Fig. 14.1
iScience canaloplasty microcatheter with laser-based fiber-optic light. Image courtesy of iScience Interventional Inc.
In most cases, CP can be performed safely under topical anesthesia, although peribulbar or retrobulbar anesthesia may also be used. A superior approach is ideal to allow for coverage of a bleb by the upper lid in case one is formed, and also for less discomfort postoperatively. If a traction suture is used to improve exposure, it should be placed a few clock hours away from the surgical site to avoid obstruction of view into the AC and/or distortion of perilimbal anatomy.
A fornix-based peritomy is created using sharp-tipped Westcott scissors while leaving a small anterior skirt of conjunctiva at the limbus (Fig. 14.2). Blunt dissection is directed posteriorly to allow the posterior edge of the peritomy to be sufficiently relaxed for creation of scleral flap (Fig. 14.3). Location of ciliary veins should be considered when deciding where to perform the dissection. One percent plain Xylocaine is injected under Tenon’s capsule to apply further local anesthesia (Fig. 14.4). Cautery should be performed lightly only as needed, and avoiding these veins and collector channels (Fig. 14.5).
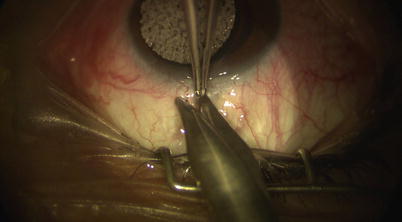
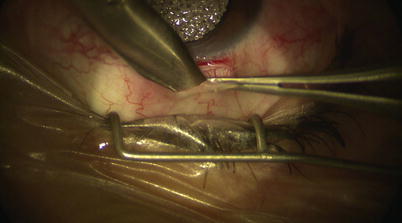
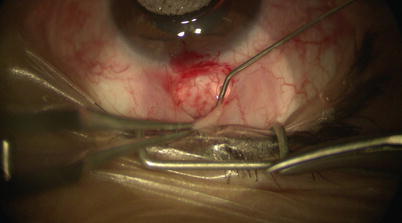
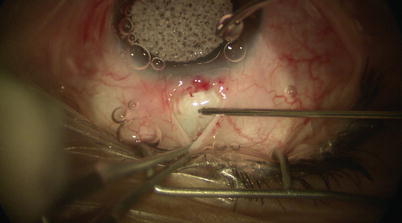

Fig. 14.2
Localized peritomy leaving a small anterior skirt of conjunctiva at the limbus

Fig. 14.3
Blunt dissection posteriorly with Westcott scissors beneath Tenon’s capsule

Fig. 14.4
One percent plain Xylocaine is applied for further local anesthesia

Fig. 14.5
Light cautery is applied only as needed
Given the high accuracy required to create the scleral flaps, diamond blades can be helpful in making incisions and dissections. A 5 mm × 5 mm superficial scleral flap of parabolic shape (which in our experience provides good watertight closure) is outlined using a diamond trifacet blade (Fig. 14.6). The flap is then fashioned with a diamond crescent knife at about one-third scleral thickness (200–300 μm) (Fig. 14.7), dissecting 1–2 mm into clear cornea (Fig. 14.8).
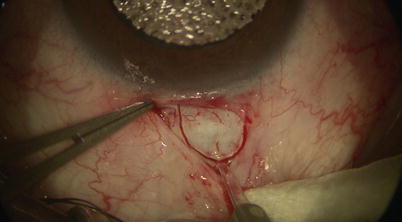
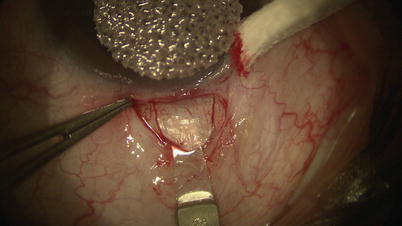
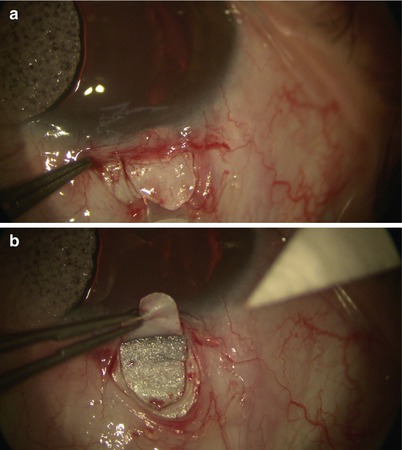

Fig. 14.6
A diamond trifacet blade is used to outline a 5 mm × 5 mm parabolic scleral flap

Fig. 14.7
A diamond crescent blade is used to dissect at a depth of approximately one-third of the full scleral thickness to create the superficial scleral flap

Fig. 14.8
Dissection is carried forward (a) until it reaches the clear cornea (b)
Then under high magnification, again using the diamond trifacet blade, a deep flap is outlined from 1 mm inside the posterior edge of the superficial flap (Fig. 14.9). This deep flap is also fashioned using the diamond crescent blade, but now at 90 % scleral thickness, leaving only about 100 μm of sclera covering the choroid (one may cut down into suprachoroidal space just at the back end of the flap to allow better determination of the required depth) (Fig. 14.10). At the desired depth, you should see irregular scleral fibers with an underlying purple hue coming from the choroid (Fig. 14.11). Sometimes a full-thickness dissection may inadvertently occur, in which case care must be taken to restart a new tissue plane leaving behind a thin layer of scleral bed. If dissection is too deep, then one risks penetration into the choroid or even the vitreous; yet if it is too superficial (more often the case), when dissection reaches the limbus, one may pass right over SC into clear cornea without exposing the canal (where it becomes even more difficult to reestablish a new plane or flap). Creation of a good deep scleral flap exposing SC is thus likely the most technically challenging step of CP. Identification of proper anatomical landmarks is crucial, and this step must be performed under high magnification. This is particularly true when dissection reaches the base of the flap at the limbus, where unintentional penetration into the AC can occur. If that is the case, which would be evident with a gush of aqueous coming from the AC, one can convert to a penetrating procedure (trabeculectomy).
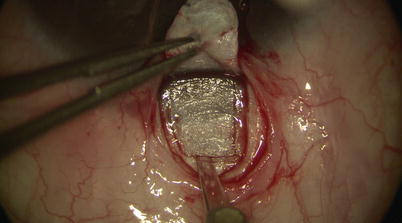
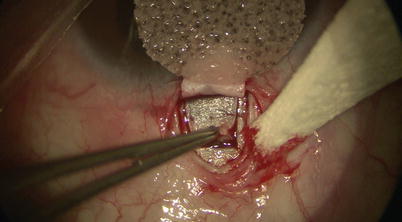
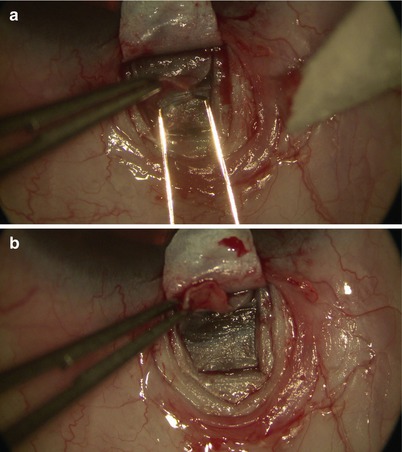

Fig. 14.9
A deep scleral flap is outlined using the diamond trifacet blade

Fig. 14.10
Suprachoroidal space is reached at the posterior edge of the deep scleral flap. This will facilitate identification of the depth required for creation of the deep flap

Fig. 14.11
The diamond crescent blade is used to fashion the deep scleral flap (a). At the desired depth, the thin remaining scleral bed has a light purple hue coming from the underlying choroid (b)
The deep flap is carried anterior enough when one sees scleral fibers changing from random to organized circumferential arrangement parallel to the limbus. At this point the scleral spur is reached. One may then either slowly and carefully dissecting forward to unroof SC (Fig. 14.12) or simply use a pair of toothed forceps to lift the deep flap up to expose fibers of the outer wall of SC at the base of the flap. Good exposure is confirmed when one sees aqueous percolating through SC, or blood reflux from cut ends of SC (Fig. 14.13).
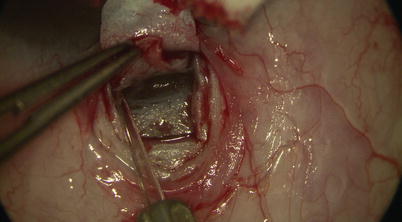
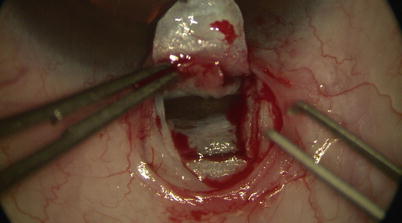

Fig. 14.12
Careful dissection is performed to unroof Schlemm’s canal at the base of the scleral flap

Fig. 14.13
Once Schlemm’s canal is reached, slow percolation of aqueous and reflux hemorrhage from the canal can be visualized
A paracentesis is then made through the clear cornea away from the surgical site (Fig. 14.14). This would allow IOP to drop gradually to almost single digits, and thus preventing bulging of Descemet’s membrane and the inner wall of SC. One should ensure gradual entry and retraction of the blade while making the paracentesis, so as to avoid sudden decompression of the eye which would risk sudden shallowing of the AC and/or intraoperative hemorrhage. This is particularly true if the reason of choosing CP (a non-penetrating procedure) instead of a penetrating procedure was because the eye is prone to such issues.
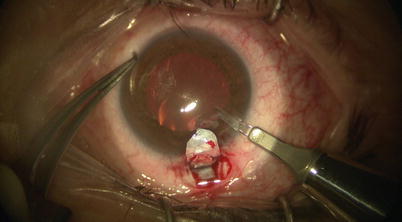

Fig. 14.14
A paracentesis is made with the diamond trifacet blade at least a couple of clock hours away from the surgical site. This will decompress the anterior chamber and prevent bulging of Descemet’s membrane at the base of the scleral flap
The deep flap is advanced forward for another 1 mm into the clear cornea, where further advancement only requires little force as it gets into the plane between Descemet’s membrane and the corneal stroma. The trabeculo-Descemet’s window (TDW) is thereby fashioned. To allow better advancement and exposure of TDW, one may use the diamond trifacet blade to gently create vertical relaxing incisions into the clear cornea at the lateral edges of the flap (with the cutting edge of the blade facing you, thus reducing the chance of penetration into the AC) (Fig. 14.15).
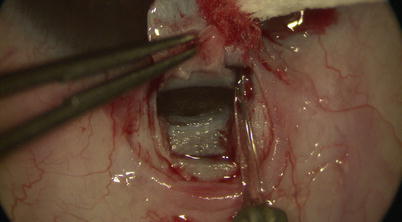

Fig. 14.15
Relaxing incisions are made with the diamond trifacet blade at the lateral edges of the base of the deep scleral flap. One has to be careful to avoid inadvertent penetration through Descemet’s membrane and enter the anterior chamber
One may use a pair of Mermoud forceps to delicately strip the inner wall of SC away to further increase aqueous percolation (Fig. 14.16). Surgical sponges such as Weck-cel (Medtronic, Minneapolis, Minnesota) can be used to carefully push down on Schwalbe’s line and Descemet’s membrane to separate corneal stroma from TDW. However, any excessive downward pressure or sudden movement may easily perforate TDW and cause inadvertent penetration into the AC. Thus, we recommend moistening the tip of the Weck-cel prior to use, as it will reduce the rigidity of the tip when it is placed on the TDW, allowing a “soft touch.” Once a satisfactory TDW is created, the trifacet diamond blade can be used to score the underside of the base of the deep scleral flap (Fig. 14.17). A pair of Vannas scissors is then used to excise the deep flap (Fig. 14.18), allowing formation of scleral lake in its place later on.
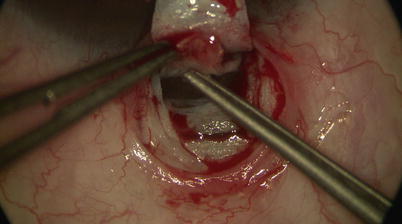
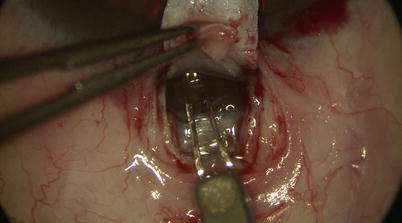
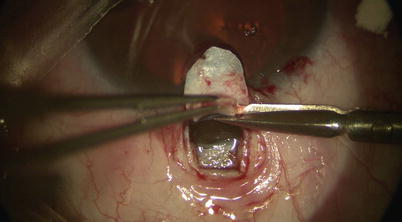

Fig. 14.16
Forceps may be used to gently strip away any overlying scleral fibers and/or the walls of Schlemm’s canal

Fig. 14.17
The diamond trifacet blade is used to mark and score the base of the deep scleral flap (but not the superficial flap)

Fig. 14.18
Vannas scissors are used to excise the deep scleral flap. This will facilitate a space beneath the superficial flap after closure, acting as the intrascleral lake
The two cut ends of SC can now be intubated with a special 150-μm diameter viscocanalostomy cannula (Grieshaber, Switzerland) (Fig. 14.19), through which a cohesive high viscosity viscoelastic such as Healon GV (Advanced Medical Optics Inc., Santa Ana, California) is injected to widen the ends of SC for easier entry of the microcatheter. The iScience microcatheter can be secured to the drape with Steri-strips (3M, St. Paul, Minnesota). Using two non-toothed forceps, the catheter is passed into one of the cut ends of SC and is advanced for 360° until the tip emerges from the other cut end of the canal (Fig. 14.20). During passage, one must pay attention to any resistance or false passage into the suprachoroidal space by observing the blinking red light at the leading tip of the microcatheter. If strong resistance or false passage is encountered, one should stop pushing further, retract, and reattempt with scleral depression at the site of resistance, or simply pull out and try passage in the opposite direction through the other cut end of SC.
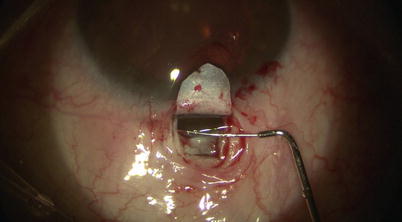
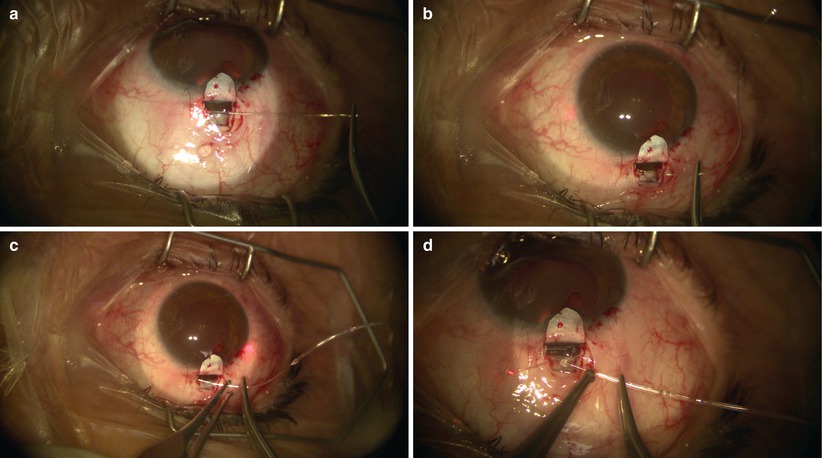

Fig. 14.19
The viscocanalostomy cannula is used to inject cohesive viscoelastic into the cut ends of Schlemm’s canal, thus dilating the entry site for the microcatheter

Fig. 14.20
The microcatheter is inserted into Schlemm’s canal using non-toothed forceps (a). A blinking red light can be visualized behind the sclera, indicating the leading tip of the catheter during passage (b, c). The catheter is passed though Schlemm’s canal for its entire circumference, until the tip emerges from the other end of the canal (d)
Once the catheter has passed 360° and the tip emerges, a 10-0 Prolene suture (polypropylene) with needles cut off is tied around the shaft of the device near its tip (by tying the two loose ends to the loop) (Fig. 14.21). The microcatheter is then withdrawn slowly in the reverse direction to which it was passed (Fig. 14.22); meanwhile viscoelastic is injected into SC using the screw mechanism by a surgical assistant. One must avoid injecting excessive viscoelastic which can cause Descemet’s detachment (approximately 1/8th turn of the screw for every 2 clock hours of passage is recommended). Reflux of blood into the AC can be expected during passage or withdrawal of catheter. Once the entire catheter is removed (Fig. 14.23), the 10-0 Prolene suture is released from the catheter tip using Westcott scissors. By now there should be 2 single 10-0 Prolene sutures in SC with 4 loose ends exposed. After confirming the corresponding ends, each suture is tied to itself with a slip knot (Fig. 14.24) with a back-and-forth movement in SC known as “flossing.” Suture tension is adjusted such that one sees a small indentation of TDW by the suture, which should sit anteriorly in SC – this is confirmed by pulling the knot posteriorly until it is barely able to reach the scleral spur (Fig. 14.25). More tension of the suture would result in more distention of SC, thus theoretically more IOP reduction (Fig. 14.26) [13]. Once satisfactory suture tension is achieved, the knots are locked and trimmed with Vannas scissors (Fig. 14.27).
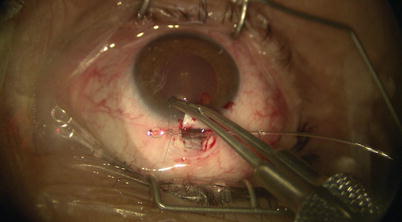
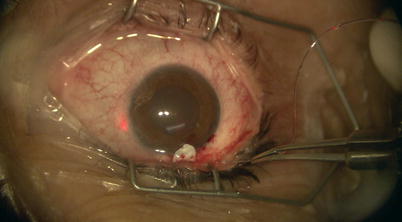
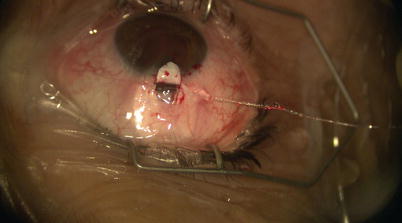
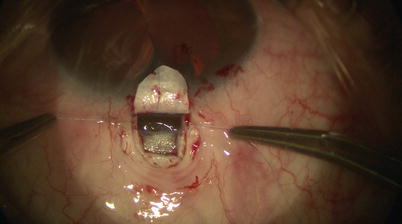
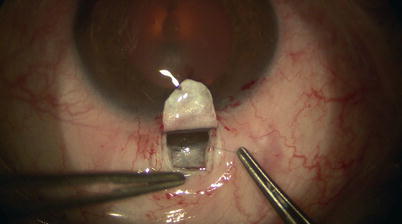
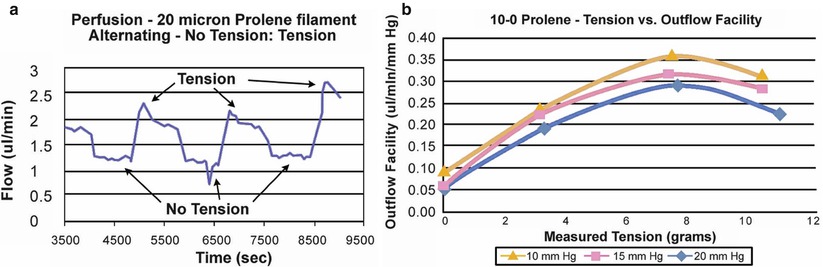
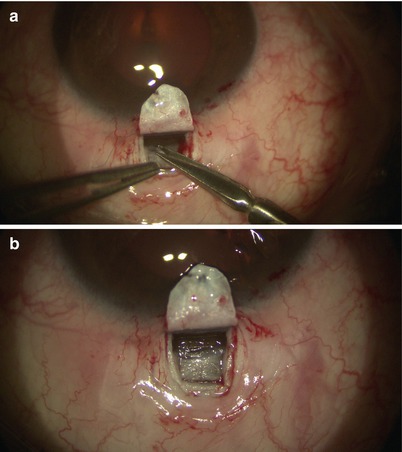

Fig. 14.21
A 10-0 Prolene suture is tied on itself to the tip of the microcatheter

Fig. 14.22
The microcatheter, now with the Prolene suture tied to its tip, is retracted through the entire circumference of Schlemm’s canal. Again the blinking red light indicates the location of the catheter’s tip

Fig. 14.23
The entire microcatheter is externalized, with the 10-0 Prolene suture now remaining within the canal

Fig. 14.24
The corresponding ends of the Prolene sutures are tied with a slip knot that allows adjustment of tension

Fig. 14.25
The desired tension is reached when the knot can barely reach the scleral spur while the sutures are being pulled posteriorly. Slight indentation of the trabeculo-Descemet’s window by the suture knot can also be seen

Fig. 14.26
Outflow facility can be seen as a function of suture tension (a, b) in an ex vivo model. 10, 15 and 20 mmHg correspond to designated levels of IOP. Data from iScience Interventional Inc.

Fig. 14.27
Vannas scissors are used to trim the knots (a). Slight indentation of the trabeculo-Descemet’s window can be seen due to tension from the suture (b)
Watertight closure of the superficial scleral flap is achieved with about five 10-0 nylon sutures placed in simple interrupted fashion. A cohesive high viscosity viscoelastic, such as Healon GV, may be placed under the flap to maintain the scleral lake (Fig. 14.28). It has been suggested that viscoelastics may limit fibrinogen migration [18], thereby minimizing scar formation which may obliterate scleral lake over time. Aqueous will percolate through the TDW and accumulate in this “reservoir” before being absorbed by the episcleral, scleral, and choroidal veins.
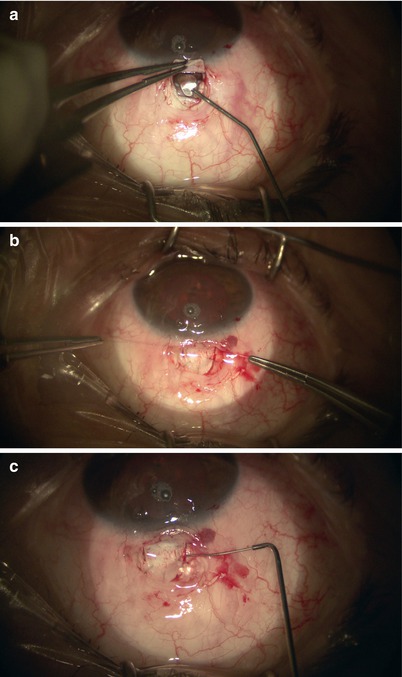

Fig. 14.28
Cohesive viscoelastic is applied beneath the scleral flap to maintain the space for the intrascleral lake (a, c). 10-0 nylon sutures were placed to close the flap (b)
Finally, the conjunctiva is closed in a watertight fashion using 10-0 or 9-0 Vicryl suture in a running horizontal mattress fashion (Fig. 14.29) as in the case of trabeculectomy. The paracentesis and conjunctiva are checked for leaks with a Weck-cel.
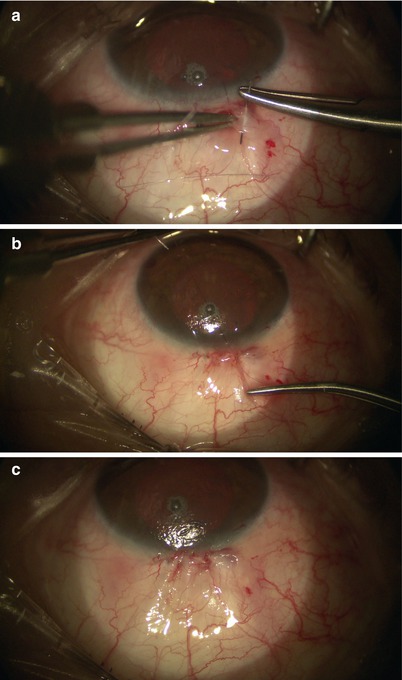

Fig. 14.29
10-0 Vicryl suture is placed in a running horizontal mattress fashion to close the conjunctiva (a–c)
Adjunctive Measures or Alternative Techniques
If cataract surgery is planned as a combined procedure with CP, it should be performed first. After cataract extraction and intraocular lens implantation, the AC should be adequately pressurized with viscoelastic to facilitate scleral flap dissection later. The viscoelastic should be removed after the excision of the deep flap with a 27 gauge cannula on a syringe with balanced saline solution (BSS) manually, rather than with automated irrigation-aspiration, in which case the irrigation part may raise the IOP abruptly causing the TDW to rupture.
Given that the most challenging step in CP is likely the creation of a good deep scleral flap without inadvertent entry into the AC, various alternative approaches to fashion the scleral flap have been described. For deep sclerectomy, Abdelrahman reported the use of a trabeculotome to intubate a segment of SC such that it can be well delineated prior to its unroofing [19]. Others have described using excimer laser [20], erbium:yttrium-aluminum-garnet (Er:YAG) laser [21], CO2 laser [22], and the Fugo blade (Medisurg Ltd., Norristown, Pennsylvania; unpublished data by authors).
Antimetabolites such as mitomycin C (MMC) is typically not used intraoperatively in CP as bleb formation is not a planned outcome. However, it has been reported as an adjunctive agent to prevent collapse of the intrascleral lake [23, 24]. MMC (in 0.25–0.30 mg/cc) can be applied in precut soaked sponges under the superficial scleral flap prior to creation of the deep flap. The conjunctiva is lifted with non-toothed forceps to avoid its contact with antimetabolites (Fig. 14.30), which may increase risk of wound leakage.
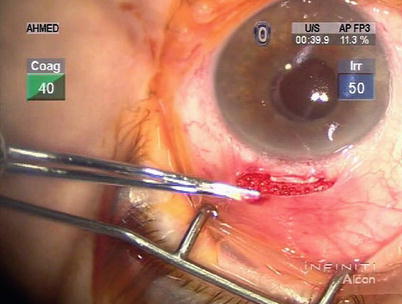

Fig. 14.30
Sponge soaked with mitomycin C is placed under the scleral flap. The conjunctiva is lifted up with non-toothed forceps to avoid contact with antimetabolite
Intraoperative Complications
Flap or conjunctiva-related complications (e.g., partial amputation of superficial flap, buttonhole of conjunctiva) can be managed similar to the case in trabeculectomy. Inadvertent perforation of the TDW is likely the most common intraoperative complication during CP. Occult microperforations may result in trace localized leakage of aqueous with no AC depth compromise and thus may be left alone and potentially even be beneficial to IOP lowering. On the other hand, macroperforations evident with gush of aqueous coming out with concurrent AC shallowing would indicate that CP should be aborted and essentially be converted to a penetrating procedure such as a trabeculectomy.
Postoperative Management
The early postoperative management of CP is quite similar to that of trabeculectomy. A topical antibiotic is used, and steroid drops are administered with a frequent dosing regimen early on (e.g., every 2 h while awake) to minimize risk of scleral lake fibrosis. A miotic agent such as pilocarpine may be prescribed at a low-dosing regimen for the first month after surgery to prevent iris apposition or peripheral anterior synechiae formation against the TDW. However, one has to be careful of its use particularly in phakic patients as it may also move the lens-iris diaphragm forward. Though relatively uncommon (see below), complications such as hyphema, hypotony, and choroidal effusion should be managed as in the case of penetrating procedures. Descemet’s membrane detachment may occur as a complication from ophthalmic viscoelastic device (OVD) injection into SC. In most cases these can be monitored and would resolve spontaneously. However, if it is large enough to compromise vision, it can be drained with a small needle, or neodymium-doped yttrium aluminum garnet (Nd:YAG) laser can be applied to open the descemetocele, with or without air descemetopexy [25, 26]. On the other hand, microhyphema on the first postoperative day had been suggested as a potential positive prognostic factor [27]. In contrast to trabeculectomy, ocular massage is often not recommended as it may result in inadvertent rupture of the TDW.
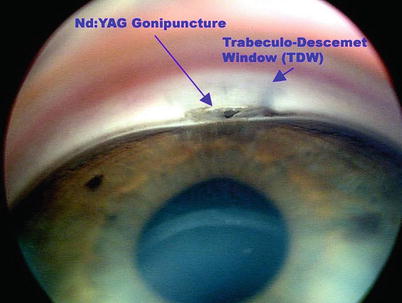
Late rise in IOP is usually due to fibrosis of the microperforations at the TDW and/or fibrosis of the intrascleral lake and episclera. The first approach if IOP rises above target would be laser goniopuncture (GPC), which is required in up to about 20 % of cases (Fig. 14.31) [28–30]. Even though this essentially converts CP into a penetrating procedure, because it is done after some degree of fibrosis has already occurred in the absence of antimetabolite use, the risk of hypotony is lower. GPC is performed with a YAG laser set to free-running Q-switch mode. Starting low at an energy level of around 4 mJ (which can be titrated gradually up to approximately 8 mJ as needed), about 4–8 shots are delivered to create 1–2 holes within the anterior part of the TDW under gonioscopic view. It can be done at any time after about 2 weeks postoperatively and may be repeated as needed. Nonetheless, a prerequisite for the effectiveness of giant papillary conjunctivitis (GPC), is that the TDW must be sufficiently thin and anteriorly dissected (thus further highlighting the importance of accurate dissection during surgery).