Anatomy of the Visual Sensory System
Alfredo A. Sadun
Joel S. Glaser
Swaraj Bose
“Those who have dissected or inspected many, have at least learned to doubt when the others, who are ignorant of anatomy, and do not take the trouble to attend to it, are in no doubt at all.”
—Morgagni GB, The Seeds and Causes of Diseases Investigated by Anatomy, 1761
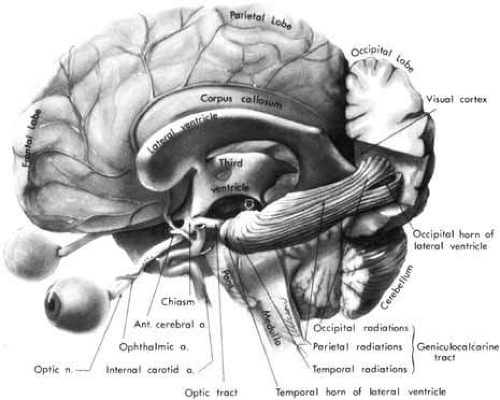
The human primary visual sensory system comprises the retina, optic nerves, optic chiasm, optic tracts, lateral geniculate nuclei, geniculostriate radiations, striate cortex, visual association areas, and related interhemispheral connections. This specialized afferent system lies principally in a horizontal plane that crosses at right angles the major ascending sensory and descending motor systems of the cerebral hemispheres (Fig. 1). The anterior portion of the visual system is intimately related to the vascular and bony structures at the skull base and undersurface of the brain. The posterior portions are closely applied to the lateral aspects of the ventricular system that extend throughout the cerebral hemispheres. Thus, defects of the visual pathways, as revealed by visual field assessment or otherwise, have great localizing value in neurologic diagnosis. The dominant role of vision in humans may be expressed numerically by considering, for example, the number of axons in the human optic nerve (700,000 to 1.4 million) as compared with axons in the acoustic nerve (approximately 31,000).1 Thus, the ratio of afferent neurons in the peripheral visual apparatus to the number in the aural system is roughly 40 to 1.
The first distinct evidence of the human eye is found in the eight somite stage of embryonic development, which occurs at about the 3rd to the 4th week of gestation.2,3 The two primordial optic bulbs extend to either side of the anterior end of the neural tube, the prosencephalon. A slight thickening between the two represents the torus opticus, that is, the primitive chiasmal anlage. The optic primordia evaginate to form the laterally placed cuplike vesicles, which contact overlying surface ectoderm and induce lens growth. The optic stalk and cups are notched ventrally (the “fetal” fissure) to permit entry of blood vessels, and the primitive retina is developing by about 5 weeks of gestation. Retinal ganglion cells differentiate, and optic nerve fibers begin to fill the optic stalk, which is now surrounded by a cellular sheath. These afferent visual neurons reach the area of the chiasm at about the 7th week of gestation as the ventral fissure closes.
At approximately 50 days of gestation, the optic nerve contains a full complement of retinal ganglion cell axons, the optic disc and scleral opening are well defined, and endochondral ossification of the spheroid begins. Actually, the ultimate number of axons in the mature human optic nerve is the result of an initial overproduction of axons during the first half of gestation, followed by a 70% reduction (“die back”) between 16 and 30 weeks of gestation. Myelination of visual axons commences at the chiasm early in the 7th fetal month and progresses distally toward the lamina cribrosa until about the 1st postpartum month.
Retinal ganglion cells direct their axons to the proper regions of specific subcortical visual centers. Despite this remarkable specificity, surgical manipulations in the perinatal period can lead to retinofugal projections to nonvisual structures in hamsters.4 In the monkey, the retinal afferents from each eye overlap in the developing lateral geniculate nucleus.5 In the mouse, the contralateral projections to the lateral geniculate nucleus precede the ipsilateral retinofugal inputs, which may then displace them.6 The relative delay of ipsilateral retinal ganglion cell projections continues until after birth. Thus, a large number of the retinofugal axons that die back are probably of temporal retinal origin and are displaced by nasal fibers from the contralateral retina.
FUNCTIONAL ANATOMYRETINA
The functional organization of the visual sensory system begins at the retina. It is beyond the scope of this chapter to fully describe the complex vertical and horizontal organization of retinal elements, synaptic patterns, receptive field physiology, and other details of visual signal propagation. However, it is important to emphasize that there is a considerable degree of retinal processing that modifies neural signals before transmission to central structures, such as the lateral geniculate nuclei and other subcortical visual areas. Ganglion cell subsets selectively encode specific aspects of visual information, such as acuity, color, image velocity and movement direction, and contrast; thus, visual signal processing takes place simultaneously but in separate “channels.”7 Visual information is already integrated at the retina and arrives at the optic nerve after considerable modulation.
In an excellent review of the primate retina, Rodieck8 described the neuronal circuitry and synaptic organization within the 12 layers of the retina that contain at least 18 specific cell types. In the human retina, Rodieck described four different percipient elements (rods and three types of cones), at least four types of bipolar cells, three types of amacrine cells, three types of horizontal cells, and at least three types of ganglion cells in addition to Muller cells. Humans, as all primates, have a nonlinear distribution of their ganglion cells across the retina.8 Axons from these ganglion cells form bundles in the nerve fiber layer en route to the optic disc. There is, at best, controversial evidence regarding the existence in primates of efferent fibers deriving from the optic nerve to terminate throughout the inner plexiform layer of the retina.9 If this pathway exists, the fibers probably are sympathetics that contact blood vessels.
There is an additional novel photoreceptor in the mammalian retina that is not found in rod or cone photoreceptors. This new photoreceptor is found in ganglion cells that communicate directly with the hypothalamus and serve to synchronize circadian rhythms with the solar day.10,11,12,13 Mice lacking rod and cone photoreceptors still retain many retinal responses to light. This has been shown to be mediated by photosensitive retinal ganglion cells that used a photopigment called melanopsin.10 These cells probably project to the suprachiasmatic nucleus of the hypothalamus that provides for photoentrainment of the circadian rhythm and possibly also for the pupillary light responses.10 Ablation of the melanopsin gene leads to nonexpression of melanopsin in these specialized retinal ganglion cells and subsequent impairments, in mice, of the pupil reflex and light-induced phase delays of the circadian rhythm.11 This system has a much lower sensitivity to light and very poor spatiotemporal resolution.12 Humans who are deprived of this visual input may suffer from disturbances of their diurnal rhythm affecting a number of neuroendocrine functions and behaviors.14
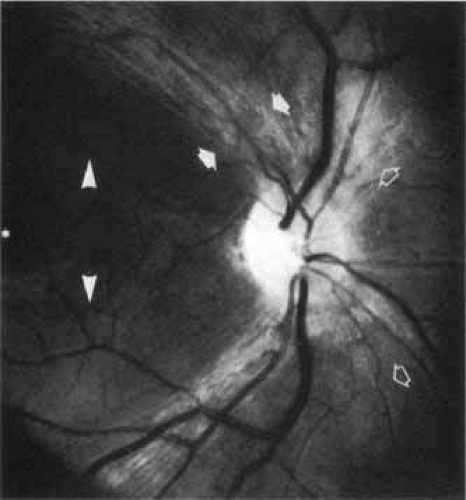
Visual function across the retina is not uniform but takes a pattern of concentric zones that increase in sensitivity toward the central retina area, the fovea, which subserves the highest sensitivity. At progressively eccentric retinal locations there is a near linear decrease in sensitivity (conversely, elevation of thresholds). Ultimately, retinal sensitivity is a manifestation of the underlying cytoarchitecture and the distribution of the percipient elements, the cones and rods. The fovea itself is essentially without rods; it is composed, rather, of about 100,000 compactly arranged slender cones. The entire posterior pole of the retina is dominated by the foveal and parafoveal cone system that occupies an area of approximately 1.5 mm in diameter. Small retinal ganglion cells subserving this central cone cell system send their small caliber axons directly to the temporal aspect of the optic disc, forming the papillomacular nerve fiber bundle. This discrete bundle of nerve fibers is relatively isolated from other retinal nerve fibers that reach the optic disc by an arcuate course above and below the papillomacular bundle, forming dense superior and inferior bands (Fig. 2).
Østerburg15 quantitatively examined the arrangement of photoreceptors in the human retina and found a skewed distribution: rod and cone populations are more dense in the superonasal retina and less dense in the inferotemporal retina. Van Buren16 performed retinal ganglion cell counts and demonstrated the same eccentric pattern, especially in the one-cell-thick layer, which reached nearly twice as far on the nasal side of the fovea as on the temporal side. This asymmetric distribution of retinal elements about the fovea is reflected in the asymmetry of nasal versus temporal visual field and accounts for the relative foreshortening of the nasal periphery.17
In mammals, the inner aspects of cones form pedicles that are wide and flat and act as a platform for synaptic terminations. Each pedicle contains several invaginations that form synaptic contact with horizontal and bipolar neurons. Rods have a smaller synaptic termination at their inner end termed a spherule that contains only one invagination. Horizontal and bipolar cells synapse with pedicles and spherules in the outer plexiform layer. Amacrine and bipolar cells make synaptic contacts in the inner plexiform layer. Ultimately, visual information progresses centripetally to the retinal ganglion cells, whose dendritic processes extend into the inner plexiform layer and whose axons form the retinal nerve fiber layer. Most recently, a sixth class of retinal neuron, the interplexiform cell, has been described in monkeys and humans.18,19 These dopaminergic cells lie in the inner nuclear layer with processes that extend to both the inner and outer plexiform layers, providing an anatomic substrate for an inner to outer (centrifugal) retinal neural signal. In humans, ganglion cells are stacked six to eight cells deep near the fovea but are found only sparsely in the peripheral retina. About 1.3 million retinal ganglion cells (down from two to three times that number prior to birth) contribute axons from 0.5 to 2.5 μm in diameter in the nerve fiber layer.20
Retinal ganglion cells have been classified since 1933, when Bishop suggested, based on electrophysiological studies of the frog, that there were three groups of axons deriving from retinal ganglion cells that conduct at different velocities.21 Morphologically, Hopp had already demonstrated different shapes and sizes to retinal ganglion cells but had not put together a rigid classification scheme. However, Polyak, in 1941, did classify primate ganglion cells of four types, including parasol and midget cells.22,23 In 1966 the work of Enroth-Cugell and Robson24 clearly established the differences between X- and Y-cell types. In 1974, Boycott and Wassle25 were able to provide the corresponding morphologic classes of retinal ganglion cells in the cat.
Enroth-Cugell and Robson also first noted differences in the spatial frequencies with which high-contrast sensitivity was obtained between X cells and Y cells. They noted that the receptive field centers of Y cells were larger than those of X cells and that X cells were more common than Y cells; therefore, they concluded that X cells subserved high-resolution pattern vision whereas Y cells subserved movement vision. Boycott and Wassle described alpha, beta, and gamma cells based on distinctions in some size, dendritic morphology, and axonal caliber. They suggested that the alpha cells corresponded to Y cells, the beta cells to X cells, and the gamma cells to W cells, which had by that time also been proposed by Rodieck.26
There are some differences in retinal ganglion cell classification schemes for primates.27 Two types of morphologically distinct cells appear to project to different divisions of the lateral geniculate nucleus (LGN; the magnocellular and parvocellular layers). This segregation continues through the radiation projections to the cortex. The magnocellular LGN projects to 4C alpha. The parvocellular LGN projects to 4C beta and layers 2 and 3 of area 17 and to the “pale stripe” region of area 18. The segregation is continued beyond the synapses to areas MT, V3, and V4 of the cortex.28 However, as in the cat, there are physiological differences between these systems as well. The parvosystem is characterized by color opponency, high spatial resolution, and low-contrast sensitivity. The magnosystem, however, is characterized by color ignorance, low spatial resolution, fast temporal resolution, stereopsis, and high-contrast sensitivity.29
In humans, Polyak22 believed that there were five or six different types of retinal ganglion cells. However, he used the overall classification scheme of large parasol ganglion cells that project to the magnocellular LGN (more recently referred to as the M-cell system) and smaller midget ganglion cells that project to the parvocellular LGN (P-cell system). Each of these two systems has a characteristic dendritic field diameter and a separate primary projection from LGN to striate cortex.30 However, to make things more complicated, midget, parasol, shrub, blue-cone, biplexiform, garland, small diffuse, giant, and displaced retinal ganglion cells have all been described.8 For many of these cells, differences in dendritic field size and orientation have been worked out.31
In advanced primates, the fovea occupies the central 3° of retina and is a roughly circular pit devoid of ganglion cells and surrounded by a multilayered annulus of densely packed small ganglion cells. Central projections, which have been traced by horseradish peroxidase, indicate that ipsilaterally projecting ganglion cells in the temporal foveal rim can generate 2° to 3° of bilateral representation in the geniculocortical pathways, because of intermingling with contralaterally projecting cells on the nasal side of the foveal pit.32 These findings provide a potential retinal neural basis for foveal (fixational) “sparing” or “splitting” that is demonstrated by perimetry in the presence of lesions of the posterior visual pathways.
Ogden33 extensively studied horizontal and vertical retinotopy in the geographic bundles of ganglion cell axons as they traverse the retina to the optic disc. Despite previous reports that the nerve fiber layer is well organized, Ogden demonstrated that horizontal retinotopic organization is present within nasal, but not temporal, nerve fiber bundles and that vertical retinotopic stratification is orderly within temporal, but not nasal, retinal bundles.28 Additionally, temporal axons in the nerve fiber layer intermingle freely along the intraretinal course of the arcuate bundles. Therefore, the segregation of fibers to conform to a retinotopic distribution must occur at the optic disc or more posteriorly in the optic nerve. Species differences do occur, with the macaque retina apparently most resembling the human nerve fiber layer.
Retinal ganglion cells (RGCs) in the mammalian retina are probably coupled by gap junctions.34 This may have important physiological significance in the integration and synchronization of signaling. That is, electrical coupling via gap junctions may explain the short-latency concerted spike activity of neighboring RGCs.34,35 Furthermore, such gap junctions, by allowing electrolyte and reactive oxygen species transmission, may permit the propagation of proapoptotic chemical messengers.36
OPTIC DISC
The optic nerve head can be seen funduscopically end on and appears as a flat disc with a central depression of variable depth, which is called the optic cup. Papilla, a term that implies a nipplelike eminence, is used less frequently. The disc is the collective exit site of all retinal ganglion cell axons (i.e., the nerve fiber layer). The optic disc is located 3 to 4 mm nasal to the fovea and represents a 1.5 × 2.0-mm hiatus in the sclera, choroid, retinal pigment epithelium, and retina proper. Mean horizontal disc diameter is 1.76 ± 0.3 mm, mean vertical diameter is 1.92 ± 0.3 mm, mean horizontal cup to disc ratio is 0.39, and mean vertical cup to disc ratio is 0.34. The number of nerve fibers appears to be positively correlated with the size of the optic nerve head–larger discs have relatively more fibers than smaller discs. Smaller discs may demonstrate optic nerve head crowding.37,38 Fiber number decreases with age.39 There are no percipient or retinal elements on the disc, which is represented in visual space as an absolute scotoma, the blind spot of Mariotte.
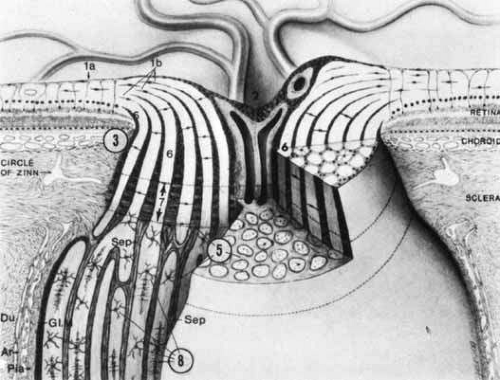
The retinal axons turn 90° over the scleral disc margin and pass through the perforations of the lamina cribrosa. This fenestrated connective tissue is lined by astrocytes, is continuous with surrounding sclera, and partitions the nerve head into prelaminar, laminar, and retrolaminar compartments (Fig. 3). The extracellular matrix of the human lamina cribrosa contains collagen macromolecules resembling central nervous system (CNS) basement membrane and does not resemble sclera in this regard.40 Axonal bundles are increasingly compartmentalized by glial–collagen pial septa as they traverse the lamina cribrosa into the distal portion of the optic nerve. The increase in interaxonal glial tissue density could play a possible role in the propagation of spread of edema caused by ischemic optic neuropathies or when intraocular pressure is raised.41
OPTIC NERVE
The optic nerve may be regarded as consisting of four segments: intraocular (1 mm), intraorbital (25 to 30 mm), intracanalicular (9 to 10 mm), and intracranial (about 16 mm). Thus, the entire length of the optic nerve from the globe to the optic chiasm is 5 to 6 cm. The intraocular portion (optic disc) may be further divided into retinal, choroidal, and scleral levels as the retinal ganglion cell axons from the nerve fiber layer turn sharply posteriorly to exit the globe (see Fig. 3). Anderson40,41,42,43,44,45 and Minckler41 extensively described the microanatomical structure of the nerve as consisting of a neuroectodermal (nervous tissue proper) and a mesodermal component (providing support and nourishment). Mesodermal connective tissue includes the sclera, fibroblasts, meningothelial cells, and blood vessels. The collagenous component of the perforated lamina (also termed the lamina cribrosa scleralis) may be considered continuous, but not identical, with the sclera and also with the perioptic meninges. The more anterior aspects of the optic nerve head contain less connective tissue, with glial components predominating. The anterior optic nerve head consists of unmyelinated axons and astrocytes. It is unusual for oligodendrocytes or myelinated axons to be anterior to the laminar scleralis; funduscopically visible myelinated (medullated) nerve fibers are associated with anomalous rests of oligodendrocytes.
Optic nerve myelination terminates at the lamina cribrosa. What is the basis of the barrier that precludes oligodendrocytes and their myelin product from going anterior to the lamina cribrosa? In the adult human optic nerve, astrocytic processes form a dense mesh through which only unmyelinated fibers pass.46 This astrocytic framework appears in the perinatal period and before oligodendrocytes have migrated from posterior side of the lamina cribrosa.46 Hence, specialized astrocytes may express inhibitory and adhesion molecules that may delimit myelin production in the eye.47
Behind the lamina cribrosa, the optic nerve abruptly increases in diameter from 3 to 4 mm. At this point, oligodendrocytes (responsible for the formation of myelin that ensheathes the axons) constitute approximately two-thirds of the interstitial cells. In peripheral nerves, Schwann cells serve this same function. Therefore, the optic nerve must be considered analogous to white matter tracts of the brain rather than to peripheral nerves; this composition makes the optic nerve susceptible to diseases of CNS tracts, such as multiple sclerosis. The orbital and intracanalicular portions of the optic nerve contain a well-developed septal system derived from pia mater. The septa form porous cylindrical walls aligned along the long axis of the nerves and may contribute to flexible movement.48 The septa divide the nerve fibers into parallel columns of variable shape and size. Astrocytes are intimately related to the pial septa and play a role in the support and nutrition of axons.
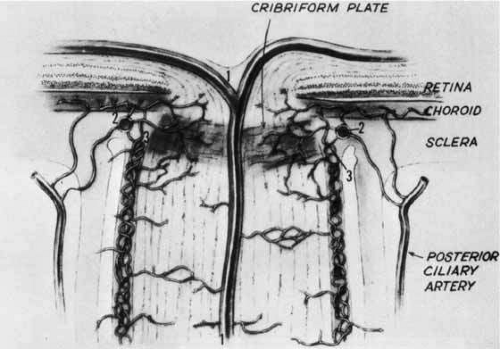
Approximately 1 cm posterior to the globe, a major branch of the ophthalmic artery pierces the inferior aspect of the meninges of the optic nerve, gains a central axial position, and emerges in the middle of the optic disc as the central retinal artery. The central retinal artery does not contribute significantly to the blood supply of the laminar and prelaminar portions of the optic nerve head. These areas of the optic nerve are supplied by an anastomotic arterial complex called the circle of Zinn-Haller. This structure receives contributions from the posterior ciliary arteries, the pial arterial network, and the peripapillary choroid (Fig. 4). Thus, the blood supply of the optic nerve head is derived primarily from choroidal and posterior ciliary vessels, as opposed to that of the retina, which is mediated by the central retinal artery.49,50
Ultra-high resolution MRI allows for a three-dimensional appreciation of the structures in the human optic nerve head. With this methodology, it is possible to see that the circle of Zinn-Haller is, in fact, a continuum of small arterial segments, in slightly different planes and axes, that join to form a complete circle.51
The intraorbital segment of the optic nerve (about 25 mm) exceeds the distance from the back of the globe to the orbital apex (less than 20 mm). Therefore, within the orbit, the optic nerve has redundancy in length and a sinuous course; this permits the nerve to move freely behind the globe during eye movements and also allows up to 6 or 8 mm of proptosis before the nerve begins to tether the back of the globe. At the orbital apex, the optic nerve enters the bony optic canal and is surrounded by the connective tissue origins of the superior, medial, and inferior recti muscles, which collectively constitute the so-called annulus of Zinn.
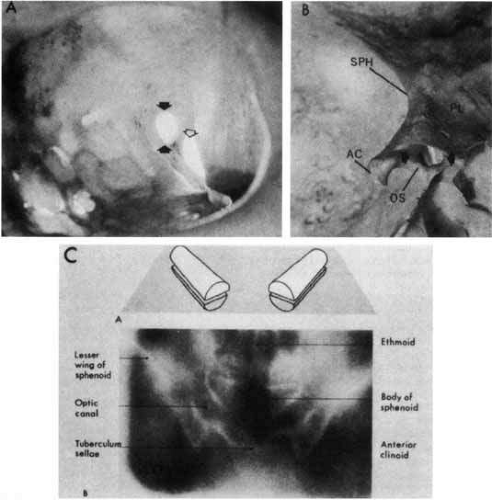
The optic canal runs posteromedially in the spheroid bone, at an angle of approximately 35° with the midsagittal plane (Fig. 5). The optic canal is 4 to 10 mm in length and contains not only the optic nerve but also the ophthalmic artery, branches of the carotid sympathetic plexus, and extensions of the intracranial meninges that form the sheaths of the optic nerve. The aural covering of the nerve and the periosteum of the canal are fused, but the arachnoid is continuous, permitting the subarachnoid space of the optic nerves to communicate freely with the intracranial subarachnoid space, both of which contain cerebrospinal fluid.
The mesial surface of the optic canal protrudes into the superolateral aspect of the spheroid sinus, and according to Fujii and colleagues,52 the optic nerves are separated from the sinus cavity by only the nerve sheath and mucosa in some 4% of specimens; in 78%, less than a 0.5-mm thickness of bone separates the optic nerves from the sinus cavity. Manipulations at the lateral spheroid sinus wall during transsphenoidal surgical procedures may damage the optic nerves.
The optic nerves are fixed at the intracranial opening of the optic canals, the upper margins of which are formed by an unyielding falciform fold of dura. This constriction may notch the superior surface of the optic nerve when sellar-based adenomas, or internal carotid artery aneurysms, elevate the chiasm. From the internal (posterior) foramina of the canals, the optic nerves converge toward the chiasm in the anteroinferior floor of the third ventricle. The two nerves ascend toward the chiasm at an angle of approximately 45° with the nasotuberculum line (Fig. 6); the intracranial nerve segment averages 17 ± 2.4 mm in length, so that the optic chiasm itself sits 10.7 ± 2.4 mm above the dorsum of the sella turcica.50 Occasionally, the intracranial optic nerves are shorter, and the chiasm may lie directly above the sella in a position that is called prefixed. More commonly, the optic chiasm is positioned 10 mm above the insertion of the diaphragma sellae onto the dorsum.53 This being the case, it should be understood that pituitary tumors must extend well above the sella before the optic chiasm is encroached upon. By the time chiasmal field defects can be found, pituitary tumors are already large and have major suprasellar extensions. Small tumors are detected clinically when signs of unilateral optic nerve compression evolve.
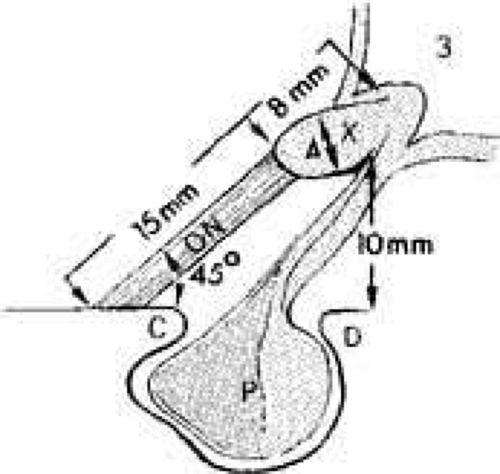 Fig. 6 Relationships of the optic nerves (ON) and chiasm (X) to the sellar structures and third ventricle (3). C, anterior clinoid; D, dorsum sellae; P, pituitary gland in sella. |
The anterior perforated substance, the root of the olfactory tract, and the anterior cerebral artery lie superior to the optic nerve in its intracranial path. The internal carotid artery is below and then lateral to the nerve, and the ophthalmic artery enters the optic canal within the dural sheath of the nerve. The inferior surfaces of the frontal lobes gyrus recti of the cerebral hemispheres are above the optic nerves. The anterior cerebral and anterior communicating arteries lie between the frontal lobes and the optic nerves (see Fig. 1). Medial to the anterior clinoid process (see Fig. 5B), the optic nerve lies just above the siphon of the intracavernous portion of the internal carotid artery and is separated from the cavernous sinus by the optic strut. Thus, expanding lesions of the cavernous sinus, such as aneurysm or meningioma, may impinge on the optic nerve. At the origin of the ophthalmic artery, aneurysms may compress the nerve from a medial and ventral direction.