Autograft ossicular reconstruction was first described in 1957 by Hall and Rytzner.1 The technique of using the patient’s own tissue to reconstruct the ossicular chain seemed to the authors to be the most sensible approach to improve success rate and to avoid prosthesis rejection, which was a significant problem with alloplastic prostheses of the time. During the late 1950s and 1960s, the techniques for using a remodeled incus were refined and increasingly adapted to become the most popular method of ossicular reconstruction.2 Over recent years, new alloplastic materials have become more popular because of their simplicity of design, reliability, and increasing stability in a well-reconstructed middle ear, but there are clear advantages of using autologous material, not least because of its ready availability and low cost.
28.2 Patient Selection
The two most common reasons for considering the reconstruction of the ossicular chain is the destruction of the ossicles caused by chronic middle ear disease and disarticulation of the ossicular chain as part of a surgical procedure to eradicate disease. Traumatic fracture of the long process of the incus or stapes superstructure rarely occurs, and, even more rarely, necrosis of the long process of the incus may occur for no clear reason, and always behind an intact and healthy tympanic membrane.3
On most occasions it is possible to retrieve at least the body of the incus to use for reconstruction because it is the long process of the incus, in isolation, that is most susceptible to erosion. The use of a refashioned incus or malleus cannot be guaranteed for a number of reasons that will be discussed later; therefore, it is sensible that the surgeon has other options available before embarking on reconstructive surgery. The decision to use an autograft is made during surgery after inspection of the autologous material at hand and the ossicular defect. The incus remnant might be unsuitable because cholesteatoma has demineralized the bone, leaving a ragged and soft residuum. This finding also raises the concern that recurrent cholesteatoma might occur because of unnoticed cholesteatoma within the bony remnant.4,5 It is difficult to say whether this does occur, but common sense dictates that a badly damaged ossicle is unlikely to serve long-term hearing restoration and, therefore, should not be used.
Ossicular reconstruction should only be considered after eradicating the underlying disease and a plan has been made for repairing the tympanic membrane. Using the patient’s own tissue to reconstruct has the inherent advantage of not being rejected, but that does not mean failure is easily avoided. It is common to operate on an ear that has persistent Eustachian tube dysfunction, and, therefore, retraction of the neotympanum must be considered. Cartilage is often employed to prevent tympanic membrane retraction in the presence of persistent negative middle ear pressure, and its uses are discussed in detail in Chapter 25. Suffice it to say that the surgeon must not only plan to deliver a high probability of hearing success, but also a tympanic membrane that will not retract around the refashioned ossicular chain, leading to recurrent disease or late hearing loss. This is one of the most difficult aspects of autograft ossicular reconstruction. The repositioned incus remnant often has an awkward shape that gets in the way of an effective cartilage tympanoplasty. The cartilage sliver must be tight against the malleus handle and scutum to avoid gaps through which the eardrum can retract.
Most autograft ossicular reconstruction techniques rely on placement of the refashioned incus between the stapes head and malleus handle. Stability and, therefore, success of such a reconstruction is reliant on a number of factors. The first is the dexterity and experience of the operating surgeon. This is not a simple technique. It requires considerable practice using cadaveric material, often after attending instructional courses and working with an experienced trainer.
The second factor is availability of a suitable incus remnant to refashion, taking into account the gap between the stapes head and malleus handle. The simplest reconstruction is one in which there is a relatively small gap between the stapes head and malleus handle and the latter lies more or less over the former. The stability of the reconstruction becomes increasingly compromised if the malleus handle lies more anterior or offset to a posteriorly placed stapes head. The more offset the two points of contact with the incus, the more unstable the reconstruction is likely to be. There are techniques described in Chapter 30 that enable repositioning of the malleus handle to reduce this gap, but surgeons should have a number of potential solutions and, once again, if one is inexperienced, then alternative alloplastic reconstruction techniques should be considered.
The third main factor that dictates success is the extent of ossicular destruction. It is often the case that the disease process only erodes the long process of the incus, but cholesteatoma or eardrum retraction can also erode the stapes superstructure. This leaves a much wider gap between the stapes footplate and malleus handle or tympanic membrane, and it is often impossible to find suitable material to bridge the gap unless a homograft incus or malleus is used.6,7 The incus is rarely long enough because the long process is almost always eroded. Fashioning an ossicle replacement graft out of a cortical bone chip8 has been described, but these are often quite large and produce an unstable arrangement. It is the authors’ experience that these wide gaps have an unacceptable failure rate using autograft material, and alloplastic reconstructive techniques should be considered.
Patient selection, outlined above, is primarily for canal wall-up procedures. Many surgeons advocate canal wall-down surgery, which can result in a narrowed middle ear cleft so that the neotympanum lies close or even touches the stapes head. If the neotympanum is resting directly on the stapes, creating a Type III stapes columella arrangement, then nothing more needs to be done, although it is often sensible to strengthen the eardrum overlying the sinus tympani with a sliver of cartilage. If there is a small gap between the eardrum and stapes head, then cartilage might suffice or, alternatively, an incus remnant or the malleus head can be used as interposition autograft material. These are simple yet robust techniques that should always be considered if the opportunity arises.
28.3 Surgical Technique
In medicine, there are often a number of ways to tackle a problem, and the management of chronic middle ear disease is no exception. No matter which surgical approach is favored by a particular surgeon, it is likely that some form of ossicular reconstruction will be necessary. The authors favor a combined approach tympanoplasty with primary reconstruction of the ossicular chain. This allows a second attempt to reconstruct at second-look surgery if there is still a conductive hearing loss after the first procedure and will ensure that the middle ear cleft is maintained at its normal height.
The reconstructed eardrum sits in a position to enable the secure placement of the prosthesis. If the surgeon favors canal wall-down surgery, then the middle ear cleft is often narrower so that the position of the tympanic membrane is in closer proximity to the stapes head. The offset between the malleus handle and stapes head is magnified, so it often is more preferable to reconstruct from the stapes to the posterior pars tensa of the tympanic membrane. Whether opting for a canal wall-up or -down approach, the surgeon is not always presented with a constant anatomical arrangement. Thus, if autologous graft material is going to be used, then the surgeon will have to be flexible and use the design that best suits that ear.
28.3.1 Ossicular Reconstruction after a Canal Wall-Up Procedure
Stapes Superstructure Present
For the purposes of clarity, primary ossicular reconstruction should only be performed if it is known that macroscopic clearance of the disease has been achieved. There is no point, for example, in reconstructing the ossicular chain if there is concern that there might be residual cholesteatoma within the stapes superstructure. Some surgeons will wait until the second-look operation before reconstructing the ossicular chain, arguing that one can be confident that the procedure is performed in a disease-free ear without evidence of tympanic membrane retraction. If this is the decided course of action, then the incus remnant or malleus head must be stored in the mastoid cavity until it is required.9 On the other hand, successful primary reconstruction in a postoperative stable ear can possibly avoid second-look surgery if there is a facility for non-echo planar magnetic resonance imaging (MRI).
The decision to reconstruct is made toward the end of the procedure once disease has been removed. It is rare not to reconstruct. The authors will only delay or avoid reconstruction if there is concern that the stapes is hypermobile (thus risking dislocation), if the footplate is partially eroded by cholesteatoma matrix, if it is impossible to be certain that all cholesteatoma matrix has been removed from the footplate, or if there is extensive middle ear tympanosclerosis fixing the stapes to its surrounding structures. On occasion there can be concern about persistent Eustachian tube dysfunction evidenced by an atelectatic adhesive otitis media and middle ear effusion. These cases can be particularly unrewarding as, even if it is possible to reconstruct the tympanic membrane using a robust palisade technique, ossicular reconstruction is rarely successful because of persistent middle ear effusion or fibrosis of the middle ear cleft.
Once the decision has been made to reconstruct, careful assessment of the residual ossicular arrangement can be made. The first step is to assess the stapes footplate. The authors always check the mobility of the stapes or whether there has been erosion of its superstructure. Sometimes the head of the stapes can be missing, but the crura are intact or that a crus of stapes has eroded, leaving a precarious arrangement that might require laser removal of the residual superstructure.
The next step is to assess the autologous material that will be used. The authors prefer the incus body over the malleus head because the incus has a thinner profile and so much of the design is already incorporated into its shape. The malleus head is a little bulky, and it can often rest on the promontory when placed between the stapes head and malleus handle.
The different methods of remodeling the incus are demonstrated in ▶ Fig. 28.1 and ▶ Fig. 28.2, and over time a surgeon will develop a technique that suits his or her particular practice. In addition, autograft reconstruction is not always the best choice, so most experienced surgeons will choose a technique and material that is best for a particular ear. The authors primarily use the incus when the malleus handle is not significantly offset to the stapes head and there is no evidence of persistent Eustachian tube dysfunction.
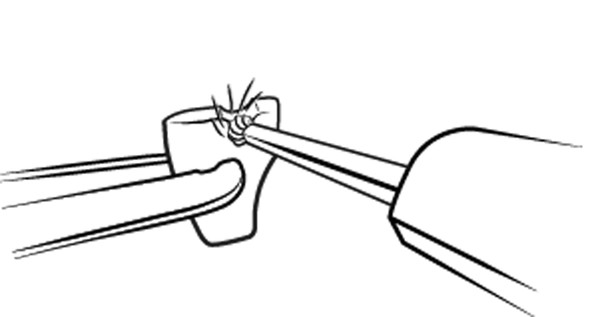
Fig. 28.1 Sculpting of incus using a diamond bur.
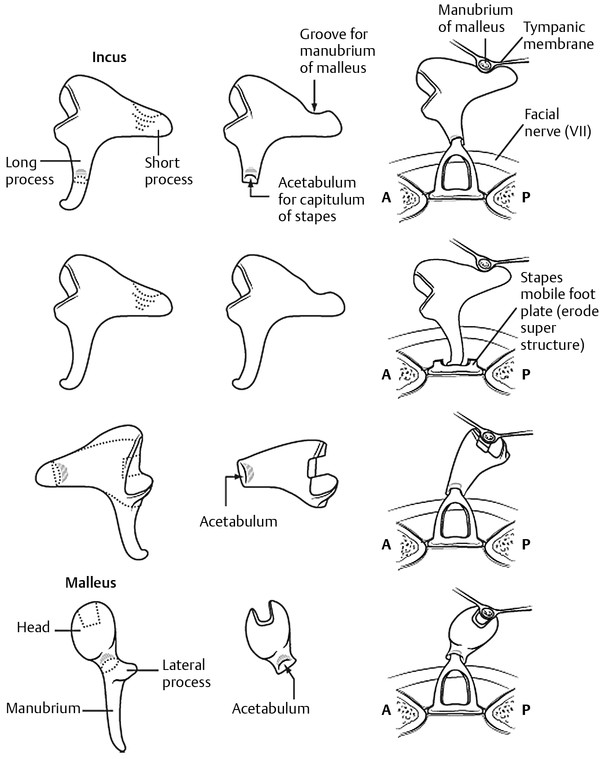
Fig. 28.2 Modifications for sculpting the incus and malleus. The authors prefer the modification shown in the third row. The modification in the second row is uncommon and is used more often with a homograft incus.
The incus remnant is scrubbed with a gauze swab to be certain that all cholesteatoma matrix has been removed, and then the body is carefully gripped with a mosquito forceps (▶ Fig. 28.3). The grip needs to be secure, and the incus should never be released until it is certain that it will not spring free. (There is nothing more disheartening than seeing the incus fly through the air and onto the floor!) A clean piece of drape should be used, and the microscope should be focused onto the incus that is held in the forceps. The mosquito forceps has a gentle curve, and the part of the incus that requires drilling faces the convexity while the index finger supports the other side within the mosquito forceps concavity. An assistant drips saline onto the incus as the drill is gently worked across the surface, refashioning the incus remnant to its desired shape without burning the bone. The incus should never be held with fingers while drilling as there is a significant risk that the surgical gloves will quickly become wrapped around the bur’s shaft. This is rather frightening and, once experienced, never repeated.
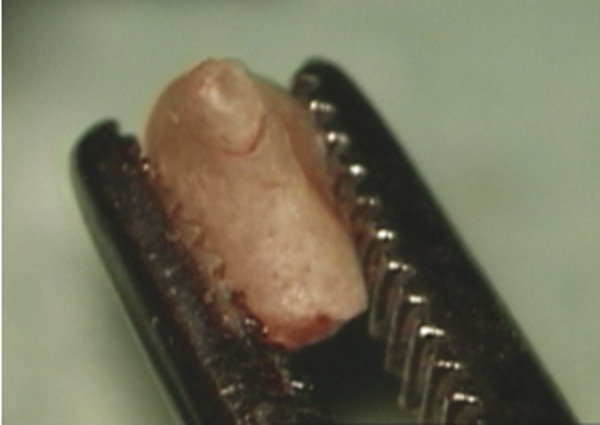
Fig. 28.3 Incus gripped between the jaws of a mosquito forceps with the short process uppermost.
The authors’ favorite incus design is one in which the short process sits on the stapes head and the disarticulated incudomalleal joint sits under the malleus handle. Any remnant of the eroded long process is removed, and a hole is drilled into the short process to accommodate the stapes head. To achieve the perfect fit, one must start with a large bur and gradually work with smaller, finer ones.
The short process is gently flattened with a 4- or 5-mm fine diamond bur. The surgeon should choose the largest and finest bur that he or she is comfortable with to reduce the risk of dislodging the incus from the mosquito forceps. A flat surface that is at least 2 mm in diameter is created, so that when the hole for the stapes head is drilled using a 1-mm fine diamond bur, there is at least 0.5 mm of healthy bone around its circumference (▶ Fig. 28.4). The hole for the stapes head lies opposite the disarticulated incudomalleal joint, which is the perfect shape to house the undersurface of the malleus handle. The refashioned incus is often tucked up against the tendon of the tensor tympani for further support. If it is too large, then more of the short process can be removed and the hole deepened for the stapes head.
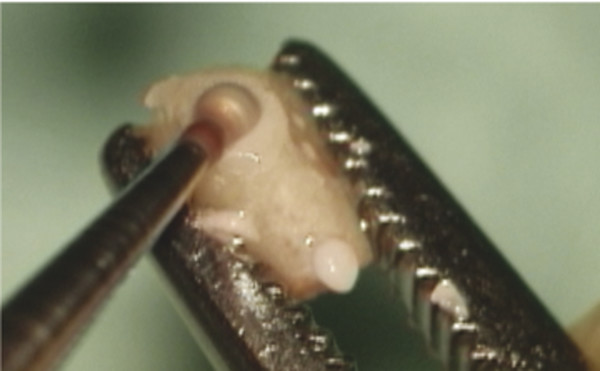
Fig. 28.4 Short process gradually reduced using a fine diamond bur so that the diameter of the flat surface is at least 2 mm. A 1-mm bur is then used to create a notch for the stapes head.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


