years and in the coronary arteries by adolescence [35, 36].
Atherosclerosis essentially involves the large arteries, although it occurs in arteries of all sizes, including the small ocular arteries. It is a patchy disease, characterized by endothelial injury and the proliferation of arterial smooth muscle cells. Endothelial dysfunction is an early event in atherogenesis, preceding formation of plaques [37, 38]. Typically there is deposition of lipid material (atheromata) under the intima, followed by connective tissue proliferation; the mass eventually undergoes hyaline degeneration and ultimately calcification. This forms an atheromatous plaque. The degenerated material may slough away to leave an ulcerated surface, upon which platelets accumulate and thrombus may form.
I have discussed the subject of atherosclerosis in detail elsewhere [39]. Atherosclerosis of the carotid arteries is an important and well-known risk factor for CRAO (see below).
Diets high in saturated fats have been implicated in thrombosis, and it has been suggested that the increased sensitivity of platelets to aggregation results from altered platelet membrane cholesterol or fatty acid contents or both [40–43]. Platelets may be activated by a diet high in saturated fat in apparently normal people [44, 45]. Platelet aggregate on the atheromatous plaques can release serotonin – a potent vasoconstrictor [27] (see below). Many experimental and clinical studies have shown that normalization of cholesterol level in atherosclerotics produces regression of atherosclerosis. In monkeys, structural [46, 47] and functional [47–52] improvements have been shown with regression of atherosclerosis. Functionally, in atherosclerotic monkeys, hyperresponsiveness to serotonin is abolished by regression of atherosclerosis [27, 47–49, 52, 53], and that effect usually happens within 4–8 months after cholesterol levels are normalized [47]. In patients, treatment of hypercholesterolemia for 6–12 months restores endothelial function in coronary arteries [51, 54–56]. Reduction of cholesterol decreases the number of cerebrovascular events by one third [57]. Regression of atherosclerosis and associated vascular abnormalities caused by dietary treatment and normalization of cholesterol levels have important clinical implications for the prevention of vascular accidents and other atherosclerotic vascular diseases.
In patients with CRAO, hyperlipidemia is a common finding, reported by a large number of studies. Following are the findings from two large studies: Schmidt et al. [58] reported hyperlipidemia in CRAO in 38 % of 208 patients with CRAO. In my study [59] of CRAO in 234 patients, cholesterol ≥240 mg/dL was seen in 31 % males and 52 % females with CRAO. The actual incidence may be higher, because many patients were already on statin drugs to lower elevated cholesterol, particularly LDL, when first seen in my clinic.
Carotid Artery Disease
The association of carotid artery disease with CRAO is well known. Atherosclerotic lesions of the common and/or internal carotid artery, and marked stenosis or occlusion of the internal carotid artery are well-established causes of CRAO. Embolism is the most common cause of CRAO in these cases. Stenosis or occlusion of the internal carotid artery can produce ocular ischemia, and that can lead to ocular ischmic syndrome (see Chap. 21). CRAO can also be associated with aneurysm of the internal carotid artery [60], dissection of the common carotid artery/spontaneous internal carotid artery dissection [61–66], internal carotid artery thrombosis [67], carotid endarterectomy [68], after carotid angiography [69], after manipulation of the neck by a chiropractor [70], and, rarely, with arterial fibromuscular dysplasia [71, 72] and pseudoxanthoma elasticum [73]. Also embolization from the external carotid artery system can cause CRAO [75].
Carotid artery disease can produce CRAO by the following three mechanisms:
1.
Embolism: This is by far the most common cause of CRAO. In 1958, Hollenhorst first described the embolization of cholesterol crystals to the retinal circulation [74]. He later reported that these retinal emboli were associated with carotid atherosclerosis, visual loss, and strokes [76]. Dunlap et al. [77], in a retrospective study of 130 consecutive patients with a diagnosis of Hollenhorst plaque, CRAO, or branch retinal artery occlusion on ophthalmoscopy, found that 73 % of patients had Hollenhorst plaque and 26 % had branch retinal artery occlusion. Hollenhorst plaques were on the right side in 48 %, on the left in 41 %, and bilaterally in 11 %. Among the patients who had carotid bifurcation lesions ipsilateral to the ocular findings, stenosis was <30 % in 70 %, between 30 and 60 % in 22 %, and >60 % in only 8 % (eight patients); of these eight patients, six had >80 % carotid artery stenosis. No patient had a definitive cardiac or aortic arch source for the retinal findings. The findings of my study [59] related to this are discussed in detail below (Table 13.1). There is a case report of a 49-year-old man who developed CRAO after chiropractic neck manipulation [78]. The patient had diffuse atherosclerotic plaques in the common carotid artery.
Table 13.1
Carotid Doppler/angiography and echocardiogram findings on the side with central retinal artery occlusion
Test finding | Non-arteritic central retinal artery occlusion |
|---|---|
Patent foramen ovale | (n = 234) |
6 (2 %) | |
Carotid Doppler/angiography: | (n = 199) |
Occlusion | |
0–15 % | 109 (55 %) |
16–49 % | 23 (12 %) |
50–79 % | 32 (16 %) |
80–99 % | 13 (7 %) |
100 % | 22 (11 %) |
Carotid Doppler/angiography: | (n = 187) |
Plaque present | 133 (71 %) |
Echocardiogram | (n = 131) |
Normal | 39 (30 %) |
Abnormal, no embolic source | 24 (18 %) |
Abnormal, with embolic source | 68 (52 %) |
Echocardiogram: | (Of n = 68 with embolic source) |
Abnormal embolic source | |
Mitral valve | 18 (26 %) |
Aortic valve | 26 (38 %) |
Mitral and aortic valves | 24 (36 %) |
Echocardiogram: | (Of n = 42 with mitral valve as embolic source) |
Mitral valve lesion types | |
Calcified valve | 24 (57 %) |
Mitral valve prolapse | 7 (17 %) |
Other types of lesions | 11 (26 %) |
Echocardiogram: | (Of n = 50 with aortic valve as embolic source) |
Aortic valve lesion types | |
Calcified valve | 39 (78 %) |
Other types of lesions | 11 (22 %) |
Arruga and Sanders [79], in 70 patients with evidence of retinal embolism, found cholesterol emboli (Hollenhorst plaque) in 74 %, calcific emboli in 10.5 %, and platelet-fibrin emboli 15.5 %. They stated that the carotid artery and the heart are the most common sources of embolism to the retinal arteries. In my study [59] of 234 eyes with CRAO, the most common abnormality and source of embolism in the carotid arteries was plaque(s) (in 71 %). Sheng et al. [80] in a review of a series of 62 patients with CRAO found ipsilateral extracranial carotid occlusive disease in 56 %; these patients did not generally have carotid bruits and had normal noninvasive carotid tests. They concluded that over half of patients with CRAO who undergo carotid angiography will have a carotid lesion on the ipsilateral side. This suggests that CRAO is a significant marker for extracranial carotid disease and should be an indication for complete carotid evaluation.
2.
Hemodynamically: The internal carotid artery has to be significantly stenosed (usually about 70 % or more), or completely occluded to produce hemodynamically induced retinal and/or ocular ischemia, by markedly reducing the ocular blood flow. This can also occur, rarely, with spontaneous internal carotid artery dissection. A fall of blood pressure, particularly nocturnal arterial hypotension (Fig. 13.11), with a markedly stenosed or occluded internal carotid artery, can result in transient CRAO [19]. Severe stenosis of the extracranial internal carotid artery is the most common identified condition associated with retinal and ocular ischemia [16, 18, 81, 82]. Sharma et al. [81] found hemodynamically significant carotid artery stenosis in only 19 % of patients with acute retinal artery occlusion. In my study [59], ≥80 % stenosis of the internal carotid artery was seen in 18 % of CRAO cases (Table 13.1). I have found that the presence of a retinal embolus is a poor predictor of hemodynamically significant carotid stenosis on carotid Doppler, as has also been pointed out by others [81, 83, 84].
3.
Serotonin (5–Hydroxytryptamine)-Induced Arterial Spasm: In our study on atherosclerotic monkeys, we found that serotonin, in the presence of atherosclerotic lesions, can cause transient, complete occlusion or impaired blood flow in the central retinal artery by producing a transient spasm [27] (Fig. 13.12). Serotonin, released by platelet aggregation on atherosclerotic plaques in the carotid artery, may produce vasospasm of the central retinal artery in atherosclerotics and thus may contribute to CRAO and retinal ischemic disorders.
Diabetes Mellitus and Arterial Hypertension
The association of diabetes and arterial hypertension with CRAO has been reported by a large number of studies [58, 59, 85]. Two large studies of CRAO have given detailed information on the incidence of diabetes and arterial hypertension [58, 59]. Schmidt et al. [58] found the incidence of diabetes mellitus at 21.6 % in 208 patients and of arterial hypertension in 80 % of 247 patients. In my study [59] of 234 patients with CRAO, the incidence of diabetes was 20 % and of arterial hypertension 52 %, and compared to the age-period matched US population, both had significantly higher prevalence (p ≤ 0.0001). The development of CRAO in a patient with metabolic syndrome x has been reported [86].
Cardiac Causes
A variety of cardiac conditions have been reported associated with CRAO due to embolism from the heart. Schmidt et al. [58], in a detailed study of 183 patients with CRAO, reported atrial fibrillation in 19 %, aortic stenosis in 13 %, atrial thrombus in one, aortic valve sclerosis in 10 %, aortic insufficiency in 2 %, prior aortic valve replacement in 1 %, patent foramen ovale in 5 %, mitral valve insufficiency in 9 %, and mitral valve prolapse in 1. In my study [59] of 234 eyes with CRAO, patent foramen ovale was present in 2 %. In 131 persons, echocardiogram was normal in 30 %, abnormal but not showing any embolic source in 18 % and showing an embolic source in 52 %. Among the embolic mitral valve sources, there was mitral valve calcification in 57 %, prolapse in 17 %, and other types of lesions in 26 %. Among the embolic aortic valve sources, there was aortic valve calcification in 78 % and other types of lesions in 22 %. There are other case reports of cardiac abnormalities, including myxoma in the left atrium [67, 87–90], patent foramen ovale [91–93], infective endocarditis [94, 95], and paradoxical brain embolism as a cause of CRAO [96], also cardiac catheterization [97]. I have seen CRAO cases due to embolism from bacterial endocarditis and aortic valve replacement.
Echocardiography is the best way to determine cardiac abnormalities. There is a common belief that echocardiography rules out the heart as the source of retinal microembolism, which is not always the case, because its resolution or the technique used may not be sensitive enough to detect very small valvular or other cardiac lesions. As has been pointed out by several studies, including mine, when routine transthoracic cardiac echography shows no abnormality, transesophageal echography may reveal abnormalities.
Following Invasive Cardiovascular Procedures
There are reports of development of CRAO or branch retinal artery occlusion following invasive cardiovascular procedures, such as stent implantation, carotid artery stenting, cardiopulmonary bypass for coronary artery bypass grafting, transcatheter balloon aortic valvuloplasty, coronary angiography, and cardiac catheterization (because of the risk of developing transient embolism in retinal circulation) [98–103]. These reports are mostly based on anecdotal case reports only.
Vasculitis and Chronic Systemic Autoimmune Diseases
CRAO has been reported, associated with acquired immunodeficiency syndrome causing CRAO alone [104, 105] or in combination with central retinal vein occlusion [106], Behçet’s disease [107–109], Fabry’s disease [110], Bartonella henselae infection (cat-scratch fever) [111, 112], herpes zoster ophthalmicus [4, 113–119], endophthalmitis [120, 121], orbital cellulitis [122–125], mucormycosis [116–128], periocular necrotizing fasciitis [129], sarcoidosis [130], Henoch-Schonlein purpura [131], Churg-Strauss syndrome [132–140], polyarteritis nodosa [141–144], systemic lupus erythematosus [145–148], Wegener’s granulomatosis [149–156], antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis [157], scleroderma [158, 159], and giant cell arteritis [22, 23, 160, 161]. Development of CRAO has been described following interferon treatment of hepatitis C [162, 163]; that is due to severe inflammatory disorder occurring during interferon-alpha therapy. Crohn’s disease is associated with vasculitis and can produce CRAO [164, 165] as well as ophthalmic artery occlusion [165]. There is a case report of CRAO combined with optic neuropathy in a young patient with miliary tuberculosis [166]. von Hippel [167] reported a case of tuberculosis of the choroid in which short stretches of two retinal arterioles were closed by intimal proliferation, with epithelioid and giant cells present. In Susac’s syndrome, there is usually branch retinal artery occlusion, but there is one report where there was CRAO [168]. Susac’s syndrome is a self-limited syndrome, involving the vasculature of the brain, retina, and inner ear. Susac et al. [169] did a brain biopsy in one case, which revealed sclerosis of the media and adventitia of small pial and cortical vessels, suggestive of a healed angiitis. The latest thinking is that its pathogenic mechanism is an immune-mediated endotheliopathy. CRAO development has also been reported following varicella [170, 171], postviral vasculitis [172], and toxoplasmic papillitis [173]. Development of CRAO in Sneddon syndrome has been described [174, 175]; it is a progressive, noninflammatory arteriopathy leading to the characteristic skin condition and to cerebrovascular problems including stroke and transient ischemic attack.
Thrombophilia
There are several reports suggesting that thrombophilia plays a role in the development of CRAO. The various thrombophilic factors reported include high homocysteine [176–181], antiphospholipid antibodies [182–185], factor V Leiden [186, 187], prothrombin G20210A mutations [187, 188], thrombocytopenic purpura [189], essential thrombocythemia [190], protein S deficiency [191], and cryoglobulinemia [192].
Salomon et al. [193], in a thrombophilia study of 21 patients with retinal artery occlusion, reported that 9 of them had at least one thrombophilic marker: 4 were homozygous for MTHFR C677T, 1 was heterozygous for factor V G1691A, 1 had a high titer of IgM anticardiolipin, 2 were heterozygous for factor V G1691A and homozygous for MTHFR C677T, and 1 had lupus anticoagulant, a high titer of IgM anticardiolipin, homozygosity for MTHFR C677T, and hyperhomocysteinemia. They concluded that homozygous MTHFR C677T was a significant risk factor with odds ratio of 3.18 (95 % CI 1.20–8.47). Greiner et al. [194] did not find an increased prevalence of activated protein C resistance in patients with CRAO or branch retinal artery occlusion when compared to the normal population. Miserocchi et al. [184] found anticardiolipin antibodies in retinal vasculitis and other inflammatory disorders of the eye. Weger et al. [195] in a study of retinal artery occlusion found the prevalence of heterozygosity for the prothrombin G20210A variant, and the distribution of factor V Leiden genotypes did not significantly differ between patients and controls.
Most of the information on thrombophilia is based generally on anecdotal case reports and/or small numbers of cases and often combined cases of central and branch retinal artery occlusion, in some cases even with central retinal vein occlusion. It is quite possible that factors, other than for CRAO, may play a role in these findings, and not CRAO per se. My study on CRAO did not confirm that thrombophilia plays a significant role. I find the recommendation that all patients with CRAO should be evaluated for thrombophilia is not advisable, unless there are some other indications for it.
There is a report of development of postpartum CRAO [196]; that patient had hypercoagulability.
Malignant Diseases
Sickle Cell Disease
CRAO has been reported in sickle cell hemoglobinopathies [207–226]. It is seen in African Americans with a history of hemoglobin sickle cell disease, often young persons. The association of sickle cell disease per se with CRAO is uncommon; most reports have described additional contributing factors, such as trauma [211] or concomitant systemic illness [207, 210], or retrobulbar injection [209, 213, 214]. In a patient with a sickling hemoglobinopathy, intravascular sickling of erythrocytes results in arterial occlusion, particularly when there is decreased perfusion. It has been suggested that vascular obstruction in sickle cell disease can involve several mechanisms, including abnormal endothelial adhesion by sickled erythrocytes, coagulation activation, and intimal injury [219, 220]. CRAO has been described much less frequently in sickle cell hemoglobinopathy (double heterozygous hemoglobin SC) than in sickle cell disease (homozygous hemoglobin SS) [219, 220], despite the nearly equal prevalence of sickle cell and SS disease [217].
Perioperative CRAO
Perioperative CRAO, i.e., following non-ocular surgery, is a well-known cause of visual loss. Surgery can cause ischemic optic neuropathy (usually posterior ischemic optic neuropathy [227, 228]) or CRAO. The most common cause of perioperative CRAO is spinal surgery [229–242], but it has also been reported following arthroplasty of the hip [243], sinus surgery [244–247], nasal septoplasty [248], cosmetic blepharoplasty [249–251], and recurrent spheno-orbital meningioma surgery [252]. CRAO development has also been described following optic nerve sheath decompression surgery [253].
The pathogenesis of development of perioperative or surgical posterior ischemic optic neuropathy is discussed in detail elsewhere [228]. Perioperative CRAO can be caused by the following two mechanisms:
1.
Compression of the Eye During Surgery: This causes a rise in IOP due to compression by prone position. In spinal surgery, there may also be severe and prolonged arterial hypotension (due to prolonged general anesthesia, surgical trauma, and massive blood loss); in spinal surgery, the surgeons often want the blood pressure to be low, to prevent bleeding. Under such circumstances, even a moderate rise of IOP may be enough to stop the circulation in the central retinal artery. This produces transient CRAO – the mechanism of this is discussed above. That can also happen in any surgical procedure where there is compression of the eyeball. The prognosis for the visual loss depends upon the duration of transient CRAO.
2.
Marked Orbital Edema in Spinal Surgery: In this, surgery is usually done in a Trendelenburg position. Prolonged surgery in this position can result in marked orbital edema and chemosis. Blood flow in the central retinal artery depends upon the difference between the arterial and venous pressures in the retinal vascular bed. As discussed above, these patients often have low blood pressure. Marked orbital edema leads to increased intraorbital pressure, which compresses the orbital veins, and that increases the pressure in the central retinal vein. Blood flow depends upon the difference between the arterial and venous pressures. Thus, a combination of low blood pressure and raised venous pressure in the retinal vascular bed can result in compromised retinal circulation, leading to CRAO. The same thing can happen in radical neck dissection, because ligation of the jugular veins can result in raised orbital venous pressure.
Systemic cardiovascular disease can further contribute to this. When there is arterial hypotension during surgery, some anesthesiologists give vasopressor agents to increase blood pressure; that can be dangerous, because these agents increase blood pressure by constricting the terminal arterioles. Constriction of the terminal arterioles has a paradoxical effect – proximal to the arterioles, there is increased blood pressure (which gives a false sense of security), but distal to the terminal arterioles (due to constriction of the arterioles), there is marked reduction of blood flow in the capillary bed, resulting in ischemic damage.
There is a case report of a 45-year-old man who underwent a nasal septal surgery for severe epistaxis and developed CRAO with third cranial nerve palsy [248]. Postoperative computerized tomography scan revealed multiple fractures of the left medial orbital wall, including one near the optic canal.
Following Orbital, Eye, or Head Injuries
There are reports of CRAO associated with severe orbital blow-out fracture [254–257], retrobulbar hemorrhage due to facial trauma [258], traumatic optic nerve avulsion [259], and traumatic carotid cavernous fistula [260, 261]. CRAO has also been reported following blunt ocular trauma [262]. Carotid cavernous fistula may develop even without any trauma and result in CRAO [263]. The causes of CRAO in these cases vary. Severe blow-out fractures can cause CRAO due to increase in IOP and/or displacement of the eyeball into the maxillary sinus through a blow-out fracture of the lower orbital wall, orbital emphysema [264, 265], and optic nerve avulsion causing tearing of central retinal artery [259, 266]. It is well known that retrobulbar hemorrhage can result in a marked rise of IOP which can obliterate the central retinal artery. In carotid cavernous fistula [260, 261], there is a rise in IOP, and that can compromise the retinal circulation, particularly in persons with other vascular factors, e.g., arterial hypertension, atherosclerosis, and diabetes.
There are two reports of patients who developed bilateral CRAO following head injury [267, 268]. In one report, a 30-year-old patient developed bilateral CRAO following head injury [267]. A scan of the head and orbits showed multiple frontal contusions and a depressed fracture of the roof and lateral wall of the left orbit. In the second case, X-ray of the skull and computed tomography scan of the brain did not reveal any abnormality except a mandibular fracture [268]. The cause of CRAO in the first report was most probably the depressed fracture of the roof and lateral wall [267]. In the second case [268], visual loss developed approximately 2 h after the initial injury, and there was no evident cause, except that there was slightly elevated cholesterol and low-density lipoprotein and those might have caused atherosclerotic change in the carotids.
Following Facial or Retrobulbar Injections
Development of CRAO has been reported after autologous fat injection in the glabellar area [269, 270], cosmetic facial filler injections [271], and following forehead injection with a corticosteroid suspension [272]. In one case, a 30-year-old woman, occlusion of the ophthalmic artery associated with CRAO developed following autologous fat injection in the glabellar area [269]. Another case was an 18-year-old man; after polidocanol-carbon dioxide emulsion into the glabellar subcutaneous hemangioma, the next day he had lid swelling, and 3 days later at referral, the visual acuity in the left eye was no light perception, because of CRAO [270]. Park et al. reported 12 consecutive patients with retinal artery occlusion caused by cosmetic facial filler injections [271]. In all these cases, most likely the material got injected into one of the branches of the ophthalmic artery under pressure, and from there that got into the ophthalmic artery and finally into various branches of the ophthalmic artery. Fluorescein angiography in such cases has shown evidence not only of CRAO but also of occlusion of the posterior ciliary artery [270]. In one of these cases, cerebral angiography showed complete obstruction of the ophthalmic artery by the material [269]. Development of acute CRAO has been reported following a local anesthetic with adrenaline on the nasal mucosa [247, 273]. CRAO can also develop following retrobulbar anesthetic injection [274–276].
Following Hemodialysis
I have seen a few patients who developed CRAO following hemodialysis, and there are several similar reports in the literature [277–288].
Mechanism of Development of CRAO Following Hemodialysis
These patients represent a unique group that almost invariably has marked vasculopathy because they suffer from diabetes, arterial hypertension, and chronic renal failure. Many of them also have diabetic retinopathy and/or hypertensive retinopathy. These factors make them highly susceptible to CRAO. It is well known that during hemodialysis there is often a marked fall of blood pressure. Also some studies [278–287] have described an increase in IOP during hemodialysis, although that is not present in every case [288]. Perfusion pressure in the retinal circulation is equal to the difference between the mean blood pressure and the IOP. Persons with all the risks factors detailed above experience a marked fall of blood pressure and at the same time a rise of IOP during hemodialysis, and that compromises the retinal circulation and results in transient CRAO.
Following Intravitreal Injection of Anti-vascular Endothelial Growth Factor (VEGF) Therapy
Intravitreal injection of anti-VEGF drugs is commonly used for macular edema due to various causes, neovascular age-related macular degeneration, and ocular neovascularization. It can cause two types of raised IOP:
(i)
Immediate Increase in IOP After Intravitreal Anti–VEGF Injection: This is transient and common, because of the volume increase in the eye. In one study [289], the mean preoperative IOP was 22.4 ± 5.5 mmHg in the supine position. Immediately after the injection, the IOP increased to 47.9 ± 15.1 (range 23–82). The mean difference between preoperative IOP and immediately after the injection was 25.5 ± 13.6 mmHg. IOP increased significantly in a considerable number of patients after receiving 0.05-ml intravitreal ranibizumab injections. IOP takes longer to reach a safe level in patients with a history of glaucoma or ocular hypertension.
(ii)
Sustained Long–Term High IOP After Intravitreal Anti–VEGF Injection: This is not uncommon; there are many such reports [290–305]. Neovascular age-related macular degeneration is common and is treated with long-term intravitreal anti-VEGF therapy; associated elevated IOP has been reported. For example, Tseng et al. [301], in a study of 25 eyes with neovascular age-related macular degeneration, reported increased IOP while the patients were receiving interval doses of intravitreal ranibizumab and/or bevacizumab. The mean highest IOP while receiving intravitreal anti-VEGF therapy was 35.8 mmHg (range 23–58 mmHg). Overall, 23 of the 25 patients required IOP management. It has been reported that there is a statistically significant IOP increase in patients treated with repeated injections of ranibizumab [305]. Sustained elevated IOP can happen even in normotensive eyes undergoing intravitreal bevacizumab treatment for neovascular age-related macular degeneration; the studies suggest that patients with preexisting glaucoma who receive either bevacizumab or ranibizumab have a high risk for further elevation of IOP, and they may rarely need glaucoma filtering surgery. I recently saw a patient with central retinal vein occlusion, who was treated by her local ophthalmologist by six intravitreal injections of Lucentis (an anti-VEGF drug), the last about one a month before I saw her; when seen in my clinic on her first visit, she had IOP in the involved eye of 60 mmHg, with IOP of 16 mmHg in the fellow eye.
Development of CRAO has been reported following intravitreal injection of anti-VEGF therapy [290, 299]. This is more likely to happen in eyes with preexisting ocular and systemic risk factors causing poor perfusion pressure in the central retinal artery, for instance, in eyes with ocular ischemia. Perfusion pressure, as discussed above, is equal to the difference between the mean blood pressure and the IOP. In eyes with low mean blood pressure for any reason, even a small rise in IOP can precipitate development of CRAO. Development of non-arteritic anterior ischemic optic neuropathy has been reported after intravitreal injection of anti-VEGF drugs [306–309]. An Internet-based survey designed to identify adverse events associated with intravitreal bevacizumab treatment showed reports of CRAO, elevation of blood pressure, transient ischemic attack, cerebrovascular accident, and even death [291]. I know of one patient who had a stroke after intravitreal injection of anti-VEGF drug for central retinal vein occlusion.
It is well established that eyes treated with intravitreal triamcinolone have a very high incidence of increased IOP. That high IOP can be a risk factor for development of CRAO in susceptible eyes.
Following Peribulbar Anesthesia
Following Intraocular Gas Expansion
Intraocular gas is used as tamponade for rhegmatogenous retinal detachment. Expansion of the gas, for one reason or another, can raise the IOP markedly, and that can result in development of transient CRAO [313–319]. Expansion of intraocular gas has been reported on climbing to a high altitude [316, 317], by nitrous oxide during general anesthesia [318], and commercial airline travel [319]. Similarly, intravitreal injection of “heavy silicone oil,” used for retinal tamponade after vitrectomy for complicated retinal detachment, can result in an increase of IOP which can cause transient CRAO [320].
Fabry’s Disease
CRAO has been reported in patients with Fabry’s disease [110, 321–325]. Fabry’s disease is an X-linked lysosomal storage disease, and it causes thrombosis. It has been suggested that that may be due to buildup of globotriaosylceramide in the endothelial cells of blood vessels [325]. Utsumi et al. [325], in a series of 65 patients with Fabry’s disease, found a high incidence of thrombosis (15 %) and had one patient with CRAO and thrombophlebitis.
Miscellaneous Causes
CRAO has been reported associated with different conditions based on one or a few cases. These include incontinentia pigmenti [326], following embolization procedure for juvenile nasopharyngeal angiofibroma [327–330], oral contraceptives [67, 331–333], radial optic neurotomy for central retinal vein occlusion [334], recreational usage of cocaine [335–338], disseminated intravascular coagulation in neonate [339], snake bite [340–344], Sydenham’s chorea [345–357] (in which it is due to embolism from the valvular lesions associated with rheumatic heart disease) [349, 351, 353, 355–357], optic disc drusen [358–360], radiation treatment [361, 362], migraine [360, 363, 364], loiasis [365], Marfan’s syndrome [366], nephrotic syndrome [367], electrical burn injuries [368], osteogenesis imperfecta [369], and YAG synechialysis [370].
The list above concerns CRAO in all age groups. I further analyzed the causes according to age, in two groups: (1) CRAO in children; the United Nations Convention on the Rights of the Child defines a child as “a human being below the age of 18 years,” and CRAO has been reported in as young as neonates [339]. (2) Young persons are defined between the ages of 18 and 44 years.
CRAO in Children
In this age group, CRAO associated with a variety of conditions has been reported in anecdotal case reports or based on a few cases. These include carotid artery disease [69, 74], cardiac myxoma [89, 90, 371], patent foramen ovale [93], infective endocarditis [95], transcatheter balloon aortic valvuloplasty with aortic valvular stenosis [99], herpes zoster ophthalmicus [4, 119, 170], orbital apex syndrome [123], orbital cellulitis [125], Henoch-Schonlein purpura [131], polyarteritis nodosa [143], Crohn’s disease [164], miliary tuberculosis [166], chicken pox [170, 171], postviral vasculitis [172], hyperhomocysteinemia [176, 180], primary antiphospholipid antibody syndrome [181], factor V Leiden and prothrombin G20210A mutations [183], leukemia [198], T-cell lymphoma [200], systemic non-Hodgkin’s lymphoma [202], disseminated intravascular coagulation [339], retinoblastoma [205, 206], sickle cell disease [219, 230, 232, 233], spinal surgery [238, 240, 256], traumatic optic nerve avulsion [259], blunt ocular trauma [262], Fabry’s Disease [321], incontinentia pigmenti [326], embolization from injection in juvenile hemangioma [327], juvenile nasopharyngeal angiofibroma [328], snake bite [341, 342], Sydenham’s chorea [345, 347, 348, 350–353, 357], and a few other miscellaneous conditions [58, 59, 372]. The underlying mechanisms of development of CRAO in the conditions in children are discussed above in the general discussion dealing with CRAO.
CRAO in Young Persons
In this age group, reports of CRAO associated with a variety of conditions have appeared, based on one or few cases. These include carotid artery dissection [61, 62], internal carotid artery thrombosis [67] and fibromuscular dysplasia [71], diabetes and arterial hypertension [67], atrial myxoma [67, 87, 371], patent foramen ovale [92], infectious endocarditis [94], acquired immune deficiency syndrome [104, 105], Behçet’s disease [108, 109], Bartonella henselae infection [111], herpes zoster ophthalmicus [116, 118], periocular necrotizing fasciitis [129], Churg-Strauss syndrome [135], polyarteritis nodosa [143, 144], systemic lupus erythematosus [145, 146, 148], Wegener’s granulomatosis [152], scleroderma [158], Susac’s syndrome [168, 169], toxoplasmosis [173], antiphospholipid antibodies [174], homocystinuria [177, 178], protein S deficiency [191], cryoglobulinemia [192], postpartum [196], systemic non-Hodgkin’s lymphoma [201], chronic myeloid leukemia [197], sickle cell disease [214, 216, 219, 221, 223–225], local anesthesia for blepharoplasty [251], intranasal anesthetic injection [247], rhinoplasty [246], spinal surgery [239, 242], orbital severe blow-out fracture [254, 255], orbital emphysema causing raised orbital pressure [264, 265], head injury [267, 268], autologous fat injection in the glabellar area [269], intralesional injection of sclerosing agent into glabellar subcutaneous hemangioma [270], retrobulbar anesthesia without retrobulbar hemorrhage [274], peribulbar anesthesia for pterygium excision [312], Fabry’s disease [322, 323], embolization in juvenile nasopharyngeal angiofibroma [329, 330], oral contraceptives [67, 332, 333], recreational usage of cocaine [335–338], use of methamphetamine [335], snakebite [340, 344], Sydenham’s chorea [346, 349, 354, 355], optic disc drusen [358, 360], melanocytoma in the optic disc [359], radiation treatment [361, 362], Sneddon disease with antiphospholipid antibodies [174], and loiasis [356]. More details of these diseases are given above.
Brown et al. [372] in 1981 in a review of the records of nine patients who developed CRAO prior to the age of 30 years found the following associated conditions: trauma in three, sickle cell disease in two, increased platelet coagulant activity in one, sickle cell with systemic lupus erythematosus and rheumatic heart disease in one, and no systemic abnormality in two.
Greven et al. [67], in 1995, studied five patients less than 40 years old with nontraumatic CRAO. They found the following associated conditions: atrial myxoma in one, internal carotid artery thrombus in one, oral contraceptive in one, diabetes mellitus and arterial hypertension in one, and none in one.
The underlying mechanisms for the development of CRAO in the conditions in young persons are discussed above, in the general discussion of causes of occlusion of the central retinal artery.
Studies Based on Large Series of Conditions Associated with CRAO
Most of the conditions associated with CRAO described above are based on anecdotal case reports or only a few cases. Following are some studies based on large series:
Karjalainen [373] in 1971, in a retrospective review of 52 CRAO patients, found associated arterial hypertension in 5, diabetes mellitus in 5, “arteriosclerosis” in 13, congestive heart failure in 3, myocardial infarction in 5, aortic valve insufficiency in 1, mitral insufficiency in 1, internal carotid artery disease in 4, bronchial carcinoma in 1, epilepsy in 1, and trauma in 2.
Schmidt et al. [58] in 2007 reported their findings in 159 patients with CRAO. They found cardiovascular risk factors in 63 %. Among these 63 %, factors were: arterial hypertension in 80 %, diabetes mellitus in 22 %, hyperlipidemia in 38 %, chronic smoking in 34 %, and internal carotid artery disease revealed by Doppler sonography in 65 % (≥70 % stenosis in 23 %, plaques in 33 %). Four patients presented with a dissecting aneurysm of the internal carotid artery. Echocardiography showed disorders of cardiac rhythm (for instance, chronic atrial fibrillation) in 19 %, aortic valve disease in 27 %, mitral valve disease in 10 %, both aortic and mitral valve disease in 4 %, and patent foramen ovale in 5 %. There was history of stroke in 5 %, transient ischemic attack in 3 %, simultaneous stroke or transient ischemic attack with retinal arterial occlusion in 2 %, and amaurosis fugax in 12 %.
Rudkin et al. [374], in a retrospective review of charts of 33 patients with CRAO, found hyperlipidemia in 36 % and arterial hypertension in 27 %. Nine patients had more than 50 % of ipsilateral carotid stenosis. One patient had significant echocardiogram findings.
In 2009, I [59] reported my findings of associated systemic abnormalities in 234 patients with NA-CRAO. There was diabetes mellitus in 20 %, renal disease in 7 %, ischemic heart disease in 26 %, stroke/transient ischemic attack in 19 %, and hyperlipidemia in 31 % of males and in 52 % of females; current smokers were 38 % men and 27 % women. Carotid Doppler/angiography and echography findings are given in Table 13.1. It shows that the most common abnormality and source of embolism in the carotid arteries is the presence of plaque(s) (66 %); significant carotid stenosis (i.e., ≥50 %) was seen in only 30 %. Echocardiography findings in this study showed 52 % with abnormal echocardiogram of an embolic source. Of those, 26 % were from the mitral valve, 38 % from the aortic valve, and 36 % from both mitral and aortic valves. The mitral valve lesions comprised 57 % calcified valve, 17 % mitral valve prolapse, and 26 % other types of lesions. The aortic valve lesions were 78 % calcified valve and 22 % of other types. Patent foramen ovale was detected in 6 patients. The finding of an abnormal echocardiogram with an embolic source was not significantly associated with stroke/transient ischemic attack (59 % in stroke/transient ischemic attack vs. 50 % without stroke/transient ischemic attack; p = 0.480) or ischemic heart disease (67 % in ischemic heart disease vs. 48 % without ischemic heart disease; p = 0.137). Relating Doppler/angiography findings to echocardiography findings showed abnormal echocardiogram with embolic source in 62 % of those with plaque and 36 % of those without plaque (p = 0.04). This means that when there are abnormalities in both Doppler/angiography and echocardiography in a patient, the embolus could have come from either the carotid artery or the heart or possibly both. One therefore has to evaluate both sources for embolism in all patients with CRAO.
In my study [59], the prevalence of diabetes mellitus, renal disease, arterial hypertension, ischemic heart disease, and stroke/transient ischemic attack in non- arteritic CRAO were significantly higher when compared to the prevalence of these conditions in the matched US population (all p < 0.0001). Smoking prevalence in non-arteritic CRAO compared to the US population was significantly higher for the male patients (p = 0.001). The data also suggested a higher smoking prevalence for female patients (p = 0.06), but that was not significant at the 0.05 significance level.
So my study [59] showed that the carotid artery and the heart are the most common causes of CRAO, by producing emboli. As discussed above, retinal emboli are usually of three types: calcific, cholesterol, and platelet-fibrin [79]. In the carotid artery, plaque is the most common source of that. In the heart, as discussed before, the sources of emboli are aortic and mitral valvular lesions, patent foramen ovale, myxoma in the left atrium, and atrial fibrillation.
There is interest in finding out about the relationship between retinal emboli and the development of stroke. Klein et al. [375, 376], in their Beaver Dam Eye Studies of 4,926 persons 43–86 years, determined the relationship of retinal emboli to the risk of stroke and ischemic heart disease mortality at 5-year [375] and 10-year incidence [376]. In their 5-year study [375], they found the prevalence of retinal arteriolar emboli was 0.9 %. After adjustments for age and sex, the prevalence of retinal emboli was associated with higher pulse pressure, arterial hypertension, diabetes mellitus, past and current smoking, and cardiovascular disease. When age, sex, and systemic factors were controlled for, people with retinal emboli had a significantly higher hazard of dying of stroke than those without retinal emboli. In their 10-year study [376], the cumulative incidence of retinal emboli was 1.5 %. After adjustment for age and sex, the incidence of retinal emboli was associated with increased pulse pressure (P = .03), higher serum total cholesterol level (P = .03), higher white blood cell count (P = .05), smoking status (P < .001), and a history of coronary artery bypass surgery (P < .001) at baseline. After controlling for age, sex, and systemic factors, people with retinal emboli had a significantly higher hazard of dying from stroke than with those without.
My study [59], by contrast, showed that an abnormal echocardiogram with an embolic source was not significantly associated with stroke/transient ischemic attack (59 % in stroke/transient ischemic attack vs. 50 % without stroke/transient ischemic attack; p = 0.480) or ischemic heart disease (67 % in ischemic heart disease vs. 48 % without ischemic heart disease; p = 0.137).
It is essential to put in proper perspective how reliable the incidence of retinal emboli in a study is. My study on fundus changes in CRAO [20] showed that migration and disappearance of retinal emboli is a common finding. (This topic is discussed below under fundus changes in CRAO.) Thus, the phenomenon of migratory emboli has practical implication in limits of determining the prevalence of retinal arteriolar emboli and their relationship with stroke or other systemic conditions.
Investigations in Patients with CRAO
From the discussion above, it is evident that patients with CRAO should have the following investigations to find the cause and to prevent or reduce the risk of any further visual problems or stroke:
1.
In all patients 50 years and older with CRAO, it is absolutely critical to rule out giant cell arteritis because that is an important ocular emergency; if it is missed, that can result in catastrophic visual loss in both eyes, which is preventable by immediate diagnosis and aggressive management with corticosteroid therapy. This fact has medicolegal implications, apart from the human tragedy involved. All these patients must have immediate evaluation of erythrocyte sedimentation rate and C-reactive protein levels – of the two, the latter is more reliable. Systemic symptoms of giant cell arteritis, when present, can be helpful. All these patients must also have fluorescein angiography because the presence of associated posterior ciliary artery occlusion is diagnostic in these cases (Fig. 13.6).
2.
Since embolism is the most common cause of CRAO, all patients with CRAO must have a complete evaluation to find the source of embolism. As discussed above, the most common sources of emboli are the carotid artery and the heart. In the carotid artery, the most common source of embolus is a plaque, so it is essential to find out if there are any plaques; unfortunately, I have found that evaluation is usually primarily focused on whether there is hemodynamically significant carotid artery stenosis, without paying attention to the presence of plaques. That can miss the actual source of emboli.
3.
Cardiac evaluation is equally essential, even if the carotid artery shows lesions, because I have found that at times a patient can have lesions in both places, and either of them can be the source of emboli. As discussed above, my study showed that the most common source of emboli in the heart is the valves, although other lesions can also be responsible (Table 13.1). Transesophageal echocardiography is the best way to evaluate heart lesions.
4.
Atherosclerosis is a common cause of lesions in the carotid artery, so it is essential to do their lipid evaluation. I have found that invariably physicians tend to look only at the total cholesterol level. I have seen many patients with perfectly normal total cholesterol levels but with high LDL, which is the most important parameter. The current guideline is to have LDL not more than 70s mg/dL. Similarly, triglycerides should also be within normal limits.
5.
As discussed above, there are many rare causes of CRAO. In difficult cases, one has to keep those in mind. In my studies, I have not found thrombophilia evaluation fruitful in CRAO.
Racial Differences in the Causes of Retinal Arterial Occlusion
Ahuja et al. [377], in a retrospective study of 29 African American and 17 Caucasian consecutive patients with a diagnosis of amaurosis fugax, CRAO, branch retinal artery occlusion, or intra-arterial retinal plaques, evaluated the causes of retinal artery occlusion. They found that their African American patients had a mean age of 61 years (range 30–77 years) and the Caucasian patients a mean age of 73 years (range 56–94 years) (P = 0.003). There was no statistically significant difference between the two groups with respect to visible emboli on ophthalmoscopy, or with regard to risk factors for arterial occlusive disease such as arterial hypertension, coronary artery disease, hypercholesterolemia, tobacco use, and history of stroke or transient ischemic attacks. But Caucasian patients had a 41 % incidence of high-grade ipsilateral internal carotid artery stenosis, compared with a 3.4 % incidence in African American patients (P = 0.002).
Cheung et al. [378] found that the prevalence of retinal emboli was lower in Asian Malaysians than in similarly aged White persons, but associated with similar cardiovascular risk factors.
Demographic Characteristics of Various Types of CRAO
Two large studies have provided detailed information on demographic characteristics of CRAO. Schmidt et al. [58] investigated 253 patients with CRAO. In that study, 6 % were under the age of 45 years, 29 % aged 45–64 years, and 65 % older than 65 years. The right eye was involved in 57 % and the left in 43 %, and 65.6 % were male.
As discussed above, my study [19] showed that CRAO is of four types. There were 244 consecutive patients with CRAO in that study. I analyzed demographic characteristics in the four types of CRAO separately. The following were the findings:
Non–arteritic CRAO: This was seen in 67 % of cases. The age range in this type was 26.5–90.4 (mean ± SD = 67.7 ± 12.3). Of these patients, 58 % were male, and 5 % had it in both eyes.
Non–arteritic CRAO with cilioretinal artery sparing: This was seen in 14 % of cases. Age range in this type was 39–87 (mean ± SD = 67.1 ± 11.6). Of these patients, 54 % were male.
Arteritic CRAO: This was seen in 4.5 % of cases. Age range in this type was 62–87 (mean ± SD = 74.4 ± 7.2). Of these patients, 36 % were male, and 18 % had it in both eyes.
Transient CRAO: This was seen in 16 % of cases. Age range in this type was 20–89 (mean ± SD = 62.7 ± 17.1). Of these patients, 46 % were male, and 5 % had it in both eyes.
Although CRAO is more common in the elderly, as is evident from the above findings, it does occur in younger age groups, and, rarely, even in infants.
Symptoms of CRAO
Typically, there is a sudden, massive loss of vision in the involved eye. It can occur at any time of the day. It was discovered on waking up in the morning in 35 % in NA-CRAO, in 29 % in NA-CRAO with cilioretinal artery sparing, in 39 % in transient CRAO, and in 30 % in arteritic CRAO [19]. When the visual loss is discovered on waking up in the morning, it may be due to embolism, thrombosis, or transient CRAO from a fall of perfusion pressure during sleep caused by nocturnal arterial hypotension [19] (Fig. 13.11). The visual loss may be preceded by transient visual obscurations, which may be due to transient migrating embolism or giant cell arteritis (GCA); some patients complain of a purplish hue to the blur – this purplish hue is highly suggestive of retinal ischemia. A history of amaurosis fugax before development of CRAO was found in 12 % in NA-CRAO, in 20 % in NA-CRAO with cilioretinal artery sparing, in 30 % in arteritic CRAO, and in 13 % in transient CRAO. Whenever a patient aged 50 years or older complains of a history of amaurosis fugax, the first essential is to rule out GCA, which is an ophthalmic emergency and, if not treated immediately, can result in massive visual loss which can involve both eyes. Hallucinations have been reported with severe vision loss associated with CRAO [237, 379, 380].
Simultaneous onset of CRAO in both eyes does not normally occur; it happens rarely, when there is compression of both eyes during a prolonged surgical procedure, or during hemodialysis.
Visual Function in CRAO
There is a classical dramatic marked visual loss. One has to consider two important issues related to visual function in CRAO. (1) Is visual loss similar in all types of CRAO? (2) What is the natural history of visual outcome in CRAO? To evaluate the beneficial effect of treatment on the visual outcome in CRAO, it is essential to have information about the natural history of visual outcome without any treatment. I investigated both these parameters in 244 patients (260 eyes) with CRAO. Moreover, visual acuity and visual field provide totally different information about the visual outcome because visual acuity is a function of the fovea only but visual fields provide information about the entire retina.
Visual acuity and visual field defects were analyzed separately for different types of CRAO, which had never been done before. As is evident from the discussion below, that is essential to get valid information. The findings are discussed at length elsewhere [19], following is a brief summary.
Initial Visual Acuity
In eyes seen within 7 days of onset, the initial visual acuity in the different types of CRAO was as follows:
NA–CRAO: 20/200–20/400 in 7 % and counting fingers or worse in 93 %
NA–CRAO with cilioretinal sparing: 20/40 or better in 20 %, 20/50–20/100 in 15 %, 20/200–20/400 in 5 %, and counting fingers or worse in 60 %
Arteritic CRAO: 20/200–20/400 in 25 % and counting fingers or worse in 75 %
Transient CRAO: 20/40 or better in 38 %, 20/50–20/100 in 7 %, 20/200–20/400 in 17 %, and counting fingers or worse in 38 %
The study showed that, among the eyes seen within 7 days of onset, initial visual acuity differed significantly among the 4 CRAO types (p < 0.0001), with visual acuity counting fingers or worse in 38 % with transient NA-CRAO but 93 % with NA-CRAO.
Final Visual Acuity
Median follow-up of eyes in this study [19] was 1.1 years (interquartile range: 0.2–3.5 years). In eyes seen within 7 days, with counting fingers visual acuity, the change in the four types of CRAO was as follows:
NA–CRAO: It improved in 22 %, remained the same in 66 %, and worsened in 12 %. Final visual acuity was 20/40 or better in 2 %, 20/200–20/400 in 19 %, and counting fingers or worse in 79 %.
NA–CRAO with cilioretinal artery sparing: It improved in 67 %, remained the same in 33 %, and did not deteriorate in any eye. It was 20/40 or better in 24 %, 20/50–20/100 in 10 %, 20/200–20/400 in 28 %, and counting fingers or worse in 38 %.
Transient NA–CRAO: It improved in 82 %, remained the same in 18 %, and did not deteriorate in any eye. It was 20/40 or better in 60.5 %, 20/50–20/100 in 10.5 %, 20/200–20/400 in 16 %, and counting fingers or worse in 13 %.
Arteritic CRAO: There were only three eyes with counting fingers visual acuity, seen within 7 days; among them, one improved, one remained the same, and one got worse.
In eyes seen more than 1 week after onset, in all types of CRAO, there was almost no change in visual acuity. Thus, the study showed that of the eyes seen within 7 days of onset and with initial visual acuity of counting finger or worse, it improved in 82 % in the transient NA-CRAO compared to 22 % in the NA-CRAO (p < 0.001). This visual acuity improvement contradicts the conventional wisdom that little visual acuity improvement can be expected in CRAO.
Visual Field Defects
As discussed above, visual acuity essentially represents the function of the foveal region and not of the rest of the retina, while visual fields plotted with a Goldmann perimeter provide full information about the function of the entire retina – after all, CRAO involves the entire retina. Unfortunately, there has been little information on the visual field defects in CRAO in the literature. Mine [19] was the first detailed, prospective, systematic study to evaluate visual fields in 145 CRAO eyes, where it was possible to plot visual fields in that study. It provided invaluable information on visual function. I plotted the visual fields with a Goldmann perimeter initially and throughout their follow-up in all eyes, because when the study was started in 1973, automated perimetry did not exit. Visual field defects in that study were divided into two categories: (1) scotomas seen in the central 30°, and (2) general visual field defects. This is because central visual field loss results in impaired visual acuity, while the peripheral visual field is essential for navigation, as discussed below.
Initial Visual Field Defects
Central Visual Field Defects
In eyes seen within 7 days of onset, in the central 30°, there was complete loss of central visual field in NA-CRAO in 48 %, in NA-CRAO with cilioretinal sparing in 22 %, and with transient NA-CRAO in 4 %. There was central scotoma in 52 % in NA-CRAO and central or paracentral scotoma in 72 % in NA-CRAO with cilioretinal artery sparing and in 62 % in transient CRAO. There was no scotoma in the central visual field in 6 % in NA-CRAO with cilioretinal sparing and in 38 % with transient CRAO.
Central scotoma was most common in all types of CRAO (Figs. 13.8, 13.13, 13.14, and 13.15); its pathogenesis is discussed below. Paracentral scotoma was the next most common.
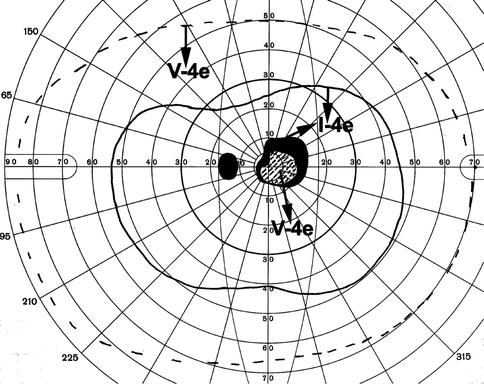
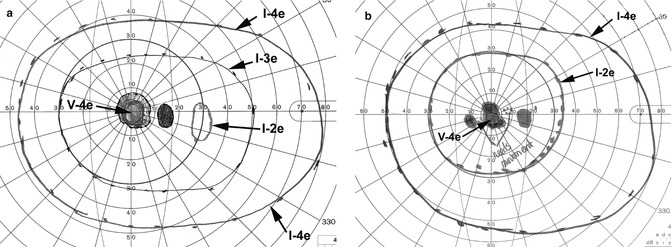
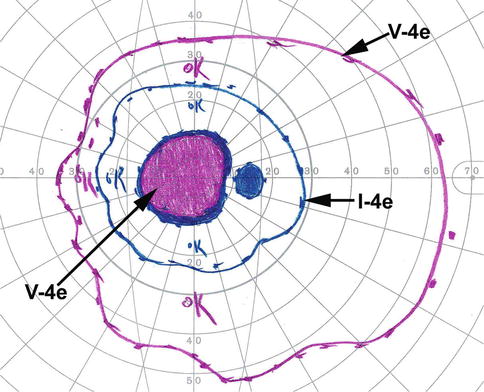

Fig. 13.13
Visual fields of the left eye, plotted with a Goldmann perimeter 2 months after the development of transient non-arteritic CRAO. It shows an absolute central scotoma, and normal peripheral visual field with I–4e and V–4e, and visual acuity of 20/200 (Reproduced from Hayreh and Zimmerman [19])

Fig. 13.14
Visual fields of the right eye, plotted with a Goldmann perimeter after the development of transient non-arteritic CRAO. (a) Initial visual fields showing small absolute central scotoma with a small temporal island field with I–2e and normal peripheral visual fields with I–3e and I–4e. (b) Visual fields 11 months later showing improvement, with smaller central scotoma and normal peripheral visual fields with I–2e and I–4e

Fig. 13.15
Visual fields of the right eye with CRAO, plotted with a Goldmann perimeter, showing a large absolute central scotoma with mild peripheral visual field constriction with I–4e and V–4e, but I–2e was not seen
Peripheral Visual Field Defects
Peripheral fields were normal in 22 % with NA-CRAO (Fig. 13.15) and in 63 % with transient CRAO (Figs. 13.8, 13.13, and 13.14); the clinical importance of intact peripheral visual fields is discussed below. The rest of the eyes had one or another peripheral visual field defect, as discussed in detail elsewhere [19]. In eyes with marked visual field loss, (1) only a temporal island field was present in 27 % with NA-CRAO, in 23 % in NA-CRAO with cilioretinal artery sparing, and in 3 % with transient NA-CRAO; (2) only a nasal island field was present in 1 % in NA-CRAO and in 3 % in NA-CRAO with cilioretinal artery sparing; (3) only a superior island field was present in 3 % in NA-CRAO; and (4) only an inferior island field was present in 3 % in NA-CRAO. Generalized constriction of the peripheral fields was more common than other types of peripheral visual field defects in eyes with NA-CRAO with cilioretinal artery sparing (in 32 %) and also in those with transient NA-CRAO (in 17 %). Eyes with NA-CRAO with cilioretinal artery sparing showed an intact central island field corresponding to the area of the retina supplied by the patent cilioretinal artery.
Final Visual Field Defects
Central Visual Field Defect
NA–CRAO: Improved in 21 %, stable in 73 %, and worsened in 6 %.
NA–CRAO with cilioretinal artery sparing: Improved in 25 %, stable in 69 %, and worsened in 6 %.
Transient NA–CRAO: Central visual field recovered to normal in 26 %, improved in 39 %, became stable in 30 %, and worsened in 4 %.
Peripheral Visual Field Defect
NA–CRAO: It recovered to normal in 6 %, improved in 39 %, became stable in 39 %, and worsened in 15 %. Because peripheral fields were normal in 22 % with NA-CRAO initially, it was finally normal in 28 %.
NA–CRAO with cilioretinal artery sparing: Improved in 19 %, stable in 75 %, and worsened in 6 %.
Transient non–arteritic CRAO: Recovered to normal in 30 %, improved in 39 %, stable in 30 %, and none worsened. Because the peripheral fields were normal in 63 % initially, it was finally normal in 93 %.
The findings of visual acuity and visual fields summarized above show that visual findings in the four types of CRAO are significantly different; combining all of them in one group results in misleading information.
Natural History of Visual Outcome in CRAO
From my findings on visual acuity and visual field loss [19], one can draw the following conclusions about the natural history of visual outcome in CRAO:
Visual acuity: Visual acuity improvement essentially occurs during the first 7 days, with minimal chance of any appreciable visual acuity improvement thereafter. Also the incidence of visual acuity improvement during the first 7 days differed significantly (p < 0.001) among the four types of CRAO. The number of cases with arteritic CRAO was too small to give any realistic corresponding data; however, since these eyes suffer from not only arteritic CRAO but also arteritic anterior ischemic optic neuropathy (see below) [22, 23], their visual loss is due to the combination of these two serious disorders, and the outcome was the worst.
Visual fields: It is evident that in all types of CRAO, both central and peripheral visual fields showed improvement in a variable number of eyes. Interestingly, in transient NA-CRAO with initial field defects, the central and peripheral visual fields recovered to normal in 26 and 30 %, respectively.
Therefore, visual outcome, as shown above, can be very different in the different types of CRAO.
Thus, my natural history study on CRAO [19] showed that, contrary to the highly prevalent impression, spontaneous improvement does occur in both visual acuity and visual fields in the first few days after onset; the extent of improvement depends very much upon the type of CRAO. As discussed below, this natural improvement of visual function has erroneously been attributed to the beneficial effect of various treatments for CRAO.
Pathogenesis of Central Scotoma in CRAO
There is a common impression that CRAO causes generalized loss of vision. However, it is not widely realized that CRAO can produce only central scotoma (the most common visual field defect in CRAO) with fairly normal peripheral visual fields (see above – Figs. 13.8, 13.13, and 13.14). The mechanism of selective development of central scotoma without any peripheral visual field defect in CRAO is as follows: It is well known that the macular region has more than one layer of retinal ganglion cells (unlike the rest of the retina), and it is the thickest part of the retina – maximal thickness being close to the foveola. Experimental [3, 8] and clinical [16, 19] CRAO studies have shown that the ischemic retinal whitish opacity and swelling are essentially located in the macular region – maximal in the perifoveolar region (Figs. 13.1, 13.3, 13.4, 13.5a, 13.7a, and 13.8a). If there is restoration of circulation in the central retinal artery, the retinal capillaries in the central, thickest part of the macular region do not refill (Figs. 13.5b; 13.7b, c, and 13.8b) because of compression by the surrounding swollen superficial retinal tissue, resulting in the “no reflow phenomenon” [9]; there is consequently permanent ganglion cell death in the non-perfused retina; the area of central retinal capillary non-filling may vary from eye to eye, depending upon the severity of retinal swelling in the macular region. This results in the variable size of the permanent central scotoma (Figs. 13.8c, 13.13, 13.14, and 13.15). Oxygen supply and nutrition from the choroidal vascular bed to the thinner, peripheral retina help in its much longer survival, and the maintenance of peripheral visual fields.
Patients with a central scotoma often learn with experience to fixate eccentrically, resulting in an apparent visual acuity improvement that does not represent a genuine improvement in the retinal function and may erroneously be attributed to a therapy. Natural history studies of various types of ocular vascular occlusive disorders have shown that for genuine visual acuity improvement, there must be simultaneous improvement in both the central scotoma and visual acuity [19, 381–385].
Therefore, it is essential to classify CRAO into its different types to make scientifically accurate observations about the visual outcome, because, as shown above, the visual outcome can be very different in the various types.
Conclusions on Visual Outcome
These findings of this study [19] on visual acuity and visual field defects show the following:
1.
The initial and final visual acuities and visual field defects are different in the four types of CRAO.
2.
Significant improvement can occur in visual acuity and visual field without treatment.
3.
Classification of CRAO is crucial for understanding differences in visual outcome. To combine all types of CRAO in one category results in misleading and invalid information on visual outcome.
Misconceptions About Visual Outcome in CRAO
There are several misconceptions prevalent about CRAO (see below). These are discussed at length elsewhere [386]. As regards the visual outcome, there are the following major misconceptions:
1.
That CRAO Causes Marked, Generalized Loss of Vision
This is an almost universal impression. As discussed above, central scotoma is the most common visual field defect in CRAO. However, it is not generally realized that CRAO can produce central scotoma only, with fairly normal peripheral visual fields (Figs. 13.8c, 13.13, 13.14, and 13.15). (The mechanism of selective development of central scotoma without any peripheral visual field defect in CRAO is discussed above.) Thus, an eye with CRAO can have only central scotoma with normal peripheral visual field.
2.
That CRAO Cannot Produce Central Scotoma Only, with Intact Peripheral Visual Fields
The above findings show that this is not true.
3.
That There Is No Chance of an Eye with Retinal Arterial Occlusion Having Spontaneous Visual Improvement
Once again, my findings show that this is not true.
4.
That Visual Acuity Testing Gives Adequate Information About the Visual Outcome in CRAO
Visual outcome most of the time is measured in the change in visual acuity only. As mentioned above, visual acuity essentially represents the function of the foveal region and not of the rest of the retina. Thus, visual acuity does not give complete information about the visual loss or recovery in CRAO. By contrast, visual fields plotted with a Goldmann perimeter provide information about the function of the entire retina. Visual field findings have important clinical implications for judging the degree of functional visual disability in CRAO. It is well established that the constant tracking provided by the peripheral visual fields is essential for sensory input in our day-to-day activity, for example, for driving and “navigating” generally [386, 387]. In view of that, to assess the visual function disability produced by CRAO, it is important to realize that persisting peripheral fields, even in the presence of central scotoma, do provide the patient fairly useful functional vision for day-to-day living, although he/she cannot read, write, drive, or do any fine work with that eye. This is particularly important if the patient loses the fellow eye. Unfortunately, this important fact is not provided by the evaluation of only the visual acuity. This is not fully realized by the majority in the ophthalmic community. Some may argue that currently automated perimetry is the method of choice to evaluate visual field defects, and not kinetic perimetry. Both types of perimetry have their advantages and disadvantages, discussed elsewhere [387]. Automated static threshold perimetry (Humphrey 30–2 or 24–2 SITA) provides information only up to about 24–30° in the periphery. Kinetic perimetry, by contrast, provides peripheral visual field information all the way to about 80–90° temporally, 70° inferiorly, 60–70° nasally, and 50–60° superiorly. In CRAO, to evaluate the functional disability produced by the visual field defect, it is essential to obtain information about the peripheral visual field loss. Kinetic perimetry, not automated perimetry, provides that information (see Figs. 12.1 and 12.2 in Chap. 12).
5.
Apparent Visual Acuity Improvement May Be Due to Eccentric Fixation
When evaluating visual acuity improvement, one has to keep this flaw in mind. As discussed above, patients with a central scotoma often learn with experience to fixate eccentrically, resulting in an apparent visual acuity improvement, but that does not represent a genuine improvement in the retinal function. Obviously, this important artifact can produce a misleading apparent visual improvement following a therapy. In my studies on various types of ocular vascular occlusive disorders, I have found that, for genuine visual acuity improvement, there must be simultaneous improvement in both the central scotoma and visual acuity [19, 381–385].
Factors Which Influence the Visual Outcome in CRAO
The devastating visual loss with CRAO is one of the most important topics in ophthalmology. Therefore, we need an in-depth understanding of the fundamental scientific knowledge behind the factors that play crucial roles in determining the visual outcome in CRAO, to comprehend the etiology, clinical features, and management of CRAO. My experimental [3, 8] and clinical [6, 15–19] studies on CRAO have provided important information on these factors (discussed at length elsewhere) [19]. These include the following:
1.
Site of Occlusion in the Central Retinal Artery in CRAO
It is generally believed that the site of occlusion in CRAO is invariably at the level of the lamina cribrosa (as revealed by histopathological studies of enucleated eyes). A detailed anatomical study of 100 human central retinal arteries showed that the narrowest lumen of the artery is where it pierces the dura mater of the optic nerve sheath (Fig. 13.16) [388, 389]. Therefore, in cases of CRAO due to embolism, the chance of an embolus getting impacted at this site is much higher than at any other site in the artery. In contrast to that, the site of occlusion in thrombosis of the central retinal artery is at the lamina cribrosa, as shown by histopathological studies. The site of occlusion is an important factor in determining the amount of residual retinal circulation in CRAO, discussed below. When the site of occlusion is in the dural sheath, multiple anastomoses by all the pial and intraneural collaterals of the central retinal artery [388, 390] distal to the occlusion site are left intact (Fig. 13.17), and they play a major role in determining the amount of residual retinal circulation in CRAO [6, 13, 19]. Also in this type of CRAO, fluorescein angiography shows the central retinal artery trunk slowly filling from within the optic nerve (Figs. 13.18 and 13.19). By contrast, when the site of occlusion is at the lamina cribrosa, no collaterals are available to establish retinal circulation (Fig. 13.17) [388, 390]; or if the central retinal artery has no branches, in these eyes angiography shows only filling of capillaries on the surface layer of the optic disc (Figs. 13.20, 13.21, and 13.22) from deeper posterior ciliary artery circulation (Fig. 13.23), without any filling of the central retinal artery trunk itself. Thus, angiography provides very useful information about the site of occlusion. The site of occlusion is an important factor in determining the amount of residual retinal circulation in CRAO, discussed below.
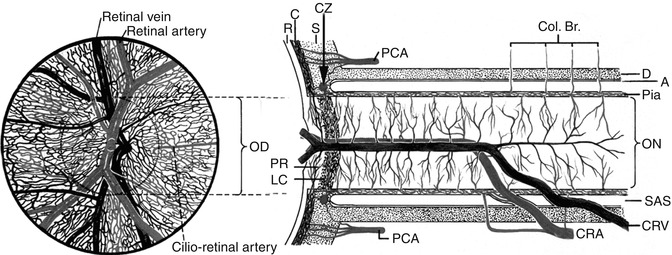
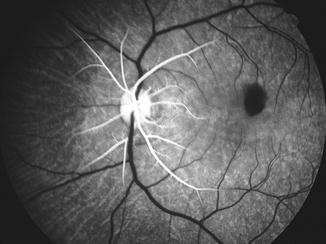
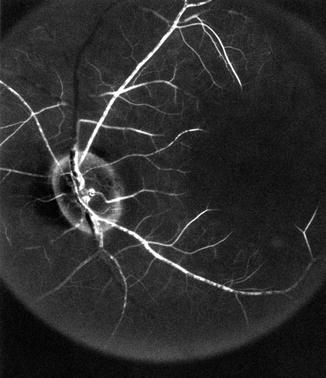
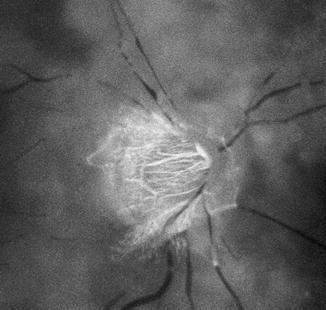
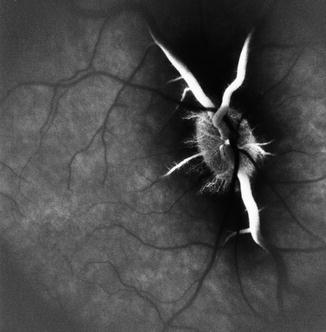
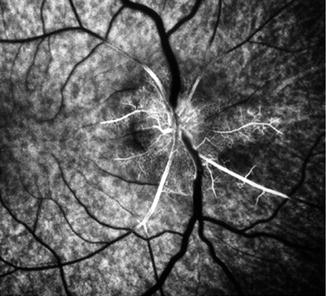

Fig. 13.16
A transverse section of the human optic nerve showing the central retinal artery (CRA) inferiorly lying within the substance of the dural sheath of the optic nerve (ON) (Reproduced from Hayreh [388])

Fig. 13.17
Schematic representation, showing on the right side the course of the central retinal artery and its branches and anastomoses, central retinal vein and its tributaries, and blood supply of the optic nerve. Left figure shows blood vessels on the optic disc and in the retina (Modified from Hayreh [391]). Abbreviations: A arachnoid, C choroid, CRA central retinal artery, Col. Br. collateral branches, CRV central retinal vein, CZ circle of Zinn and Haller, D dura, LC lamina cribrosa, OD optic disc, ON optic nerve, PCA posterior ciliary artery, PR prelaminar region, R retina, S sclera, SAS subarachnoid space

Fig. 13.18
Fluorescein fundus angiogram (14 s after injection of the dye) of the left eye of a rhesus monkey, immediately after experimentally cutting of the central retinal artery at its site of entry into the optic nerve. It shows slow filling of the central retinal artery and retinal arterioles in the posterior part of the fundus in spite of cutting of the central retinal artery (Reproduced from Hayreh [386])

Fig. 13.19
Fluorescein fundus angiogram of the left eye of a rhesus monkey, immediately after experimentally cutting of the central retinal artery at its site of entry into the optic nerve. It shows slow filling of the central retinal artery and retinal arterioles in the posterior part of the fundus in spite of cutting of the central retinal artery

Fig. 13.20
Fluorescein fundus angiogram of the right eye of a patient with CRAO, 29 s after injection of the dye, showing normal filling of the choroid and optic disc vessels (supplied by the posterior ciliary artery circulation), but no filling of the central retinal artery and retinal vasculature at all as yet. The retinal vessels, particularly the veins, show typical “cattle-trucking” or “boxcarring” (Reproduced from Hayreh [386])

Fig. 13.21
Fluorescein fundus angiogram of the right eye of a rhesus monkey with CRAO, showing normal filling of the choroid and optic disc vessels (supplied by the posterior ciliary artery circulation), but very slow filling of the main retinal veins in the peripapillary region but no filling of the central retinal artery

Fig. 13.22
Fluorescein fundus angiogram of the right eye of a rhesus monkey with CRAO, showing normal filling of the choroid and optic disc vessels (supplied by the posterior ciliary artery circulation), but very slow filling of the peripapillary retinal vessels

Fig. 13.23
Schematic representation of blood supply of the optic nerve head. Abbreviations: C choroid, CRA central retinal artery, LC lamina cribrosa, NFL nerve fiber layer, ON optic nerve, P pia, PCA posterior ciliary artery, PLR prelaminar region, R retina, RA retinal arteriole, S sclera
2.
Residual Retinal Circulation in CRAO
Fluorescein fundus angiography in eyes with CRAO initially almost always shows a variable amount of residual retinal circulation, with sluggish filling of the retinal vasculature, and it is rare to see complete absence of retinal vascular filling in these eyes. This has erroneously led to a widespread belief that the artery is usually only partially occluded in CRAO [392, 393], which belief, in turn, is used as a justification for the claims of visual improvement for many advocated therapies, even when therapy is instituted many hours or even days after the onset of CRAO. For example, Watson [394] very well reflected the prevailing view that if there is even minimal retinal circulation after CRAO, it is possible to obtain a considerable recovery of vision in these eyes. To place this misconception in its proper perspective requires answers to the following two important questions:
(a)
Is It True That Occlusion of the Central Retinal Artery Is Almost Always Incomplete in CRAO Eyes with Residual Retinal Circulation? The subject is discussed at length elsewhere [386]. Briefly, my experimental [3–14] studies on CRAO are conclusive on this issue, clearly demonstrating that this assumption is mistaken, for the following reasons. In these experimental studies, CRAO was produced by either clamping or cutting the central retinal artery under direct observation at its site of entry into the optic nerve, by exposing the artery in the orbit. Immediate post-occlusion fluorescein fundus angiography showed a variable amount of slow filling of the retinal circulation in the vast majority of the eyes (Figs. 13.18, 13.19, 13.20, 13.21, and 13.22), in spite of the presence of a cherry-red spot and marked retinal infarction – this clearly indicated massive retinal ischemia [3, 4, 6–8, 11–13]. Similarly, in my studies on patients with CRAO [6, 15, 17, 18], I have found identical slow filling of the retinal circulation in the vast majority of eyes. This definitely proves that, although the central retinal artery is completely occluded in CRAO, there is usually still some (highly variable) residual retinal circulation, as shown by fluorescein angiography. Moreover, my serial angiographic studies in both experimental and clinical CRAO eyes have always shown marked improvement in retinal circulation after several days or weeks [3, 6–8, 11, 12, 15]. The mechanism of this residual retinal circulation with complete occlusion of the central retinal artery is discussed at length elsewhere [6, 13]. If a patent cilioretinal artery of medium or larger size is present, my angiographic studies showed that that appreciably improves the slow filling of the retinal vascular bed (Fig. 13.24).
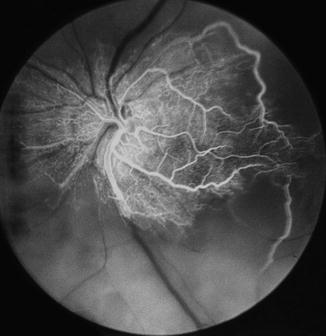

Fig. 13.24
Fluorescein fundus angiogram of the left eye with non-arteritic CRAO with cilioretinal retinal sparing. It shows the area supplied by the cilioretinal artery and retrograde filling the adjacent retinal circulation
(b)
Is It Possible to Obtain a Considerable Recovery of Vision in Eyes with Even a Minimal Residual Retinal Circulation After CRAO? I investigated this issue specifically in 101 eyes of rhesus monkeys in two experimental CRAO studies [3, 8–10]. Both clearly showed that there was no correlation between the residual retinal circulation and recovery of visual function, as judged by electrophysiological and morphologic studies. This is presumably due to very low oxygen tension in the blood of residual retinal circulation, inadequate to maintain retinal viability [3, 8, 9]. Both studies showed that it is almost always the duration of CRAO which is the principal determining factor in the production of irreversible retinal damage, i.e., the longer the ischemia, the more marked the retinal damage. The only exception was in the presence of a large patent cilioretinal artery, where relatively quicker and better filling of the retinal circulation seems to have some protective effect, particularly in the macular retina.
3.
Causes of Occlusion in CRAO
It is generally believed that the CRAO is always either embolic or thrombotic in origin. My studies [17, 19] and others discussed above have shown that embolism is far more common than thrombosis in CRAO, as was pointed out almost a century ago by Coats [395, 396]. Very rarely, vasculitis, trauma, or other conditions may be associated with CRAO (see above).
Giant cell arteritis is an important and a well-known cause of CRAO – an ophthalmic emergency because of its high risk of bilateral visual loss. My detailed studies on GCA during the past 45 years have revealed that when these patients develop CRAO, although ophthalmoscopy reveals a classic picture of CRAO with or without optic disc edema, fluorescein fundus angiography almost invariably discloses combined occlusion of the central retinal artery and one of the posterior ciliary arteries (Fig. 13.6) [22, 23]. This is because the two arteries often arise by a common trunk from the ophthalmic artery (Fig. 13.25) [389, 397]. GCA has a special predilection for involving the posterior ciliary artery. When GCA causes occlusion of the common trunk, both arteries are occluded; that finding is characteristic of arteritic CRAO but extremely rare in NA-CRAO. Therefore, these eyes actually have combined arteritic CRAO and arteritic anterior ischemic optic neuropathy, resulting in a much worse visual loss than in NA-CRAO [19]. Fluorescein angiography is the only way to diagnose this (Fig. 13.6).

Fig. 13.25
An example of a common trunk of origin of the central artery of the retina and posterior ciliary arteries arising from the ophthalmic artery, as seen from below (Reproduced from Hayreh [388]). CAR central artery of the retinal, LPCA lateral posterior ciliary artery, MPCA medical posterior ciliary artery, OA ophthalmic artery, ON optic nerve, PPS point of penetration into the sheath by central retinal artery, * common trunk of origin of CRA and PCA
A fall of perfusion pressure below the critical level in the retinal vascular bed usually causes transient CRAO. Its mechanism is discussed above (see above).
4.
Length of CRAO
This is by far the most important factor. My experimental study of CRAO in elderly, atherosclerotic, hypertensive rhesus monkeys (similar to most patients with CRAO) showed that the retina suffers no detectable damage with CRAO of up to 97 min, but, after that, the longer the CRAO, the more extensive the irreversible damage [3]. The study suggested that CRAO lasting for about 240 min results in massive, irreversible retinal damage [3, 398]. It [3] also showed that, contrary to the prevalent impression, the retina of old, atherosclerotic, hypertensive rhesus monkeys can tolerate ischemia for a much longer time than we found in our exactly identical study in younger, normal monkeys [9, 10]; the reasons for that discrepancy are discussed at length elsewhere [3]. I also found in my experimental CRAO studies that in eyes where the retinal circulation was restored to normal after CRAO of more than 2- but less than 4-h duration, retinal function did not show signs of major improvement until many hours or even a day or more after restoration of circulation – the longer the ischemia, the longer the lag before any improvement of function started. This is highly important finding clinically, because it has unfortunately fostered the mistaken notion that visual recovery can occur in eyes with CRAO lasting for much longer than 4 h; this notion is a highly pervasive in the studies dealing with various advocated treatments for CRAO. However, unless retinal circulation has returned to normal within 4 h, treatment instituted more than 4 h, or even days, after the onset of CRAO cannot claim any scientific rationale for improvement of vision.
5.
Presence and the Area of Supply by a Patent Cilioretinal Artery in CRAO
This can have a major impact on the visual outcome in CRAO, as shown above. Cilioretinal arteries are discussed in Chap. 3. Justice and Lehmann [399], in a fluorescein angiographic study of 2,000 eyes of 1,000 consecutive patients, found one or more cilioretinal arteries in 49.5 % of all patients or in 32.1 % of the eyes, with a great deal of variability in their number and distribution (incidentally, I have seen two patients whose entire retina was supplied by multiple cilioretinal arteries, similar to that I described previously in rhesus monkeys [400]). In CRAO, if the patent cilioretinal artery supplies the foveal region (Figs. 13.2 and 13.24), the visual acuity is almost always normal, with an intact central island visual field corresponding to the distribution of the artery [19, 373, 401–403]. Importantly, when the supply by the cilioretinal artery just touches the foveola, in CRAO initially there is a marked fall of visual acuity, but within 2–3 weeks the visual acuity spontaneously improves markedly (to almost 20/20) which, again, may erroneously be attributed to any treatment currently being administered [386]. However, if it supplies only a small peripapillary region (Figs. 13.3, 13.4, 13.5a, and 13.7a) or retina nasal to the disc (Fig. 13.4), a small island visual field may be present corresponding to the region of supply by the patent cilioretinal artery (Fig. 13.5), with poor visual acuity.
To sum up: all these factors play important roles in the visual outcome in CRAO and require evaluation.
Anterior Segment of the Eye
This shows no abnormality specifically related to the CRAO. In these eyes, initially the IOP often tends to be slightly lower than in the fellow normal eye, similar to what is seen with retinal vein occlusion [404], but, unlike in the latter, the lower pressure does not last long. Iris and angle neovascularization is seen in CRAO eyes associated with ocular ischemic syndrome [17, 18].
Ophthalmoscopic Findings in CRAO
Nettleship [405] in 1891 published a detailed and comprehensive description of the acute ophthalmoscopic appearance of CRAO in the English literature. He stated: “The classical dense, white haze of the central region of the retina with a well-marked clear patch (“cherry red spot”) at the yellow-spot was very well shown; there were no haemorrhages; the arteries and veins were of about the normal size, but no pulsation could be produced in any of them by pressure with the finger upon the globe.” He went on to describe the appearance of the blood vessels: “The second feature of interest …. is the stagnation of the arterial blood stream without diminution in the size of the arterial column. ….. the large size of the vessels appeared to be inconsistent with the conclusion that their supply was cut off. This phenomenon is by no means uncommon in recent cases of embolism or thrombosis of the arteria centralis, and, … is quite compatible with almost, if not quite complete extinction of sight; on the other hand in some cases the arteries are, as is well known, found to be extremely shrunken from the first.” He also gave an excellent diagram of cilioretinal collaterals on the surface of the optic disc seen in a patient 12 months after development of CRAO.
The late fundus findings of CRAO have been described by Lorentzen [406] and Karjalainen [373] in a retrospective review of the records of 37 and 50 CRAO patients, respectively – the authors, however, had not seen the patients during the acute phase.
While the classical ophthalmoscopic findings of acute and late CRAO are well known, there was limited information concerning the prevalence and temporal development of the fundus findings drawn from large, prospective studies, in part due to the predictability of the clinical picture and the rarity of CRAO. I investigated prospectively the incidence, pattern, and evolution of fundus changes in 248 eyes (of 240 consecutive patients) with CRAO [20]. This study provided information on many of these aspects of CRAO that retrospective studies could not. My experimental CRAO studies (101 eyes) [3, 8] in rhesus monkeys also provided useful information, particularly since it is possible to evaluate invasively, serially and systematically, the whole evolution of CRAO – the fundus prior to CRAO, at the onset of CRAO, CRAO lasting for varying lengths of time, and the resultant progression of disease. In these experimental studies, almost all eyes had transient CRAO. Combined information from clinical [20] and experimental [3, 8] studies on CRAO provides a complete picture, discussed below.
As previously discussed, my clinical study [19] on CRAO has shown that in order to make scientifically accurate observations about visual outcome and fundus changes, it is crucial to classify CRAO into its four different types, i.e., permanent CRAO (arteritic and non-arteritic), permanent CRAO with cilioretinal artery sparing, and transient CRAO.
Retinal Changes
The earliest characteristic change in CRAO is the development of retinal opacity, located essentially in the posterior pole, mostly within the temporal arcades, with normal-looking peripheral fundus (Figs. 13.3, 13.4, 13.5a, 13.7a, and 13.8a). One potential misunderstanding arises from the concept that retinal whitening must involve the entire retina to justify the diagnosis of CRAO. The opacity is essentially due to opacification of the retinal ganglion cells caused by acute ischemia and is seen ophthalmoscopically mainly where there is more than one layer of ganglion cells, that is, mainly in the macular region. Since the number of ganglion cells progressively increases toward the center of the macula, the retinal opacity and associated swelling become progressively more marked toward the center of the macula (Figs. 13.1, 13.2, 13.3, 13.4, 13.5a, 13.7a, and 13.8a) [19]. However, since there are no ganglion cells in the foveola and also it is an avascular region, it is not affected by the retinal opacity. Consequently, the normal color of the fundus is still seen through the normal transparent foveolar retina, giving rise to the “cherry-red spot.” I believe that the absence of retinal opacity in the peripheral retina is due to thinner retina with a single layer of ganglion cells, so that nutrition of the inner layers of the retina, along with those of the outer layers, can be maintained by the choroidal vascular supply. That would explain the presence of either normal or varying amounts of peripheral visual fields still present in these eyes [18]. There is almost a universal impression that the “cherry-red spot” is due to the red color of the normal underlying choroidal blood. That is not true. The color of the normal fundus is due to the color of the retinal pigment epithelium, not the blood in the choroid. That is the reason why when African and other races with dark skin color develop CRAO, this spot is actually dark or brown in color instead of “cherry red” – and their blood is the same color as that of whites! (The same is also true of rhesus monkeys.)
In my experimental CRAO primate model, the central retinal artery was occluded temporarily for 15 min to 4½ h [3, 8]. In the primates, at the earliest examination of the fundus, i.e., 7 min after CRAO, the retina in the posterior pole already showed a mild degree of opacity. The severity of retinal opacity progressively increased, so that after CRAO of 60 min it was marked and after 70 min or longer it was of severe degree. The “cherry-red spot” was evident with CRAO of about 20 min or longer. In eyes with short-term transient CRAO, multiple retinal opacities, similar to cotton-wool spots, were the only findings at the posterior pole (Fig. 13.10a). This finding of multiple cotton-wool spots has often resulted in misdiagnosis of CRAO.
Late ophthalmic findings depended upon the duration of CRAO. For example, with transient CRAO of 90 min, the fundus looked normal within 4½ h and remained normal during follow-up (Fig. 13.10). In eyes with transient CRAO for 2¼ h, it took about a day for the retina to look normal. With long-term transient CRAO, the retinal opacity started to resolve in about a week and usually resolved completely within about 3 weeks. Once the retinal opacity resolved, the retina regained its transparency, but histopathology in eyes with long-term CRAO showed permanent damage to the inner retina [3]. When the CRAO lasted for several hours or was permanent, the macular retina looked atrophic. Some eyes developed macular retinal pigment epithelial granularity.
In my clinical study [19], in complete CRAO, the retinal opacity was still present in 17 % of the eyes after as much as 1 month. By contrast, in transient CRAO and CRAO with cilioretinal artery sparing, the opacity had resolved within 30 days; the reason for the briefer period of opacity in these two types of CRAO was that retinal circulation was restored within few hours or so in transient CRAO, and in eyes with cilioretinal artery sparing, the retinal capillary bed was gradually perfused by retrograde blood flow (seen on fluorescein angiography) from the patent cilioretinal artery. Since in the clinical study the follow-up of patients was not as frequent and regular as in the experimental CRAO study, it is not possible to give as precise information as in the experimental CRAO (above).
Retinal Vascular Changes
In experimental transient CRAO [3, 8], soon after onset of CRAO, the appearance of the retinal vessels varied markedly, irrespective of the duration of CRAO, from being extremely attenuated to normal looking. Boxcarring (“cattle-trucking”) was usually seen in eyes with severe retinal opacity although it was sometimes seen even with less marked opacity; boxcarring was usually in both arteries and veins (Figs. 13.4 and 13.6). In boxcarring, the blood column is fragmented and segments of blood column oscillate jerkily, finally becoming stationary, beaded columns. On long-term follow-up, in eyes with transient CRAO of less than 105 min, the retinal vessels remained normal looking at all times; however, in those with CRAO for longer than that, the retinal vessels became attenuated.
In the clinical study, during the initial stages of CRAO, as in the experimental study, the appearance of retinal vessels varied markedly, from being extremely attenuated to normal looking. Both experimental and clinical studies showed that normal-looking retinal vessels do not rule out complete CRAO, as Nettleship [405] also pointed out in 1891 (see above). These data highlight the need for clinical awareness that a normal retinal vascular appearance does not exclude a recent CRAO. Boxcarring, during the first week after onset of CRAO, was seen in both retinal arteries and veins in about 20 % of eyes with complete CRAO; however, unlike experimental animals, human patients are usually not seen immediately after the onset of CRAO or even for several days, and that may explain the low incidence of boxcarring in them. Retinal arteries can develop sheathing, which is of two types. (a) That seen during initial stages (Fig. 13.2): it is rare. (b) That seen later on during follow-up: the estimated probability of developing sheathing later on in complete CRAO was 10 % within 3 weeks after onset and 15 % within 4 months and longer (Fig. 13.26). Attenuation of retinal vessels in CRAO is also of two types: (a) that seen during initial stages of CRAO (Fig. 13.7) and (b) seen later on, during follow-up (Fig. 13.26) – the latter is much more common than the former. Most of the eyes with permanent CRAO showed attenuated retinal arteries months after the incidence, but NOT all; by contrast, most eyes with transient CRAO showed normal retinal arteries, but NOT all. There is no universally consistent pattern; that is typical of normal biological variation. But it raises the problem: how does one classify the CRAO as permanent or transient, when a patient initially presents months after the onset of visual loss with normal-appearing retinal arteries? In such cases, fluorescein angiography does not provide useful information about that; however, the combined information from visual outcome [19] and the fundus findings are nearly always adequate to make that differentiation.