Anatomy of the Zonular Apparatus
Barbara W. Streeten
The zonular apparatus is a three-dimensional complex of microfibrils identical to the microfibrils of the elastic system in the rest of the body. These microfibrils are noncollagenous 10-nm beaded microfibrils composed predominantly of the protein fibrillin, and aggregate into fibers passing from the inner surface of the ciliary body to the outer surface of the lens capsule (Fig. 1). They are the only structural component directly holding the lens in place and are also designed to mediate movement forces of the ciliary muscle to the lens for accomodation. By attachments to the anterior hyaloid membrane, the posterior zonules may help to maintain this membrane and the anterior vitreous in apposition to the ciliary body and lens, which may play an additional role in accomodation.
Because of great difficulties in understanding and even describing the elusive zonule, many terms have been used for it since the first complete description by the anatomist Zinn in 1753,1 including the zonule of Zinn, the annular ligament of Zinn, the suspensory ligament of the lens, the ciliary zonule, and the tertiary vitreous.
DEVELOPMENT AND SYNTHESIS OF THE ZONULE
Zonular fibers are first seen at the end of the third fetal month, lying within the secondary vitreous at its base and passing through the matrix of the tunica vasculosa lentis to attach to the lens capsule.2 The mechanisms and factors guiding their highly oriented and predictable attachment to sites on the lens capsule are still unknown. In late fetal life the adult pattern is already apparent, with silvery zonular fibers passing over the tops of the low ciliary processes while others dip into the valleys between the processes, and some change direction by running up along the sides of the processes (Fig. 2). The primary candidate for synthesis of the zonule is the ciliary nonpigmented epithelium. Autoradiographic studies on fetal and young mammalian eyes showed an early accumulation of labeled carbohydrate components and amino acids including cys-teine (present in high concentration in the zonule)on zonular fibrils close to the epithelium of the pars plana and posterior pars plicata.3,4 This proximity suggests that new protein is added at the ciliary end of the zonule and that the ciliary nonpigmented epithelium is its source. In culture, these cells as well as lens and ciliary pigment epithelial cells are capable of producing fibrillin microfibrils,5,6 as do many basement membrane-secreting epithelial cells. By in situ hybridization, the ciliary nonpigmented epithelium of the fetal mouse is indeed positive for fibrillin-1, the main protein of zonular and other elastic system microfibrils.7 However, the lens epithelium in the metabolically most active equatorial region has some fibrillin-1 positivity at the same developmental stages and may contribute as well to the early zonule.8 It is not known whether zonular synthesis is continuous postnatally through at least the most active period of ocular growth, or whether a continuous low-grade renewal process occurs.
COMPOSITION AND MOLECULAR GENETICS
Because the zonular fibers arise within the vitreous, they were for many years assumed to be collagenous like the vitreous, whose small type II collagen fibers are similar in size to those in the zonule, resulting in the zonule being termed the tertiary vitreous.2 In recent decades they were shown to be composed of noncollagenous glycoprotein,9,10,11,12 consistent with their positivity to the periodic acid-Schiff (PAS) stain and association with both O- and N-linked oligosaccharides.13
Electron microscopy allowed the first significant insight into the nature of the zonule. In 1971 Raviola noted that the 10-nm size and tubular ultrastructure of the zonular fibers and their susceptibility to α-chymotrypsin digestion were similar to characteristics of the newly described microfibrils of elastic tissue.14,15 These two types of microfibrils were found to be strongly cross-reactive immunologically16 and to have similar biochemical profiles, including an unusually high cysteine content.11,12
With the advent of the molecular era, the primary component in all elastic system microfibrils was identified as a 350-kDa glycoprotein and named fibrillin,7 coded for by a gene on chromosome 15 q21.1.17,18 Immunoreactivity of zonular fibers for fibrillin is strong7,19 (Fig. 3). Classic Marfan syndrome with ectopia lentis was quickly linked to mutations on the fibrillin gene,17,20 usually of the missense type. More than 70 such mutations have now been reported, including some mutations in cases of simple dominant ectopia lentis.21 Another fibrillin protein, fibrillin-2, was found at the same time on chromosome 5, causing congenital contractural arachnodactyly when mutated, but no ocular involvement.17 More than 10 proteins besides fibrillin-1 have since been found associated with the elastic system microfibrils, many of them also with zonular microfibrils, from which one of the new proteins was isolated.22,23 It is unclear how many of these proteins are intimately involved in the structure of the microfibril, or what functions they may have. Most are known by acronyms referring to their microfibril glycoprotein nature, such as MAGP-1 and -2, MFAP-1 to -4, MAP, GP115/emilin, and LTBP-2, but also included are more familiar ubiquitous proteins including fibronectin and vitronectin.24
 Fig. 3. Insertion of the anterior zonular fibers, strongly reactive with fibrillin-1 antibody on a completely nonstaining lens capsule. |
The fibrillin-1 molecule has a large number of repeating domains resembling epidermal growth factor (EGF-like), all but two of the calcium-binding type, where most of the Marfan mutations have occurred.24 This is consistent with evidence that the presence of calcium is important for the stability of this unique beaded microfibril.25,26 The stability of the zonular bundles and their resistance to solubilization imply the presence also of strong cross-linkage sites, besides the great capacity for disulfide linkages between the many cysteines. Transglutaminase-induced glutamyl-lysine cross-linkages have also been identified in elastic microfibrils from several tissues.27 Sites in the molecule for RGD-integrin binding to cell membranes andmatrix are also recognizable.28 The adhesion mechanisms used by the zonular fibers are still incompletely known.
FUNCTION OF THE ZONULAR MICROFIBRILLAR SYSTEM
The elastic system is composed of three units,29 the first composed entirely of microfibrils, as far as can be determined identical to the zonular microfibrils. These are the first units to be synthesized during elastogenesis,15 serving as a template on which the completely different elastomeric protein, elastin, may be deposited. Many bundles of microfibrils in the body like the zonular type never become elasticized, and these are sometimes called oxytalan (acid-resistant) fibers.30 Like zonular bundles, they attach particularly to basement membranes of surfaces under stress, connecting them to each other just as zonular fibers connect the ciliary nonpigment epithelial basement membrane to the lens epithelial basement membrane. In deeper tissues these microfibrils can become partially elasticized bundles called elaunin fibers.29 These then connect to the larger fully elasticized elastic fibers, completing the third unit of the system. The long-recognized elasticity or stretchability of the zonular bundles clearly has a different basis than that of the elastomer elastin and appears to reside in reversible relationships of the fibrillin filaments to each other and to their associated beads, as well as to adjacent microfi-brils, when stretched. As part of the widespreadelastic system, the zonule and fibrillin-1 appear toshare in relatively few other systemic diseases be-sides the Marfan syndrome, including the Weil-Marchesani syndrome31 and the pseudoexfoliation syndrome.19,32,33
ZONULAR BUNDLE, FIBER, AND FIBRIL ARCHITECTURE
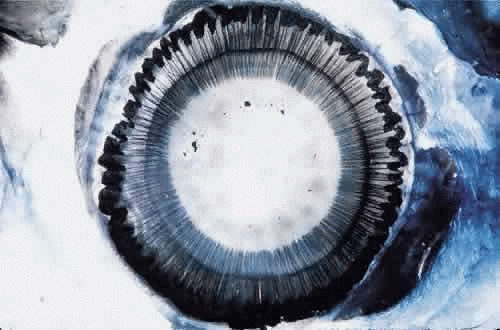
When seen during life through an iridectomy or under the dissecting microscope, the zonular fibers appear as glassy, straight rods of 5 to 30 μm in diameter (Fig. 4), occurring in diverging groups that appear as individual glassy rods when cut (Fig. 5). These groups of fibers are sticky when handled with instruments and show moderate elastic recoil after stretching by forceps. In one study of human eyes age 48 and 56 years, zonules attached to the lens capsule could be stretched for four times their normal length from capsule to ciliary body without apparent breakage or disinsertion.34 Because ofthis extensibility, zonular fibers and bundles canvary considerably in diameter, depending on processing methods.
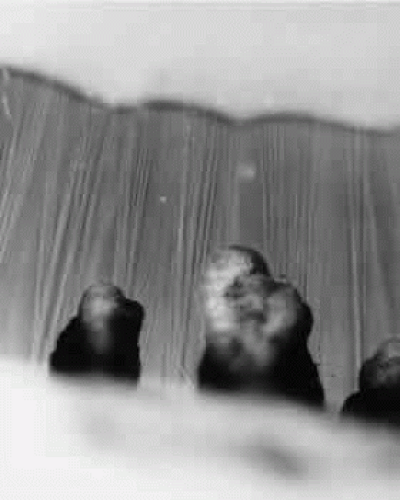 Fig. 4. Unstained zonules seen in situ in a whole eye under the dissecting microscope. Zonular bundles stretch between the dark ciliary processes below to the lens above (× 150). |
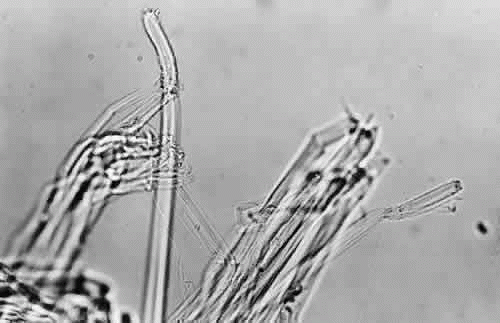 Fig. 5. When cut, the zonular bundles in Figure 4 appear as individual glassy rods (× 300). |
Understanding of the three-dimensional architecture of this almost invisible zonular system has been slow to build up by use of many types of microscopy and special stains,35,36,37,38 but the combination of transmission and scanning electron microscopy has been the best tool to prove that the zonule is a system of fibers with a course that can be fairly accurately defined.
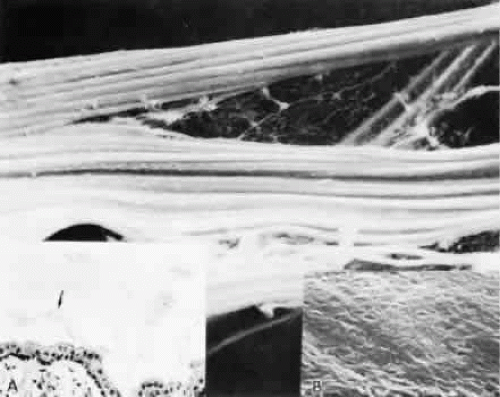
By light microscopy, zonular fiber bundles are eosinophilic, PAS-positive structures, often appearing to be ribbons connected by a paler interbundle “membrane” (Fig. 6). By scanning electron microscopy, the ribbons appear as groups of striated fiber bundles held together by a loose fibrogranular meshwork. Each fiber is composed of fibrils 10 nm in diameter, highly oriented and closely aggregated. Ultrastructurally, these fibrils have a tubular profile in cross section and a microperiodicity of 12 to14 nm seen along the fibril (Fig. 7). Dyes that precipitate and bind to polysaccharides show irregular granules and rod-like associated material on and between the fibrils.
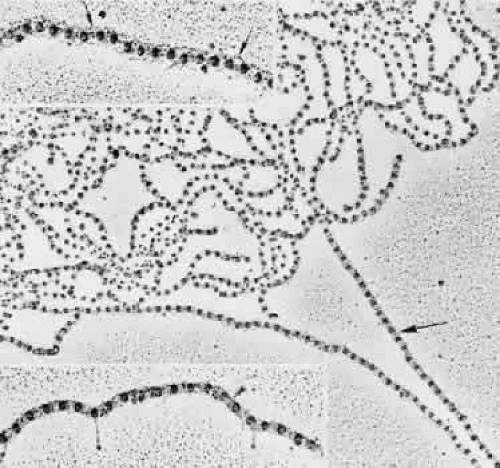
The remarkably fine structural details of these zonular fibrils, or microfibrils as the identical fibrils are called in the rest of the elastic system, are shown best by rotary shadowing and electron microscopy of freshly homogenized zonular fibers.39,40 The individual zonular fibril is a beaded structure (“beads on a string”) consisting of 29- to 32-nm electron-dense beads held together by filaments that may be four in number and 2 to 3 nm in diameter (Fig. 8). The filaments can be seen more clearly when the fibrils are in a looser conformation. When they are in tighter conformation, many show cross-banding across the filaments between two beads. The beads’ usual periodicity is 50 to 55 nm, but it can vary from 35 to 57 nm or more with different methods of preparation and 80 to 100 nm when stretched, without visible breakage of the filaments.41 Several studies suggest that changes in periodicity of the beads cannot fully explain the high degree of elasticity exhibited by fibrillin-containing microfibrils in tissue such as the zonular fibers, or in arteries of lower forms, perhaps requiring some reorientation of the fibrils.42,43 The nature of the bead is still not known, but the 148-nm filaments, the predicted length of a molecule of fibrillin, are thought to be arranged in a parallel staggered pattern with about one third overlap of adjacent molecules,27 although other patterns have also been suggested.44,45 The structure of elastic-system microfibrils throughout the body shows identical beaded fibrils.
ORIGIN OF THE ZONULE
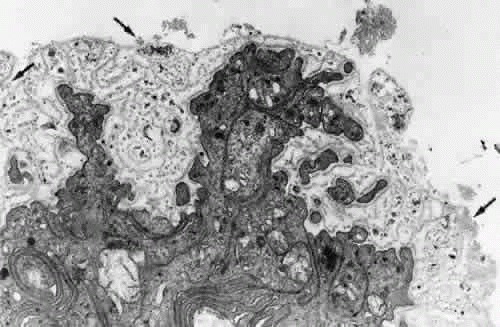
The ciliary zonular fibers originate in the pars plana as fine fiber bundles on the nonpigment epithelial basement membrane from 0.5 to 1 mm anterior to the ora serrata. They stain incompletely in gross specimens due to the overlying vitreous gel, narrowing to thin fibers as they near the ora (Fig. 9). By electron microscopy most appear to originate as small fibers containing 5 to 10 or more fibrils, adherent to the basement membrane of the epithelial cells, some becoming incorporated in the redundant infolding of the multilaminar basement membrane that develops with aging (Fig. 10). Small bundles are sometimes found superficially between these cells with no intervening basement membrane. Microfibrillar bundles found between some pigmented epithelial cells near their bases and in and under their basement membranes37,46 have been suggested to be zonular but are more likely a mixture of the abnormal elastic microfibrils found in the pigment epithelium’s aging basement membrane and/or related to inserting microfibrils from elastic fibers of the underlying ciliary body stroma.
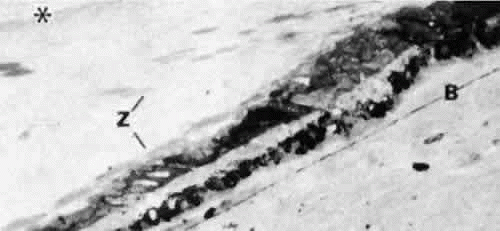
 Fig. 9. Zonules (Z) on the pars plana taper toward the ora serrata (OR) between two retinal teeth (Gomori’s hematoxylin, × 45). |
CILIARY COURSE OF THE ZONULE
Over the pars plana, the zonular fibers aggregate into bundles with the same pale membrane between them as illustrated in Figure 6, with multiple rows of these ribbons seen intermittently as new bundles are added (Fig. 11). The layer closest to the ciliary epithelium continues to have fine 0.1- to 0.5-μm fibers arising from the ciliary epithelium to join the main zonular stream over the rest of the pars plana, seen best in coronal sections, and sometimes is associated with small nodules of proliferated epithelium, called cap cells, appearing to be pulled inward by the fine attachments. These zonular bundles lie within the vitreous up to the mid-pars plana, where the anterior hyaloid membrane separates from the ciliary body to become an independent structure, and the zonules lie free in the posterior chamber (Fig. 12).
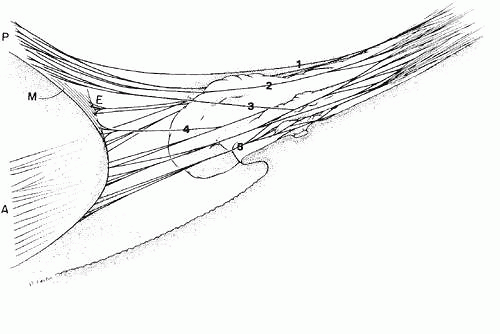
The course of the zonule from the beginning of the pars plicata has been more difficult to elucidate and depends to some extent on the modality used to visualize it, because all have their artifacts and limitations. Scanning electron microscopy was a great step forward in understanding the three-dimensional nature of the zonule,46,47,48,49,50,51,52,53,54,55 although defects in methodology and low resolution in the early years led to erroneous impressions. A longtime controversy of importance in accomodative theory is whether some zonular fibers originate on the pars plicata and have a different function from those that are continuous from the pars plana to the lens.47 Recent investigators of both human and primate zonules have tended to conclude that most zonularfibers are continuous from pars plana to the lens,53,55 but they differ in estimating the number that may originate or terminate on the pars plicata and the degree of zonular adherence to this tissue. The following description is a synthesis of views about zonular anatomy in the pars plicata, obtained by several different methods and diagramed in Figure 13.
With increasing height and complexity of the ciliary processes, the zonular bundles from their first entry between the posterior ciliary processes and minor plicae show considerable variation in distribution, but some general principles are apparent. Most of the bundles destined to insert on the posterior lens capsule become somewhat elevated above the level of the main zonular stream in the middle to anterior pars plana region. The one to three large bundles that pass over the crests of each ciliary process are the most highly elevated, especially over the prominent ciliary processes often found nasally. These bundles usually remain attached to the anterior hyaloid membrane up to the level of the ciliary processes. They then have some attachments to the crests and sides of the processes by direct adhesion or by joining branch bundles from below before passing to the posterior lens capsule at about a 30-degree angle, some still maintaining an attachment to the anterior hyaloid membrane. By the same direct and indirect attachments, the remaining posterior zonules become strongly adherent in the valleys between the ciliary processes or on the sides of the minor plicae, at intervals of 0.2 to 0.4 mm in from the posterior end of the pars plicata. These posterior zonulesin the valleys exit from the ciliary body at an angle of 50 degrees or more (Fig. 14). There is a continual exchange of fibers between bundles throughout their attachments to the valleys and walls of the pars plicata. Fibers from the deeper group of posterior zonules become associated with the anterior hyaloid membrane again only as it lies over their attachments on the lens posterior capsule.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree