Fig. 13.1
Kontur© methafilcon A hydrogel bandage lens over KPro Type I
Base curve | 6.8–9.8 mm |
Diameter | 12.0–24.0 mm |
Sphere power | +10.00 to −20.00 D |
Toric power | −0.75 to −5.00 D |
Although their use is somewhat debatable, we have occasionally used frequent replacement silicone hydrogel material lenses as bandages. These lenses have higher oxygen permeability than Kontur© lenses, are less expensive, are available in boxes of six lenses, and are useful for incorporating power changes. However, these lenses are prone to develop significant deposits and are only available in limited parameter. Our current silicone hydrogel lens of choice is the Air Optix Night and Day contact lens (Alcon, Ft Worth, TX). It is available in a single small diameter (13.8 mm), two base curves (8.4 and 8.6 mm), and a wide range of powers (+6.00 to −10.00 D). This lens is FDA approved for extended wear up to 30 nights.
13.3 The Lens Fitting Process
The fitting process is very similar to a conventional soft contact lens fitting in a normal eye. The initial lens placed after surgery acts as the patient’s first trial lens. During evaluation of the lens, it is important to have proper lens centration and complete corneal coverage. Decentered lenses or lenses that have a small diameter may expose corneal tissue and increase the risk of dellen formation or corneal melt. It is also important the lens is not fit too flat, showing signs of edge fluting, or too tight, with signs of air bubbles or vascular compression (Fig. 13.2). Lens movement seems to be less important than with traditional lens fits, yet, the lens should be moveable by manual push-up. If the lens shows signs of central folding or buckling, this may be an indicator that the lens is too steep or the diameter is too large. In these cases, the fornices may exert forces on the lens causing it to pop out or wrinkle, disrupting the visual quality.
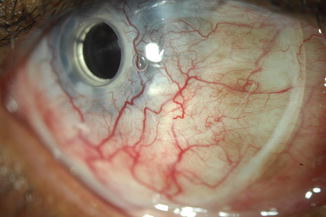

Fig. 13.2
Vascular compression or impingement from a tight-fitting bandage lens
13.4 Power Evaluation
It is important to verify that the contact lens has the best proper correction by measuring visual acuity and performing an over-refraction over the contact lens at each visit. Although the KPro device is chosen specifically for each patient with the expected aphakic or phakic power, unexpected refractive errors or fluctuations may occur and a new soft lens is less expensive than frequent replacement of spectacles [3]. The majority of patients will have a small amount of residual spherical refractive error that can be incorporated into replacement contact lenses.
13.5 Lens Wear and Replacement Schedule
Patients are instructed to wear their bandage lens continuously and not to handle the lens. It is recommended that the contact lens be removed at each follow-up visit for cleaning and disinfection. A daily cleaner for soft contact lenses is recommended; however, these cleaners are becoming increasingly more difficult to find. With the discontinuation of the Miraflow Daily cleaner, a generic version available at CVS Pharmacies called CVS Extra Strength Daily Cleaner combined with a saline rinse can be used for surface cleaning and debris removal [4]. The frequency of lens removal and replacement varies greatly from clinician to clinician. It is not uncommon for a lens to be replaced only when it falls out or when there are signs of debris accumulation [6]. Other clinicians see KPro patients monthly for bandage contact lens planned replacement. A report by Wilhelmus et al. recommends monthly replacement of therapeutic lenses due to a fungal colonization rate of up to 27 % [7] (Fig. 13.3). We tailor the follow-up schedule to each patient, seeing higher-risk patients with frequent lens loss or heavy deposits more frequently and stable patients at 3-month intervals.
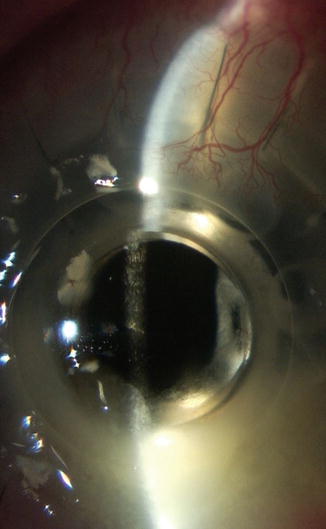

Fig. 13.3
Fungal infection after KPro Type 1 implantation (Courtesy Amy Nau, OD, FAAO)
13.6 Follow-up Schedule and Procedures
Follow-up schedules vary depending on each patient’s unique circumstances. In routine, stable KPro cases, patients are seen by the corneal specialist and contact lens specialist every 3 months. It is rare to go longer than this and common to see patients more frequently when problems occur with the contact lens, KPro, or cornea. As mentioned, some clinicians see their KPro patients monthly. Currently, there is no evidence-based recommendation for recommended follow-up at this time.
13.7 Medications
Topical and oral medications that are used concurrently with a bandage lens wearing KPro patient need to be carefully reviewed. For topical medications, the lens may limit the ocular penetration of the drug by an unknown amount. Additionally, suspensions and oil-based emulsions may interact negatively with the lens material, resulting in reduced optical quality, increased deposits, and an irregular, poor wetting lens surface and ensuing discomfort.
13.8 Culturing of Lenses
There is no current standard of care for culturing the bandage lenses or conjunctiva of patients who have had KPro surgery. Patients are at lifetime risk for infection and complications and require close clinical monitoring for signs of infection. Soft contact lenses, as well as vancomycin and prednisolone use, have been reported as risk factors for fungal infections [1]. A series of 70 surveillance cultures of the ocular or lid surface around the KPro optic in 36 uninfected eyes had four definite and one probable culture; however, surveillance cultures did not predict infection [5]. Though there is no standard of care, some clinicians report culturing the contact lens and conjunctiva. If growth is found, the therapeutic regimen may be adjusted by the cornea specialist.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


