Fig. 18.1
(a) Cross section of pontomedullary junction at level of lateral recess of the fourth ventricle (4th Vent.). DCN (Dorsal Coch. Nucl.) and VCN (Ventral Coch. Nucl.) are located adjacent to the lateral recess (Lat. Recess) of the fourth ventricle. DCN underlies a prominence, while VCN is more deeply imbedded in brainstem and does not produce as great a prominence on surface of brainstem. (b) Retrosigmoid view. Flocculus has been elevated to expose junction of vestibulocochlear nerve with side of brainstem at pontomedullary junction. Foramen of Luschka (For. Luschka), which is positioned dorsal to glossopharyngeal nerve (CNIX), is partially covered by choroid plexus. (c) Choroid plexus has been retracted rostrally to expose dorsal cochlear nucleus sitting in floor of lateral recess. Ventral cochlear nucleus is positioned in area between lateral edge of dorsal cochlear nucleus and junction of vestibulocochlear nerve (CN VIII) with brainstem [16]
18.4 Complications Specific to Auditory Brainstem Implantation
Compared to standard cochlear implantation, stimulation of nonauditory neurons, electrode migration, and cerebrospinal fluid (CSF) leakage are listed as ABI-specific complications. Up to 42 % of multichannel ABI users suffered from nonauditory sensations, including nausea, tingling in the throat, jittering of the visual field, and shoulder contraction, that were probably caused by activation of the vagal nerve, glossopharyngeal nerve, flocculus of the cerebellum, and accessory nerve, respectively [17, 18]. Incorrect positioning of the ABI electrode array is usually associated with nonauditory sensations. Accurate placement of the electrode array on the surface of the cochlear nucleus is essential in auditory brainstem implantation; however, a large tumor often compresses the pontomedullary junction and creates difficulty in identification of the landmarks surrounding the cochlear nucleus. Changing the program of the speech processor, including deactivating the responsible electrodes and decreasing the amplitude of electrical stimuli, is usually effective to reduce these aversive ABI-mediated symptoms [17, 19]. Unlike the CI with an electrode array that is inserted into the cochlea, the electrode array of the ABI is placed on the surface of the brainstem with an unstable fixation. The fixation of the electrode array has been improved by the development of silicone backing, Dacron mesh, and nonelastic wire, but migration and dislocation of the electrode array still occur, especially in NF2 patients with large tumors compressing the brainstem. In these patients, the shape and position of the brainstem may change after tumor resection, which probably increases the risk for postoperative migration of the electrode array. Migration of the electrode array is usually associated with nonauditory sensations and deterioration of auditory perception; accordingly, these manifestations suggest the necessity of evaluating the position of the electrode array using high-resolution CT. The electrode lead of the ABI penetrates the meningeal dura and CSF may leak from the subarachnoid space to the mastoid air cells or the subcutaneous space along the electrode lead, regardless of the surgical procedure and choice of translabyrinthine or retrosigmoid approach [20, 21]. The percentage of CSF leakage in the ABI population is reported to range from 3.3 to 11 % [17, 19]. CSF leakage is usually controlled conservatively with or without lumbar drainage, but revision surgery is sometimes necessary.
18.5 Hearing Outcomes for Auditory Brainstem Implantation
During the last two decades, several groups have published the results of auditory brainstem implantation in NF2 and non-NF2 patients [17, 18, 21–24]. In general, hearing outcomes were lower in ABI recipients compared with outcomes for patients with CIs, but ABI-aided audiological performance varied widely according to the etiologies of deafness. As Colletti et al. established following long-term observation of 80 ABI adults with or without NF2 (Fig. 18.2) [23], trauma-induced cochlear nerve disruption and cochlear ossification were associated with favorable ABI outcomes and open-set speech discrimination scores greater than 50 %, while subjects with NF2 and auditory neuropathy spectrum disorder (ANSD) showed only limited improvement of auditory performance, even though the majority of these patients had ABI-aided benefits in daily life, particularly in combination with lip reading. Patients with congenital inner ear malformations showed moderate ABI-aided speech discrimination scores that were better than those in the NF2 patients, but worse than those in patients with trauma-induced cochlear nerve disruption and cochlear ossification. Considering that both populations (NF2 patients and patients with cochlear nerve disruption and cochlear ossification) suffered from postlingual deafness, the limited benefit of ABI in patients with NF2, which was also reported by other studies [17, 22, 25], should be related to NF2-specific problems such as direct damage to the cochlear nucleus caused by chronic tumor compression on the brainstem, as well as surgical procedures for tumor removal. ANSD is diagnosed by (1) absent or abnormal ABR and (2) present otoacoustic emissions and/or cochlear microphonics. Several reported etiologies causing sensorineural hearing loss meet these criteria, suggesting heterogeneity among patients with ANSD. Several studies have demonstrated that ANSD is highly associated with CND in the congenitally deaf population [26, 27]. In patients with an otoferlin-encoding (OTOF) gene mutation, synaptic release from inner hair cells is primarily affected, whereas demyelination and axonal loss may be responsible for hearing loss in systemic neurodegenerative diseases such as Charcot-Marie-Tooth disease. In Colletti’s study, there was no description of the etiologies about ANSD; therefore, the exact reason for poor ABI-aided hearing outcomes in patients with ANSD was unclear. These results demonstrated that hearing loss of different etiologies was associated with varied hearing outcomes following auditory brainstem implantation.
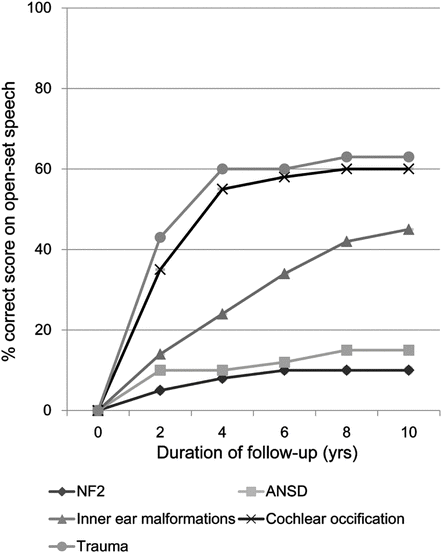

Fig. 18.2
Improvement of open-set speech discrimination scores (% correct) in patients with different causes of hearing loss after brainstem implantation [23]. NF2, neurofibromatosis type 2; ANSD, auditory neuropathy spectrum disorder; trauma, head trauma
18.6 Auditory Brainstem Implantation Compared with Cochlear Implantation
Previous studies showed that peripheral stimulation with CIs usually results in better outcomes than central stimulation of the auditory neural system using an ABI, suggesting that cochlear implantation should be tried before ABI whenever possible [10]. Utilization of the simple linear tonotopic organization in the cochlea and the natural sound processing mechanisms in the auditory nerves and neurons in the cochlear nucleus might contribute to favorable outcomes for cochlear implantation. The relatively high probability of ABI-related complications, including nonauditory stimulation, electrode migration, and CSF leakage, requires surgeons to carefully consider the indications for auditory brainstem implantation. As discussed previously, patients with Michel deformity or sacrifice of the cochlear nerve during tumor resection are definitely ABI candidates [8–10]. Trauma-induced bilateral cochlear nerve disruption is also an indication for auditory brainstem implantation, although this situation is unlikely to occur without the subject incurring fatal damage to the brain [8]. In patients with cochlear ossification and advanced otosclerosis, cochlear implantation may be followed by placement of an ABI [8]. For patients with cochlear aplasia or absence of the cochlear nerve or VCN, a more comprehensive analysis would be required to conclude the advantage of ABI. Due to the limitations of spatial resolution, high-resolution CT and MRI cannot exclude the possibility that a small number of cochlear nerve fibers innervate the inner ear, indicating the necessity for other functional evaluations. Since there are only a few reported cases of patients undergoing cochlear implantation followed by auditory brainstem implantation on the same side [28], the evidence for the advantages of an ABI compared with a CI for speech perception has not been fully established. In clinical application, a CI might be recommended for the initial operation in these patients. If the CI fails to provide sufficient auditory sensation, then an ABI may be the only solution. It should be noted that the efficacy of auditory brainstem implantation may decrease after the critical period for development of auditory perception if effective auditory stimulation is not available during the first several years after birth. Therefore, prelingually deaf children should undergo implantation of an ABI as early as possible if an ABI is indicated [9]. To resolve the dilemma of the need for careful decision-making regarding the indications for ABI and the importance of brainstem implantation at an early age, electrophysiological evaluation of the auditory system might be effective. As described in Chap. 15, intraoperative CI-mediated EABR testing may be useful to predict outcomes of CI [29]; thus, electrophysiological examination may be an effective method to determine the necessity for an ABI during cochlear implantation.
18.7 Future Prospects
As described above, different causes of hearing loss were associated with different hearing outcomes following implantation of an ABI [23]. Since the ABI directly stimulates the cochlear nucleus, ABI outcomes should be influenced by the severity of pathological changes in the cochlear nucleus. Compared with SGNs, neurons in the cochlear nucleus are less susceptible to loss of hair cells, but transneuronal loss of neurons in the cochlear nucleus occurs when the cochlea is damaged during an early period of development [30]. Interestingly, prior to exposure to normal environmental stimuli during the developmental period, spontaneous neural firing is observed in the central auditory system. Similar to other sensory systems, these spontaneous neural activities are thought to facilitate maturation of auditory neurons and their synapses [31]. Therefore, the congenital hypoplasia of SGNs that is associated with CND can attenuate synaptic input from the cochlear nerve to the neurons in the cochlear nucleus, which may lead to disorganization or immature development of neuronal connectivity in the cochlear nucleus. In this respect, CI-mediated intracochlear stimulation preceding auditory brainstem implantation may be effective to maximize the use of the remaining SGNs and promote development of the central auditory system, even if a CI would provide only limited auditory sensation. As discussed in Chaps. 28 and 30, the combination of a CI with the SGN regeneration or extension of SGN afferent dendrites by pharmacological or genetic methods can increase the responsiveness of SGNs to CI-mediated electrical stimuli, which in turn increases synaptic input from the cochlear nerve to the cochlear nucleus.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


