(1)
Department of Ophthalmology and Visual Sciences, University of Iowa Hospitals and Clinics, Iowa City, IA, USA
Anterior segment ischemia is not a common disorder but it can result in serious visual loss. In this condition, there is usually ischemia of the iris and ciliary body but it may involve the cornea and sclera. The blood supply of the anterior segment is discussed at length in Chap. 6.
Schmidt [1] gave the first clinical description of anterior segment ischemia in 1874. Crock [2] reviewed the history of anterior segment ischemia in 1967. The following account is based on his review. Clinical and pathologic features were described by Harbitz [3] in 1926. Leinfelder and Black [4] in 1941 described it on the experimental transposition of the extraocular muscles in monkeys. Skipper and Flint [5] in 1952 described symptoms and signs in 18 cases. Ask-Upmark [6] in 1954 described atrophy of the iris, cataract, and corneal opacities in Takayasu’s arteritis. Knox [7] reviewed the literature in 1965. Wilson and Irvine [8] first reported it after retinal detachment surgery in 1954.
Clinical Study of Anterior Segment Ischemia After Strabismus Surgery
This can be associated with a variety of clinical conditions. There are many reports, mostly anecdotal or based on only a few cases, of the development of anterior segment ischemia with ocular ischemic syndrome [7, 9–37], and the literature on the subject was reviewed in my study on ocular ischemic syndrome [37]. Other conditions include surgery involving cutting recti, for the treatment of strabismus [4, 38–52] or retinal detachment [2, 8, 40, 44, 53–64], giant cell arteritis [65–74], following cyclocryotherapy [75, 76], sickle cell hemoglobinopathy [59, 77], chronic myelogenous leukemia [78], chronic lymphocytic leukemia [46], Takayasu’s arteritis [6], occlusion of a posterior ciliary artery [79–81], Fuchs’ heterochromic cyclitis [82], in angle-closure glaucoma [83], after intravitreal silicone injection [84], quinine toxicity [7], chloramine mustard [2], herpes zoster [2], rubella [2], irradiation for malignancies around the orbit and the globe [2], and after perfusion of intractraial malignancies with nitrogen mustard [85].
Anterior segment ischemia is well known to develop after the cutting of various rectus muscles for the treatment of strabismus [4, 38–52] or in retinal detachment [2, 8, 40, 44, 53–64] after cutting one or more rectus muscles, with extensive diathermy applications to the uveal tract, including the long posterior ciliary arteries (PCAs), and encircling procedures. The anterior segment is supplied by various anterior ciliary arteries (ACAs) in all the rectus muscles and by the long PCAs. I investigated, by experimental and clinical studies, the effects of cutting various ACAs or long PCAs on the development of anterior segment ischemia.
Experimental and Clinical Studies
Anterior Segment Ischemia After Recession of Various Recti [51]
I carried out recession of various recti (in the combinations shown in Table 20.1) in 40 normal adult cynomolgus monkey eyes, to evaluate the effects on the anterior segment. The various types of recession were combined to evaluate their effects on the anterior segment; such combinations may be considered desirable in clinical practice, but a number of them are not done by ophthalmologists for fear of producing anterior segment ischemia. These procedures were done as primary, secondary, tertiary, and fourth (in one eye) procedures (each separated by several days, weeks, or months), ultimately involving all four rectus muscles in the eyes. The successive surgeries were always separated by a time interval that enabled the eye to recover normal filling of the iris vessels on fluorescein angiography, so that preoperative iris angiography in every case showed normal circulation in the iris.
Table 20.1
Details of recession of the various recti in different combinations and other additional procedures
First surgery | Interval (days) | Second surgery | Interval (days) | Third surgery | |||
|---|---|---|---|---|---|---|---|
Procedure | Totaleyes | Range (mean) | Procedure | Totaleyes | Range (mean) | Procedure | Totaleyes |
MR + LR | 3 | 51–100 (77) | SR + IR | 3 | 126–168 (147) | All 4 recti | 2 |
SR + IR | 4 | 163–169 (166) | MR + LR | 4 | 125–141 (135) | All recti | 3 |
MR + IR + SR | 6 | 54–80 (63) | All 4 recti | 3 | – | – | – |
7 | LR | 1 | |||||
7 | APACO | 1 | 2 | LR | 1@ | ||
LR + IR + SR | 6 | 142,149 | All 4 recti | 2 | – | – | – |
SR + MR | 2 | 70, 120 | IR + LR | 2 | 160,217 | All 4 recti | 2 |
SR + LR | 2 | 60,128 | IR + MR | 2 | 154,159 | All 4 recti | 2 |
All recti | 7 | – | – | – | – | – | – |
APCAO + LR + MR | 5 | – | – | – | – | – | – |
APCAO + LR | 1 | – | – | – | – | – | – |
LPCAO + LR | 2 | 125 | All 4 recti | 1 | – | – | – |
APCAO | 1 | 6 | LR + MR | 1 | 7 | SR + IR | 1 |
APCAO + all 4 recti | 1 | – | – | – | – | – | – |
Tenotomy of the recti cuts the anterior ciliary arteries (ACAs), but the anterior segment is also supplied by the long posterior ciliary arteries (branches of posterior ciliary arteries – PCAs) (see Chap. 6); therefore, in some eyes, the long PCAs were also cut, alone, or in combination with different recti, to find the effect. The anterior segment was evaluated by slit lamp examination and fluorescein iris angiography. The findings are discussed in detail elsewhere [51]; what follows is a brief description of the main features following the major primary procedures.
Simultaneous Recession of the Horizontal Recti (Four Eyes)
Iris Circulatory Defects
There were transient filling defects in the corresponding horizontal segments in two for 1–6 days, in the nasal half only in one, and no defect in one.
Anterior Segment Changes
The main finding was mild flare and cells, with no other abnormality.
Simultaneous Recession of Vertical Recti (Four Eyes)
Iris Circulatory Defects
There was a defect involved ¾ of the iris, leaving the nasal ¼ unaffected, in 3 of the eyes and in one eye the superior segment was spared. The defect lasted for 7–21 days.
Anterior Segment Changes
One eye showed marked flare, mild hyphema, folds in the Descemet’s membrane, dilated iris vessels, and sectoral iris atrophy in the superior quadrant. One eye showed iris neovascularization. The remaining two eyes showed no abnormality.
When it was a secondary procedure, it still showed almost identical changes.
Simultaneous Recession of Superior, Inferior, and Medial Recti (Six Eyes)
Iris Circulatory Defects
There was a defect involving ¾ of the iris in all the eyes (Fig. 20.1), sparing the nasal quadrant in three, superior nasal in two and temporal in one. The defects lasted for 5–62 days.
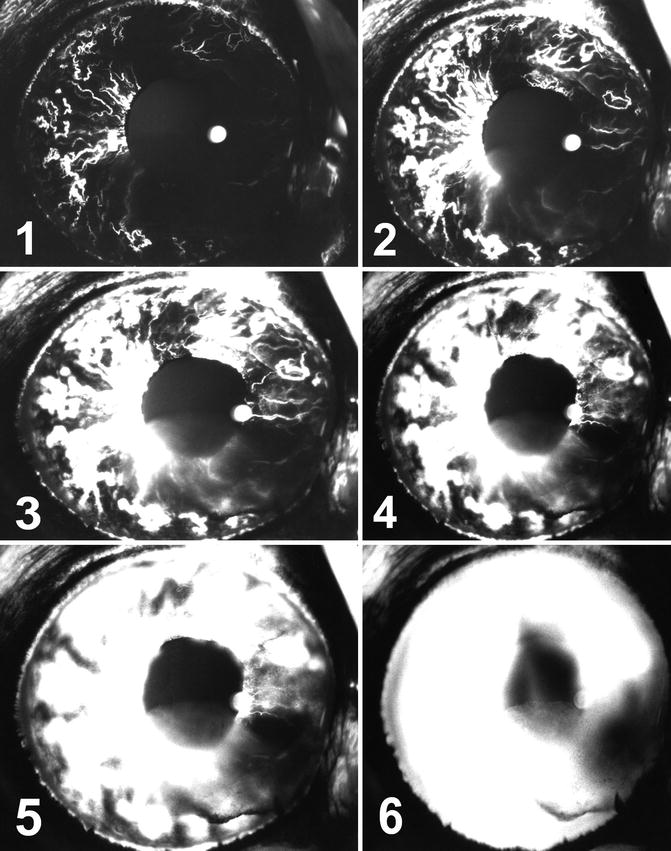

Fig. 20.1
The eye in cynomolgus monkey with simultaneous tenotomy of medial, superior, and inferior recti 6 days after surgery shows extensive filling defects and anterior segment ischemia with iris neovascularization and fluorescein leakage. Angiograms (1) 8.9, (2) 11.9, (3) 14.9, (4) 21.8, (5) 36.8, and (6) 56 s after the dye injection
Anterior Segment Changes
The anterior chamber in all the eyes developed mild to moderate flare and cells, and in addition exudates developed in one eye. Pupils were dilated in five eyes, and normal in one. Iris neovascularization (Fig. 20.1) developed in two eyes on day 5. Later on the iris was normal in five eyes and showed sectoral iris atrophy in one. A fall in intraocular pressure (IOP) developed in four of the eyes, a rise in one, and one had normal IOP. Fluorescein leakage from the iris vessels was seen in four eyes. Cornea and lens were normal in all eyes.
Simultaneous Recession of Superior, Inferior, and Lateral Recti (Six Eyes)
Iris Circulatory Defects
There was a defect involving 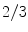 to ¾ of the iris (Fig. 20.2), sparing the nasal part in four eyes, confined to the superior and inferior sectors in one, and the temporal half in one eye. The filling defect lasted from 5 to 31 days.
to ¾ of the iris (Fig. 20.2), sparing the nasal part in four eyes, confined to the superior and inferior sectors in one, and the temporal half in one eye. The filling defect lasted from 5 to 31 days.
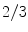 to ¾ of the iris (Fig. 20.2), sparing the nasal part in four eyes, confined to the superior and inferior sectors in one, and the temporal half in one eye. The filling defect lasted from 5 to 31 days.
to ¾ of the iris (Fig. 20.2), sparing the nasal part in four eyes, confined to the superior and inferior sectors in one, and the temporal half in one eye. The filling defect lasted from 5 to 31 days.
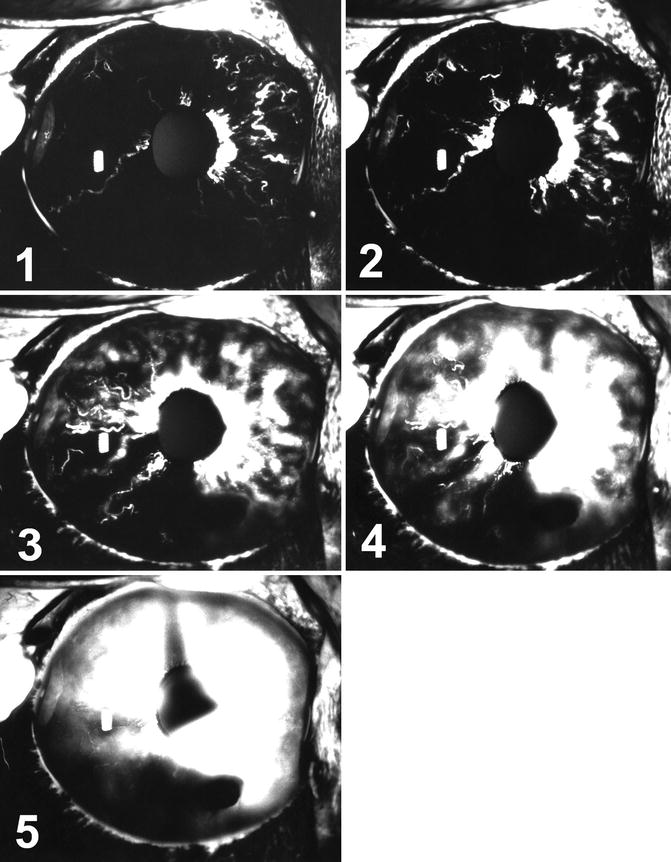
Fig. 20.2
The eye in cynomolgus monkey with simultaneous tenotomy of lateral, superior, and inferior recti 6 days after surgery. It shows extensive filling defects and anterior segment ischemia with iris neovascularization and fluorescein leakage and hyphema inferiorly. Angiograms (1) 7.8, (2) 10.7, (3) 18.6, (4) 32.2, and (5) 122.2 s after the dye injection
Anterior Segment Changes
The anterior chamber in all eyes showed flare, cells, and pigment dispersion. The pupil showed sectoral loss of reaction to light in five eyes, almost corresponding to the areas of iris non-filling. In the 2 eyes with the longest delay in iris filling on angiography, iris atrophy developed in the temporal and inferior temporal regions corresponding to iris filling defects on angiography. Iris neovascularization developed in five eyes (Fig. 20.2) and all of them had a fall in IOP.
Simultaneous recession of both vertical recti combined with one of the horizontal recti, although producing a fairly marked anterior chamber reaction with pupillary changes during the immediate postoperative period, resulted in minimal long-term complications, with the cornea and lens normal and the iris showing sectoral atrophy in 3 of 12 eyes.
Simultaneous Recession of All Four Recti as Primary Procedure (Seven Eyes)
Iris Circulatory Defects
There was defect involving ¾ of the iris in five eyes, sparing nasal quadrant in 4 and superior temporal quadrant in one. The blood supply of the entire iris was absent in one eye. In the remaining eye, the defect was confined to superior and inferior sectors. The filling defect lasted from 6 to 117 days.
Anterior Segment Changes
The anterior chamber in all eyes showed fairly marked flare, cells, and hyphema, and in some exudate and pigment dispersion. The pupil was dilated in all, with complete or segmental loss of reaction to light. Corneal edema, folds in Descemet’s membrane, and an increase in corneal thickness occurred in five eyes. In one eye, the cornea became totally opaque and phthisis bulbi finally developed. In one eye, there was pigment migration and temporary corneal cloudiness. Iris neovascularization developed in five eyes. Iris atrophy developed in all the eyes. A fall in IOP occurred in all eyes, coinciding with the onset of iris neovascularization.
This shows that when all four recti were recessed simultaneously, as a primary procedure, the anterior segment changes were the most severe and the eyes were left with permanent lesions (e.g., cataracts in five of six eyes, atrophic iris in all, corneal vascularization and opacity in two). However, when the simultaneous recession of the four recti was a secondary procedure (preceded by a simultaneous recession of both vertical recti and one horizontal rectus, about 2–5 months earlier), the anterior segment changes were comparatively much fewer and milder (e.g., corneal or lens changes did not develop in any of the six eyes, and iris atrophy did not even develop in two of the eyes). When the simultaneous recession of the four recti was a tertiary procedure, the anterior segment changes were once again fewer and milder than after the primary procedure. This indicates that simultaneous recession of all four recti as a secondary or tertiary procedure (after a previous recession of three to four muscles, 2–5 months earlier) did not produce serious permanent ocular complications.
Simultaneous recession of one vertical and one horizontal rectus (or both horizontal or both vertical recti), as a primary or secondary procedure, almost invariably produced no changes in the anterior segment (apart from angiographic filling defects in the iris) despite the fact that in all of these eyes, after the second procedure, all four recti had been recessed in two stages. As an exception to this, however, one eye, with simultaneous recession of the vertical recti as a primary procedure, showed a moderate anterior segment ischemia during the immediate postoperative period.
Simultaneous Occlusion of Both Long PCAs and Recession of the Horizontal Recti
In five eyes with this as a primary procedure, evidence of anterior segment ischemia during the acute phase was very common, and later iris neovascularization developed in four of the eyes, corneal neovascularization in two, iris atrophy in one, cataract in one, and prolonged hypotony in three. The anterior segment changes were thus much more severe in this group than after the recession of the corresponding recti alone or occlusion of the PCAs alone, indicating a compounding effect of the two procedures. Occlusion of the long PCAs and simultaneous recession of both the horizontal recti may be done during retinal detachment surgery. So the risk of producing anterior segment ischemia with this procedure is high, and the eye may be left with permanent serious complications.
Some strabismus surgeons suspect that fornix conjunctival incisions, which may cause less disruption of perilimbal conjunctival circulation, help protect the eye from anterior segment ischemia after recession of rectus muscles. In this experimental study, as in standard strabismus surgery, the conjunctival incision was made 4 mm from the limbus. In monkeys, unlike white patients, there is marked pigmentation of the perilimbal conjunctival area, so that it is not possible to outline the perilimbal vessels on fluorescein angiography of the anterior segment. Moreover, immediately after the strabismus surgery, there is subconjunctival hemorrhage and postoperative tissue reaction in that region, masking the underlying perilimbal and episcleral vessels; thus, in most cases, it is almost impossible to evaluate the circulation in the perilimbal region by fluorescein angiography, neither in monkeys nor in humans. In my fluorescein angiographic studies on humans (see below) [48], in the eyes with tenotomy of one of the vertical recti and satisfactory angiograms of the perilimbal vessels, I found that most of the eyes showed filling defects in the corresponding iris as well as the perilimbal vessels, because the perilimbal region is supplied by the corresponding episcleral branches of the anterior ciliary arteries (Fig. 20.3). In these eyes, the filling of the perilimbal and iris vessels was usually related. There are free anastomoses between the adjacent perilimbal vessels (through the episcleral circle – Fig. 20.3). This thereby suggests that the iris vessels in the region of a cut vertical rectus may fill from the perilimbal vessel via the following route. When the ACAs of one rectus muscle are cut, a retrograde flow of blood may take place from the perilimbal vessels to the episcleral branches of the cut ACAs and then via the perforating branches to the iris vessels (Fig. 20.4). It is therefore possible that extensive extraocular muscle surgery associated with destruction of the episcleral and perilimbal vessels in the corresponding region may render the eye susceptible to anterior segment ischemia.
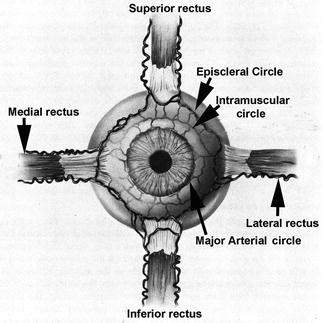
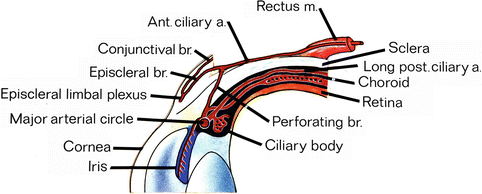

Fig. 20.3
Diagrammatic representation of anterior ciliary arteries running along the borders of the four recti muscles and also of episcleral, intramuscular, and major arterial circles (Modified from Van Buskirk [86])

Fig. 20.4
Pattern of blood supply of iris and anterior segment (Reproduced from Hayreh and Scott [48]) Abbreviations: a artery, br branch, m muscle
The findings of my study also show that iris angiography is a very useful test to evaluate ischemic changes in the anterior segment because it shows, apart from the presence of filling defects in the iris, fluorescein leakage from the iris vessels, which is an important sign of ischemia.
It could be argued that these findings from the monkeys may not be applicable to humans. Based on my anatomical studies in humans and primates, as well as long clinical studies in humans and experimental experiences with monkeys, I think that they are indeed applicable, because the vascular pattern and other anatomical and physiological patterns of the anterior segment are identical in humans and monkeys. The other objection that could be raised is that there were not large numbers of eyes in each group; however, I think there were a reasonable number of eyes in the important groups to give us realistic information.
Conclusions
1.
The nasal and temporal sectors (especially the former) have a dual blood supply – primary from the ACAs of the horizontal recti and subsidiary from the long PCAs; the former source plays a far more important role than the latter. Because most of the temporal perforating arteries are branches of the ACAs (Fig. 20.4) of the vertical recti [87], they constitute the major source of blood supply to the temporal sector. When recession of the medial rectus was combined with that of one of the vertical recti, the nasal iris filling defects were more frequent and more extensive than those seen after recession of the medial rectus alone. This suggests that even in the nasal region, the ACAs of the vertical recti play some role in the blood supply.
2.
The superior and inferior sectors are mainly supplied by the ACAs of the vertical recti, with no long PCAs in those regions. Cutting the vertical ACAs usually produces an abnormal filling defect in those areas, and the defect mostly extends to involve a part of the adjacent temporal sector also, because the ACAs of the vertical recti also generally contribute to the temporal region [87]. This study indicates that the ACAs of the vertical recti play a crucial role (more important than either the ACAs of the horizontal recti or the long PCAs) in the blood supply to the anterior segment.
3.
The contribution by the long PCAs to the anterior segment is usually not very important. This study, as well as my experimental studies on the occlusion of the long PCAs (see below) or parent trunks of the long PCAs (i.e., the main PCAs [88–91]), has clearly shown that occlusion alone does not produce any anterior segment ischemia. This was further confirmed by my patients with giant cell arteritis in whom fluorescein fundus angiography showed complete occlusion of the main PCAs, without any evidence of anterior segment ischemia. On the other hand, in seven eyes, with normal PCA circulation but simultaneous recession of all four recti as a primary procedure, immediate postoperative iris fluorescein angiography showed some filling of the nasal quadrant in four and of a small temporal sector in two, and no filling of the iris at all in one eye, with severe anterior segment ischemia developing in all eyes. The latter indicates that the long PCAs, even when they contribute to the blood supply of the anterior segment, do not protect it from acute ischemia in the event of occlusion of the ACAs. The evidence of this experimental study also indicates a frequent additive effect of occlusion of long PCAs with the occlusion of the horizontal ACAs, as in retinal detachment surgery.
4.
With regard to the disparity between the postmortem anatomical studies and in vivo occlusion studies, I conclude the following. Although the former studies show the presence of extensive collateral anastomoses in the anterior segment (i.e., via the episcleral circle, intramuscular circle, and major arterial circle of iris), in vivo occlusion studies indicate that these anastomoses are usually not functioning in the living eye. Similarly, in the PCA circulation and in the choroidal vascular bed, my in vivo occlusion studies clearly demonstrated that those anastomoses do not function as collaterals to establish circulation during the immediate post-occlusion period in the living eye [88, 90, 92, 93], although postmortem injection anatomical studies revealed extensive anastomoses. The reason for this disparity between the postmortem and in vivo studies is still not clear.
5.
My studies [94] established that there is marked interindividual variation in the distribution by the PCAs in humans, and this experimental study suggests that the same may be true of the distribution by the ACAs and long PCAs. This would explain the interindividual variation in the response after recession of all the four recti as well as in other groups. It seems that either certain sectors of the iris have more than one source of blood supply with interindividual variation or the various arterial systems are independent and are connected to each other through collaterals, the situation, number, and efficacy of which determine the type of iris filling defect, and the degree of iris ischemia.
Long Posterior Ciliary Artery Occlusion [91]
The long PCAs supply the anterior segment of the eye. There are many studies on the effects of experimental occlusion of the long PCAs on the anterior segment of the eye and the IOP [96–103]. Most were carried out in rabbits; the production of ocular hypotony, phthisis bulbi, iris neovascularization, cataract, and anterior segment necrosis has been recorded. Interference with the long PCAs has been attributed to the occurrence of anterior segment ischemia following diathermy, scleral buckling, and some other procedures for retinal detachment [96].
In view of this, I investigated the effect of cutting a long PCA. This was done in two studies:
In all cases, the anterior segment remained normal.
Retinal detachment surgery involving cutting of one or more rectus muscles, with extensive diathermy applications to the uveal tract, including the long PCAs, and encircling procedures, have been known to produce anterior segment necrosis. In such cases, the occlusion of the long PCAs has erroneously been considered to be the major factor in the production of anterior segment ischemia. The role of the cutting of the ACAs discussed above, and interference with the anterior uveal venous drainage [95] as the major factor in these cases, has been ignored or minimized, because of the wrong concept created by the rabbit experiments. In rabbits, the major blood supply to the anterior segment of the eye is by the long PCA [104], but that is not the case in primates or humans. The observations in primates on occlusion of the long PCAs indicate that this by itself plays no significant role in anterior segment ischemia in man, although it may act as a minor contributing factor in combination with interruption of the ACAs, and interference with venous drainage [95] from the anterior uveal tract due to liberal diathermy and indentation procedures in retinal detachment surgery. As shown above, this is further demonstrated by the absence of any lesions on follow-up in the anterior segment of eyes with occlusion of one or both of the long PCAs.
Vortex Vein Occlusion
Venous drainage from the anterior segment is into the vortex veins. If venous outflow is blocked, that can result in arterial blood flow impairment – if the blood cannot get out, it cannot get in. Therefore, venous occlusion from the anterior segment can result in ischemia of the anterior segment. Clinically, retinal detachment surgery may be associated with vortex vein occlusion, caused by an encircling band located anterior to the vortex veins or accidental injury to the vortex veins, and extensive diathermy of the uveal region. I investigated experimental occlusion of the vortex veins in 40 eyes of rhesus monkeys [95]. The findings of that study are discussed in detail in Chap. 27.
Briefly, when all the vortex veins were occluded, the result was anterior segment ischemia and phthisis bulbi, with corneal stromal vascularization, iris atrophy with complete or partial anterior and posterior synechiae, cataractous lens, and permanent marked hypotony. Occlusion of 3 vortex veins resulted in sectoral iris atrophy with posterior synechiae only. Occlusion of one or two vortex veins produced no evidence of anterior segment ischemia.
Early clinical signs of complications of vortex vein occlusion include flare and in severe cases proteinous material and gelatinous deposits in the anterior chamber, and even hyphema; these may be mistaken for uveitis or infection. Fluorescein angiography shows a marked leak of fluorescein from the posterior chamber into the anterior chamber after intravenous injection of the dye.
Clinical Study of Anterior Segment Ischemia After Strabismus Surgery
Ischemia of the anterior segment of the eye is a well-established complication of strabismus surgery [4, 38–52]. I investigated this in patients with blue or green eyes, who underwent surgical correction of their strabismus. Patients with blue or green eyes were selected so that iris fluorescein angiography could provide information about the filling defects in the iris on iris fluorescein angiography; in brown eyes angiography does not provide that information. Their ages varied between 10 and 64 years. Table 20.2 gives details of patients in this series with strabismus operation, and iris vascular abnormalities on fluorescein angiography after various procedures are described in detail elsewhere [48]. Since then, I have seen more such cases, and information from some of those is also included.
Table 20.2
Details of patients in this series with strabismus operation
Procedure
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|
|---|