Fig. 11.1
(a) New design of the Boston keratoprosthesis. Screw threads have been eliminated and dimensions have been changed. (b) Diagram of the assembled device. The KPro has been incorporated into a corneal graft
With these design modifications came the recommendation of using healthy donor corneal tissue as a carrier, compared to the historical acceptance of any donor tissue, including tissue that was previously frozen or stored in glycerin. Healthy donor corneal tissue has been associated with better coupling of the donor carrier around the neck of the prosthesis and decreased incidence of corneal melts [9–15]. That being said, there have been reports recently of the successful use of eye bank-supplied gamma-irradiated corneal tissue that is essentially devoid of functioning keratocytes and endothelial cells [16].
Surgical complications may be divided into two parts, those associated with the keratoprosthetic device itself and others specific to the inherent morbidity in the surgical eyes requiring this type of surgery:
11.1.1 Complications with the Boston Type I Device
1.
Centration. Although this seems like a simple step, it is not uncommon to obtain significant decentration of the 3.0 mm trephination hole in the middle of the donor graft, especially when the donor, most commonly an 8.5 mm graft, is prepared before the central trephination is made. Several modifications in surgical technique have been suggested to circumvent this problem. An effective technique is to simply place the 3.0 mm opening in the center of the donor corneoscleral tissue before performing the 8.5 mm trephination. The trephination may then be centered around the 3 mm opening. In the event of an eccentrically placed central hole in the donor graft, a similar decentration may be created in the recipient cornea. This allows the positioning of the optical element over the presumed visual axis with appropriate rotation of the keratoprosthesis–donor graft complex within the trephination opening in the recipient cornea.
2.
Device assembly. It is straightforward with few challenges [4]. The donor carrier corneal endothelium must be protected with a viscoelastic device, and care should be given not to unnecessarily damage the endothelium. The posterior plate (PMMA or titanium versions) must be placed correctly (concave side up) onto the threadless stem, which is part of the front plate and constitutes the optical element. The titanium locking ring does not always snap in place easily. It is sometimes too tight for the stem. In that scenario, two heavy needle holders may be used to enlarge the ring circumference slightly to allow for it to snap easily into place. Damage to the device components is unlikely, but dropping one of the components off the sterile field can happen easily during a moment of inattention!
3.
The 8.5 mm posterior plate is used in most instances. This is usually also the size of the carrier donor graft. The opening in the recipient cornea is usually undersized at 8.0 mm, making the introduction of the KPro–donor corneal complex into the opening a small challenge that can be easily overcome. If, however, the surgeon chooses a smaller trephination opening in the recipient, then consideration should be given to the use of the smaller 7.0 mm (“pediatric”) backplate.
4.
An adhesive patch is usually used to stabilize the front plate during assembly. This may leave a gummy residue on the front plate that must be removed after the KPro–corneal carrier complex is sutured into position. The best way to achieve that is with a squeegee posterior capsular polishing cannula.
11.1.2 Complications with the Recipient Eye
1.
Many eyes requiring a permanent keratoprosthesis device have corneal neovascularization, frequently 360° around. Care should be given to minimize/eliminate bleeding into the eye that may delay visual rehabilitation unnecessarily and act as a nidus for intraocular inflammation. Light cautery is very effective in this regard.
2.
The line of sight behind the optical element must be free of any obstruction. After the placement of an iridectomy, an iridoplasty may be necessary to position the pupil behind the optical element [17]. Do not sacrifice any iris tissue unnecessarily!
3.
A plan must be in place for the management of the crystalline lens or IOL. Since the keratoprosthesis comes in pseudophakic and aphakic versions (the aphakic KPro’s power is chosen based on axial length), the surgeon must have a strategy on removal of the crystalline lens with appropriate IOL insertion (including the option of a plano IOL), retention of the IOL, or IOL exchange. Remember: The chosen KPro has already been assembled around the carrier tissue! The surgeon must also be familiar with vitrectomy techniques to address any unwanted vitreous especially in the proximity of a tube shunt.
4.
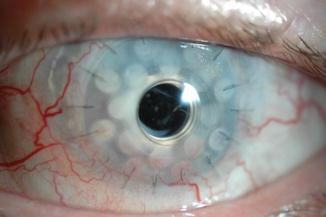
While suturing the KPro–donor corneal complex in place with 9-0 or 10-0 suture, the optical element MUST be covered to protect the macula from microscope light damage (Figs. 11.2 and 11.3).
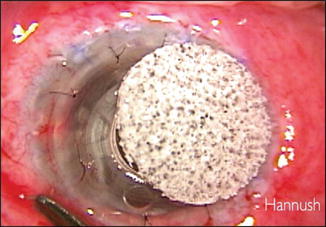
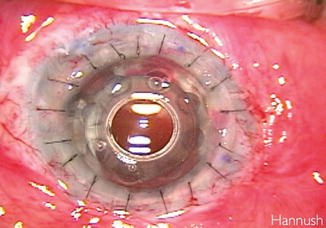

Fig. 11.2
The keratoprosthesis optical element covered by a light shield to protect the macula from the microscope light during the suturing of the graft

Fig. 11.3
The Boston keratoprosthesis (KPro) imbedded in the full-thickness corneal graft at the end of the procedure
11.2 Sterile Keratolysis
After Boston type 1 keratoprosthesis implantation, dellen formation, epithelial defects, stromal thinning, melt, and tissue recession from the edge of the front plate may occur, leading in some cases to exposure of the backplate, aqueous leaks, or extrusion of the device (Fig. 11.4). With previous models of the Boston keratoprosthesis featuring solid backplates, the incidence of corneal melt after implantation was over 50 % (1). The realization that nutritional support from the aqueous humor was important for survival of the carrier corneal graft led to the development of newer models that incorporated multiple holes to the backplate design. This simple device modification, together with the use of a bandage contact lens to prevent ocular surface desiccation postoperatively, significantly decreased the occurrence of corneal melt to a now overall reported incidence of 10–18 % [10, 18, 19].
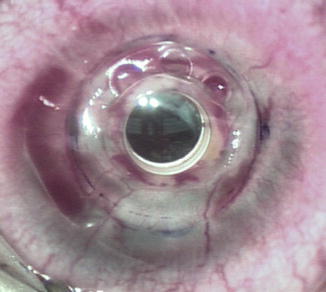

Fig. 11.4
Boston type 1 keratoprosthesis with corneal melt and device extrusion
The use of a bandage soft contact lens was introduced several years ago in replacement of the conjunctival flap previously performed to promote ocular surface stability. The junction between the front plate and the carrier graft is a vulnerable area to evaporative forces with risk of dellen formation, retarded epithelial healing, and even corneal ulceration. The use of a therapeutic well-fitted soft contact lens has proven effective in maintaining adequate lubrication to this area and therefore decreasing complications secondary to tissue dehydration [20, 21]
Despite improvements in device design and postoperative management, patients with a history of inflammatory disease especially those with Stevens–Johnson syndrome/toxic epidermal necrolysis syndrome (SJS/TENS) and mucus membrane pemphigoid (MMP) still have a poorer prognosis [22]. In this group the incidence of tissue necrosis and retraction surrounding the stem is higher and is thought to be triggered by a chronic inflammatory response that could possibly be exacerbated by contact with the keratoprosthesis materials [23, 24]. In addition, their compromised ocular surface allows increased microbial activity also contributing to persistent inflammation and corneal necrosis [24]. Aggressive treatment of the underlying autoimmune disease is likely the key to better outcomes in these difficult patients.
The presence of a persistent epithelial defect (PED) is a known risk factor for the development of keratolysis in general and is associated with a higher risk of sterile corneal necrosis and infectious keratitis in patients with implanted Boston type I keratoprosthesis [23, 25]. Although limbal stem cell deficiency (LSCD) may predispose to epithelial breakdowns, an isolated factor has not been found to be associated with a higher incidence of keratolysis in the absence of underlying inflammatory disease [23], Fig. 11.5.
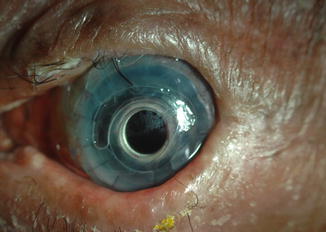

Fig. 11.5
Slit-lamp photograph of a Boston 1 keratoprosthesis showing corneal melt and exposure of the backplate underneath the contact lens in the supero-nasal quadrant
Similarly, the presence of eyelid abnormalities is often a cause of surface exposure and difficulty retaining the bandage contact lens that may lead to PED and keratolysis [21]. Therefore, surgical correction of eyelid incongruities and misalignments is recommended in selected patients to aid in contact lens retention and reduce surface exposure [26].
Sterile keratolysis also occurs more frequently in patients with clinical evidence of retroprosthetic membrane (RPM). Due to corneal opacification in a large number of keratoprosthesis patients, only presence of a retro-optic RPM may be observed clinically at the slit lamp. In cases of a clear cornea, the presence of RPM may be observed through the backplate holes, which are sometimes filled with tissue. However, the thickness of this membrane cannot be estimated accurately on slit-lamp examination. Anterior segment optical coherence tomography (AS-OCT) can be used for further characterization of retro-backplate RPM. Evidence shows that patients with retro-backplate RPM are three times more likely to develop corneal melt and that retro-backplate RPM thickness correlates with the risk of melt [27]. The mechanism by which RPMs are associated with corneal necrosis could resemble that of the solid backplate in years past, by acting as a physical barrier between the cornea and the aqueous humor. Thus, the presence of this retro-keratoprosthesis fibrous tissue can, in some cases, limit the contact between the aqueous and the corneal graft enough that stromal necrosis and epithelial degeneration ensue. In our experience, keratolysis associated with RPM is characteristically seen around the stem of the keratoprosthesis, which is the most distant site from other sources of nutrition such as limbal vessels and, in general, is not associated with an aqueous leak or an inflamed eye. We recommend the use of AS-OCT as an adjunctive tool to diagnose retro-backplate RPMs and identify patients that may be at increased risk for developing keratolysis (Fig. 11.6).
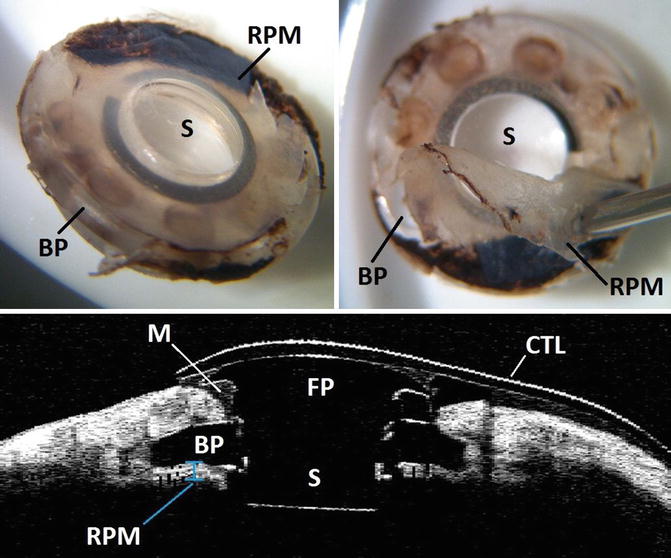

Fig. 11.6
Explanted Boston Keratoprosthesis showing thick retroprosthetic membrane covering the backplate holes (top right and left). Anterior segment optical coherence tomography of the same patient prior to explantation showing corneal melt surrounding the stem and a thick retro-backplate membrane (bottom). BP backplate, M corneal melt, RPM retroprosthetic membrane, S:stem, CTL contact lens, FP front plate
When detected early, sometimes identification and treatment of underlying mechanisms may stop the necrotic process. Initial management may involve optimization of the tear film, professional fitting of the bandage contact lens to improve lens retention, and lateral tarsorrhaphy. Agents with anticollagenase activity such as medroxyprogesterone and oral tetracycline can be tried [28]. In patients with autoimmune disease, systemic immunosuppression to better control the underlying disease might be needed.
Different surgical alternatives to reinforce the ocular surface have been tried with various successes including conjunctival flap, buccal mucosal graft, donor corneal lamellar graft, or crescenteric amniotic membrane graft [28, 29]. In the presence of an aqueous leak, cyanoacrylate glue may also be used as a temporary measure. However, if the necrosis is full thickness and there is exposure of the keratoprosthesis backplate, replacement of the keratoprosthesis is recommended. Even in the absence of an aqueous leak, treatment should not be delayed to decrease the risk of epithelial downgrowth (Fig. 11.7).
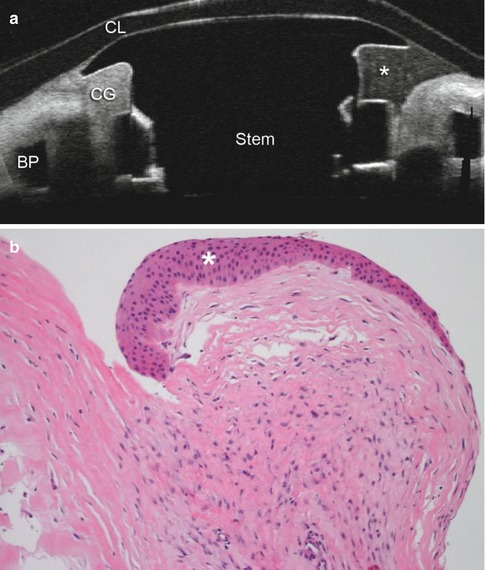

Fig. 11.7
(a) Anterior segment optical coherence tomography of a patient with Boston type 1 keratoprosthesis and corneal melt 15 months after implantation. BP backplate, CL contact lens, CG corneal graft, * area of corneal melt. (b) Histopathology of the retroprosthetic membrane extracted from the explanted keratoprosthesis in the same patient. There are fragments of fibrovascular tissue, lens remnants, iris tissue, and basement membrane with evidence of epithelial downgrowth. There is little inflammation present. * epithelial downgrowth
If adequately treated, most patients fair well, and despite causing morbidity during the postoperative period, keratolysis does not always lead to significant vision loss [19]. However, 25 % of patients with a history of graft stromal necrosis have a recurrent episode, stressing the importance of indentifying and addressing possible risk factors.
Key Points
The mechanisms involved in the development of sterile keratolysis are likely multifactorial and may differ from patient to patient depending on their underlying disease process and comorbidities.
Although more common in patients with inflammatory disease, corneal melt occurs even in noninflammatory conditions.
Retro-backplate RPM thickness correlates with risk of corneal melt.
PED and poor bandage contact lens retention are also associated with higher risk of corneal melt.
Treatment usually requires replacement of the KPro and modification of underlying risk factors.
11.3 Retroprosthetic Membranes
Retroprosthetic membranes (RPM) are the most common complication after Boston KPro surgery, occurring in 25–64 % patients in a 1-year follow-up [30, 31]. According to the multicenter study group results (265 cases), the overall RPM formation rate is 31.7 % (mean follow-up: 17.8 ± 14.9 months) [30]. Most cases of RPM develop within the first 6 months postoperatively, being the mean time to development of 104–216 days postoperatively [30, 31], depending on the series, with a wide range.
These fibrous membranes originate from activated host stromal cornea cells that migrate through gaps in the posterior graft–host junction [32]. They are supposed to be a reliable measure of the degree of postoperative inflammation [7].
Histologically, we can find three layers in them: The first of them consists of stromal fibroblasts, from the host cornea, behind the backplate. Sometimes it is a fibrovascular layer. The second layer is the stromal iris joined to the posterior surface of the membrane. The third is composed by metaplasia of the lens epithelium [32].
Many risk factors for the development of RPM have been advocated, but only few of them have been statistically confirmed. Statistic studies are quite difficult in KPro cases due to many different concomitant factors that appear in these patients, making univariate analysis frequently cannot be proven by multivariate analysis.
RPM formation seems to be more prevalent in individual with chronic inflammation such as autoimmune diseases and uveitis [33].
Infectious keratitis [14] has been demonstrated as a risk factor to develop a higher rate of RPM and a faster development of them, even by using multivariate tests.
Conversely, chemical injury seems to show a non-statistically significant trend to lower rate of RPM and longer time to develop them, not confirmed when controlling other possible predictors of RPM development [30]. However, according to other authors, chemical burns have higher incidence of RPM development [7].
When we use intracameral steroids, no difference in the rate of RPM is seen, but it seems to take statistically longer to be developed [30].
Aniridia [30] is also an independent factor that increases the rate of RPM.
The titanium backplate has demonstrated a much lower rate of RPM development, diminishing from 41.8 % (6-month follow-up period) when PMMA backplate is used to 13 % (6-month follow-up) when 8.5 mm diameter titanium backplate is used [7]. This rate is 0 % (>6-month follow-up) when a 9.5 mm diameter titanium backplate is used [34]. Titanium has a high tissue tolerance, and very thin and strong backplates can be manufactured (edge thickness of 0.25 mm, the fourth part when compared to PMMA). Even more, in vitro cornea cells have increased cell proliferation and decreased cell death when kept in contact with titanium compared to PMMA [7].
It is not clear if RPM is a risk factor for glaucoma development.
Treatment for RPM is diverse. Some of them do not interfere visual acuity so much, so only observation is needed [31]. If we focus only on the cases that need intervention, 90 % of the cases are solved by using YAG laser and 10 % need surgical membranectomy. “Can-opener” approach with 1.2 mJul peak power has been described as safe with no damage in the optic [33]. In our experience, “Cross” Nd:YAG approach with less than 2.0 mJul peak (usually less than 20 peaks) power achieves good results, Fig. 11.8. More than 2.0 mJul peak power can produce pits in the optic of the KPro, Fig. 11.9. Even more, we do not use the Abraham lens for capsulotomy, although many surgeons do. YAG laser produces free radicals and mediators that could increase inflammation inside the eye [33]. In fact, we found high rate of cystic macular edema after Nd:YAG laser in KPros, so we usually use concomitant peribulbar triamcinolone and topical steroids and NSAIDs tid for a week.
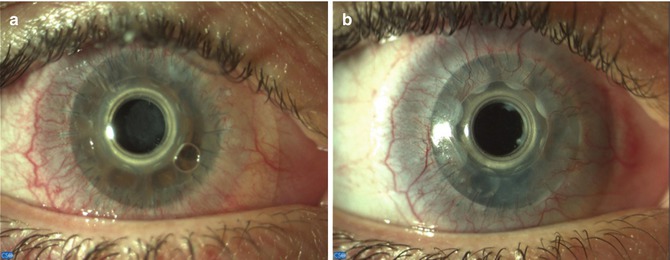
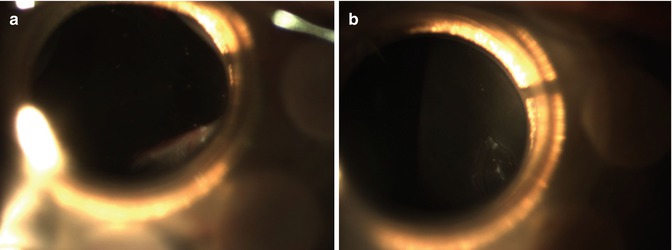

Fig. 11.8
(a) RPM 2 months after Boston KPro type I surgery in a chemical burn case (surgery performed 1 year after chemical burn). VA diminished to 20/40. (b) After Nd:YAG laser “cross” approach, VA improved to 20/20

Fig. 11.9
Fibrovascular RPM reduced VA to 20/400 in case (b). After Nd:YAG laser, VA came back to 20/200. YAG peak power was 2.4 mJul. Some pits in the optic that do not affect VA appear
When surgical RPM excision is needed, surgery is quite easy for aphakic KPro but could not be so easy for pseudophakic KPro due to RPM which is located in a virtual space, not always easy to be surgically found, between the backplate of the KPro and the intraocular lens.
It has been suggested that RPMs seem to be related to melting of the cornea [35] and probable subsequent extrusion of the KPro. A proposed mechanism for this point is that the presence of RPM impedes nutrient diffusion into de carrier cornea by occluding backplane holes. A recent study [36] has shown that 66.7 % melting cases had previous RPM but only 18 % non-melting cases had previous RPM. Furthermore, it seems to be a trend toward thicker RPMs on average, measured by OCT, among eyes with melt. In order to control corneal collagenolysis, the appearance of RPM could lead us to increase the dose of oral tetracyclines and/or topical medroxyprogesterone. YAG laser in the fibrous plugs of backplate holes has also been described to improve nutrition of the carrier cornea [36].
11.4 Infectious Keratitis
Infectious keratitis is a serious complication that can lead to stromal melting with aqueous leak and keratoprosthesis extrusion. Also, surface organisms can easily track into the eye along the optic stem, via the potential space between the KPro anterior plate and corneal stroma, resulting in endophthalmitis. For this reason, an urgent intervention is needed to take the appropriate diagnostic and therapeutic measures, and a detailed slit-lamp examination may reveal the need for vitreous cultures in addition to corneal scrapings.
The rate of infectious keratitis varies from 7 to 17 % [14, 23, 25, 37] in different series with a significant number of cases and follow-up. In our published series [38], we did not have any severe case of infectious keratitis. Out of these series, we have had two other cases which are shown in Figs. 11.10, 11.11, and 11.12, and 11.13. During the 1990s, most of the infectious keratitis and endophthalmitis in patients with Boston keratoprosthesis were caused by Gram-positive bacteria (Staphylococcus and Streptococcus). Following the introduction of topical vancomycin in the postoperative treatment, the infections by these germs have been significantly reduced, and the percentage of infections by Gram-negative bacteria and fungi has increased. In the last decade, the maintained use of a soft contact lens (which protects the ocular surface from drying) and the postoperative treatment with broad-spectrum antibiotics have improved results with Boston keratoprosthesis. However, these factors associated with prolonged use of corticosteroids have increased ocular surface colonization and corneal infection by fungi.
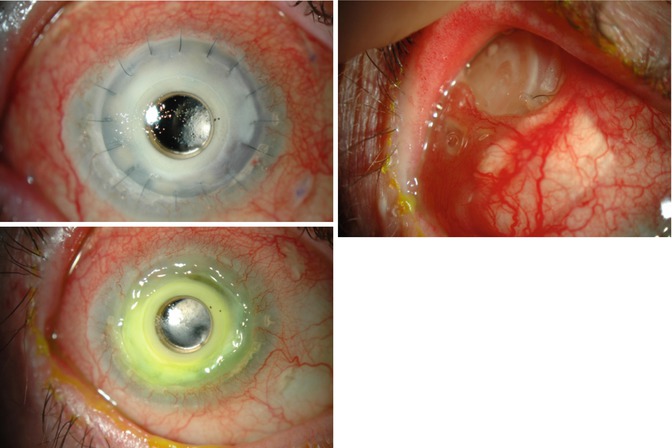

Figs. 11.10, 11.11, and 11.12
Pseudomonas keratitis (possibly initiated by an infection on the Ahmed valve). The valve and the KPro were exchanged

Fig. 11.13
Fungal Keratitis 7 years after KPro surgery. Under control with chronic topical voriconazole
Risk factors for infectious keratitis and endophthalmitis include a diagnosis of cicatrizing conjunctivitis (Stevens–Johnson syndrome, ocular cicatricial pemphigoid, or chemical injury) and a history of persistent epithelial defect [25, 39, 40]. While some authors found no increased risk of infectious keratitis with topical vancomycin, contact lens use, or systemic immunosuppression [25], others have associated the use of topical vancomycin and contact lens with an increased incidence of fungal keratitis [41].
Although most patients with infectious keratitis have symptoms (decreased vision or pain), some patients have no complaints. For this reason the importance of frequent follow-up must be emphasized to all patients. The most common presenting sign is a corneal infiltrate (90 % of cases), sometimes adjacent to the optic stem. For diagnosis, a corneal scraping of the infiltrates for Gram stain and bacterial and fungal cultures must be done. The rate of positive culture using standard methods varies from 40 to 70 %, and PCR techniques have also been recommended. The initial management includes the increased frequency of broad-spectrum antibiotics. When fungal infection is suspected or in cases with infiltrate around the stem, with risk of intraocular involvement, an antifungal topical treatment is also recommended (usually amphotericin 0.15 % or voriconazole 1 %). Later, treatment will be revised according to laboratory sensitivity results.
Currently, there is no consensus about the best postoperative prophylactic treatment to reduce the rate of infectious keratitis. Although contact lens use is considered a risk factor for fungal colonization and infection, its use is still recommended because therapeutic contact lens is essential for ocular surface hydration and the prevention of stromal melt and necrosis in KPro patients [41]. Topical vancomycin (14 mg/ml) and a fourth-generation fluoroquinolone is, probably, the most used combination for antibiotic prophylaxis. Some authors use both antibiotics indefinitely. Others prefer to stop vancomycin several weeks after surgery and only sustain vancomycin in monocular patients or those with underlying autoimmune conditions [42]. The low rate of fungal keratitis does not justify chronic use of antifungal drugs. Although it has been suggested by some authors, fungal infections cannot be predicted by obtaining periodic surveillance cultures of the peri-KPro ocular surface in uninfected eyes (fungal colonization fluctuates spontaneously over time and a positive culture is not predictive of subsequent infection) [41]. However, if there is visible fungal colonization on the KPro surface or contact lens, an aggressive medical management to prevent subsequent infection is recommended (cultures of the contact lens and KPro and topical treatment with amphotericin or voriconazole four times daily for several weeks). Heavy colonization may appear as tiny, white, mulberry-shaped precipitates on the keratoprosthesis surface or contact lens [41].
11.5 Glaucoma Surgery-Related Complications
The number of Boston keratoprosthesis (KPro) implants performed throughout the world has increased exponentially during the last years. The reasons for this increase are changes in the KPro design that decrease the risk of corneal necrosis and the chronic postoperatory use of topical antibiotics and corticosteroids that have reduced the number of endophthalmitis after this procedure.
Despite dramatic initial vision improvement following KPro, ocular hypertension (OHT) and secondary glaucoma or previous glaucoma progression are some of the most frequent and devastating complications, as they may cause, in many of these patients, severe and irreversible visual loss.
Glaucoma coexists in 36–75 % of the candidates for a KPro implantation and develops “de novo” in 2–28 % of the implanted patients [9–11, 19, 31, 37, 38, 43].
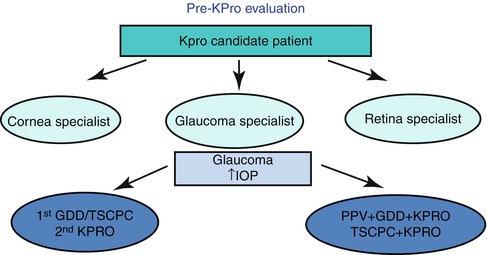
In order to avoid this irreversible complication, a multidisciplinary approach should be made among the cornea, glaucoma, and retina specialists (Fig. 11.1).