Adenotonsillar disease usually presents in children with either recurrent throat infections or sleep-disordered breathing (SDB). Twenty years ago, recurrent tonsillitis was the most common indication for tonsillectomy. Presently, the most common indication for tonsillectomy and adenoidectomy (T&A) is SDB. This shift comes as a result of an evolving understanding of the impact of SDB on childhood health and well-being and a renewed focus on ensuring evidence-based indications for surgical intervention.
Untreated SDB can negatively impact a child’s quality of life, behavior, and school performance (
1,
2,
3). Similarly, children with recurrent throat infections have more bodily pain and poorer general health and physical functioning compared to healthy children (
4). In the appropriately selected child, T&A can eliminate or improve the morbidity of SDB and recurrent tonsillitis.
This chapter reviews the current knowledge of the clinical presentation and management of adenotonsillar disease in children.
EPIDEMIOLOGY
In 2006, the most recent data available, 530,000 tonsillectomies were performed in children younger than 15 years of age in the United States (
5). The only procedure performed with greater frequency was myringotomy with tube insertion, estimated at about 667,000 in the same year (
5).
In the early and midportion of the 20th century, T&A was the most common surgical procedure in the United States, with the indication primarily being recurrent throat infections (
6). Secondary to a public backlash and scrutiny of tonsillectomy for unscrupulous indications and unproven efficacy in the late 1970s and early 1980s, there was a decline by about 50% to 75% in the number of T&A procedures performed. However, since then, the frequency of T&A has almost doubled, with the most common indication at present being SDB as opposed to recurrent throat infections (
6,
7,
8). The incidence of SDB is known to peak during early childhood and to affect 1% to 4% of children (
9). In addition, older children and young adults are more frequently being treated for SDB with T&A (
10,
11).
EMBRYOLOGY
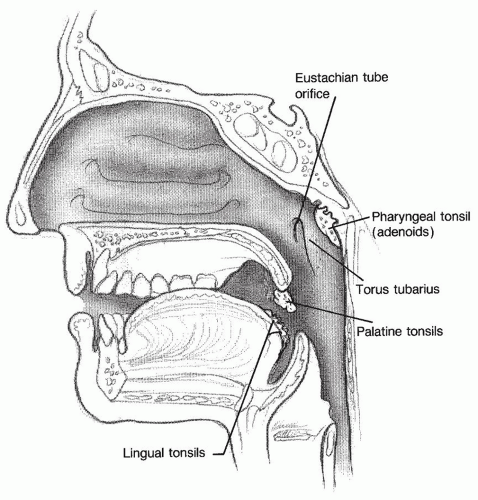
Tonsils are lymphoid organs that develop from components of surface epithelium enveloped by mesenchymal stroma and subsequently populated by lymphocytes and immunologic mediators. Adenoids, also known as the pharyngeal tonsils, develop in close approximation to mucous glands along the posterior surface of the nasopharynx.
The basic structure of the adenoids is characterized by multiple shallow sagittal folds and plicae covered by respiratory epithelium. Invaginations and ingrowth of endoderm followed by programmed cell death creates the characteristic crypts of tonsillar tissue. Lymphoid follicles develop around these crypts and adjacent glandular ducts. Crypts begin to develop at 3 months of gestation and are fully developed by the seventh month. Immunoglobulin populations have been found present in embryonal adenoid tissue. The adenoids will often increase in size until the sixth to seventh year of life at which point they begin to diminish and subsequently atrophy by early puberty. The opening of the Eustachian tube is also variably lined by lymphoid tissue, the tubal tonsils (
12).
The palatine tonsils derive from the endoderm of the ventral portion of the second pharyngeal pouch. This epithelium, in a similar manner to the adenoids, invaginates and branches within the surrounding mesenchyme and ultimately undergoes apoptosis to produce characteristic crypts. The capsule of the tonsil develops from a condensation of mesenchyme. Unlike the adenoids, the palatine tonsils are covered by squamous epithelium. Lymphocytes appear in the reticulum by the third month of gestation and become arranged as lymphatic nodules. Lymphocyte
proliferation and maturation then takes place within the surrounding mesenchyme. Definitive lymphoid organization around these crypts can be recognized by the final trimester of development (
12).
The lingual tonsils develop along the posterior region of the base of tongue. This area is covered by mucous glands, which are subsequently infiltrated with lymphoid cells during the fifth month of development. Crypts of the lingual tonsils appear at birth and are more shallow and less complicated than their palatine counterparts (
12).
Taken as a whole, the adenoids and tubal, palatine, and lingual tonsils form an anatomic arrangement, which is collectively known as Waldeyer ring (
Fig. 95.1). The adenoids and tubal tonsils form the posterosuperior extent of the ring, the palatine tonsils provide the lateral portion, and the lingual tonsils at the base of the tongue provide the anteroinferior segment. The tissue of each portion has similar histologic appearance and function. Waldeyer ring serves as a component of the mucosa-associated lymphoid tissue for the upper aerodigestive tract.
Table 95.1 summarized the distinctive properties of each component of Waldeyer ring.
The adenoid tissue lies at the superior aspect of the nasopharynx, extending laterally to the Eustachian tubes and the walls of the pharyngeal recess and lying in general approximation to the tubal tonsils, which may be present and surround the opening of the Eustachian tube. The blood supply of the adenoids is from the pharyngeal branch of the external carotid artery with some minor supply from the internal maxillary and facial arteries. Sensory innervation is provided by the glossopharyngeal and vagus nerves. The general shape of the adenoid tissue is that of a loosely defined pyramid with the adenoid apex pointed toward the nasal septum and base toward the posterior wall of the nasopharynx (
12).
The palatine tonsils lie at the entrance to the oropharynx between the palatoglossus muscle, also known as the anterior pillar, and the palatopharyngeus muscle, or posterior pillar. The superior constrictor fascia serves as the lateral margin of the tonsillar fossa. The palatine tonsils are the only portion of Waldeyer ring with a discrete capsule and are the largest tonsils within the ring. The tonsillar capsule is a specialized portion of the pharyngobasilar fascia that covers the surface of the tonsil and extends into it to form septa that conduct the nerves and vessels. This capsule adheres to the surrounding fascia by loose connective tissue, forming a relatively avascular plane which is taken advantage of during tonsillectomy. During times of hypertrophy, the palatine tonsils may extend in a more caudal direction to become nearly confluent with the base of tongue and lingual tonsil tissue. Immediately lateral to the superior constrictor is the glossopharyngeal nerve, which may be injured during tonsillectomy. In cadaveric studies (
13), the lingual branch of the nerve was shown to be just lateral and in about a quarter of cases adherent to the tonsillar capsule itself. Pain and dysgeusia are potential sequelae from injury to this nerve.
The blood supply to the palatine tonsils is variable, but in general, they are supplied by several branches of the external carotid artery: ascending pharyngeal, ascending palatine, and branches of the lingual and facial arteries. The blood supply enters from the lower portion of the palatine tonsil pole. The internal carotid artery (ICA) lies approximately 2 to 2.5 cm deep and posterolateral to the palatine tonsil; however, case reports (
14) exist of aberrant ICA courses which come within 1 cm of the inferior pole. The ICA may have a tortuous and convoluted course of which the surgeon must be cognizant. Venous drainage is by way of a peritonsillar venous plexus, which surrounds the capsule and drains into the lingual and pharyngeal veins (
12).
The lymphatic drainage of the tonsils is primarily to the superior deep cervical and jugular lymph nodes, which may become tender and inflamed during times of infection. The sensory innervation is from the glossopharyngeal nerve and some branches of the lesser palatine nerve via the sphenopalatine ganglion.
The histologic structure of the tonsils is closely related to its function as an immunologic organ. The epithelium of the crypt system acts as the immune system access route for inhaled and ingested antigens. These crypts are lined by a system of antigen-presenting cells that deliver the antigen to the immunologically active lymphoid cells in the tonsillar stroma.
IMMUNOPHYSIOLOGY
The tonsils and adenoids play an integral role in host defense, local immunity, and immune surveillance against invading antigens of the aerodigestive tract. Because the tonsils and adenoids do not have afferent lymphatic channels like other secondary lymph tissues, they are fed by surface epithelia. The aforementioned crypts serve to increase the surface area and effectively trap foreign material, both ingested and inhaled. These crypts are surrounded by numerous aggregates of cellular mediators of the immunologic system. Aggregates of B lymphocytes with more sparsely intervening zones of T lymphocytes and a diverse population of antigen presenting cells (macrophages, B cells, dendritic cells) are found in abundance within the subepithelial tissue of the tonsils (
15).
These tissues survey the locally presented antigenic stimuli, destroy them, and generate effector and memory lymphocytes to be disseminated to other mucosal sites. They serve as an inductive site for local and distant antibody responses and generate immunologic memory independent of the systemic immune system. This function is most active during childhood and declines with age (
16,
17).
Microscopically, the adenoids have three types of surface epithelium: ciliated pseudostratified columnar, stratified squamous, and transitional epithelium. Chronically infected adenoids tend to have a predominance of squamous epithelium. One study demonstrated a significant transition trend to squamous metaplasia in chronically affected adenoid tissue (
18).
Given the role in immunologic surveillance, some have questioned the impact of T&A on immunologic integrity. The results of several studies indicate that such interventions do not compromise the immune functions of children. The immunity of children after T&A has been shown to recover compared to their preoperative immune status, and to be similar to their age-matched healthy controls when the surgery is performed in young children and in late adolescence (
19). Other studies have concluded that the effect of removal is essentially “clinically insignificant” and argue that no increase in the frequency of immunomodulated diseases is to be expected (
20).
CLINICAL PRESENTATION
Adenotonsillar disease is characterized by either infections involving the lymphoid tissue, or hypertrophy or a combination of both. The disease processes can, and often will, affect the adenoids, tonsils, or both. Less often, adenotonsillar disease can be neoplastic.
Table 95.2 provides a broad overview of adenotonsillar disease.
Infections
Viruses
Viruses play an important role as initiators of mucosal inflammation, crypt obstruction, and ulceration leading to bacterial superinfection. The clinical presentation of viral tonsillopharyngitis is similar to that of bacterial infection, but often with less severe symptoms, including sore throat, odynophagia, otalgia, headache, and fever. There is often tonsillar erythema and enlargement with or without exudates. The implicated viruses are the same as those involved in a variety of upper respiratory illness: rhinovirus, coronavirus, adenovirus, herpes simplex virus, parainfluenza virus, Epstein-Barr virus, and cytomegalovirus (
21).
Of special note is herpangina caused by coxsackievirus. Often affecting young children, this disease may present as small vesicles with erythematous bases that eventually ulcerate and are typically present on the anterior tonsillar pillars, palate, and posterior pharyngeal wall.
Acute infections with Epstein-Barr virus may present as tonsillopharyngitis with dramatic tonsillar hypertrophy, sore throat, fever, cervical and diffuse lymphadenopathy, palatal petechiae, and malaise (
21). The diagnosis is confirmed on the basis of clinical presentation, greater than 10% atypical lymphocytes on a peripheral blood smear, and a positive monospot (heterophile antibody) test (
22). Care should be taken to not inadvertently prescribe amoxicillin or ampicillin as the risk of morbilliform rashes can approach 95% with this treatment (
22).
Bacteriology/Microbiology
Any discussion of adenotonsillar infectious disease has to include several important observations. First, most infections can be linked to the presence of beta-lactamase producing organisms that distort the normal aerodigestive bacterial milieu and can cause commensal organisms to become pathogenic. Second, infections are often polymicrobial in nature and often include anaerobic pathogens. Third, obstruction of tonsillar crypts can serve as a nidus for bacterial stasis and can further perpetuate a condition of chronic infection, suppuration, and fistulae (
23).
Studies of the bacteriology of adenoid tissue obtained from pathologic postoperative specimens demonstrate the presence of several aerobic species: alpha- and gamma-hemolytic streptococci,
Haemophilus influenzae, Staphylococcus aureus, group A beta-hemolytic streptococci (GABA), and
Moraxella catarrhalis. Anaerobic species noted include members of
Peptostreptococcus, Prevotella, and
Fusobacterium. Similar findings have been confirmed in tonsil isolates as well (
24,
25). Due to these findings and the previously mentioned premises, one must conclude that surface cultures have limited utility for identifying the causative organism, especially in chronic inflammation.
Generally, most authors attribute 15% to 30% of bacterial cases of acute tonsillopharyngitis to GABA, with a varying contribution from other pathogens, including group C beta-hemolytic streptococci,
Neisseria gonorrhoeae, Corynebacterium diphtheria, Chlamydia pneumoniae, and
Mycoplasma pneumoniae (
21). Studies have shown that the presence of tonsillopharyngeal exudate and anterior cervical lymphadenitis increase the likelihood that the infectious agent is group A streptococcus (
26).
The chronicity of adenotonsillar disease may be attributed to multiple overlapping infections caused by different pathogens within fistula and purulent pockets of the tonsil parenchyma even in the absence of clinical symptoms (
23).
Hypertrophy
Although we distinguish between hypertrophy and infectious clinical presentations, it is true that most adenotonsillar hypertrophy is a result of low grade or chronic infectious disease of the involved tissues. Studies support a bacterial etiology for tonsillar hypertrophy (
27). Some authors have argued for an etiologic role for
H. influenzae in the pathogenesis of tonsillar hypertrophy in children (
28). Adenoid hyperplasia has been demonstrated to be directly proportional to the number of lymphocytes and the bacterial load of the tissue (
28). For the purposes of
this discussion, we will consider hypertrophy as a separate clinical entity, one which does not present with the signs and symptoms of acute or chronic infection—erythema, tenderness, lymphadenopathy, or fever. In practice, many children may have components of both infectious and hyperplastic morbidity.
Adenoid hypertrophy is a common cause of nasal obstruction in children. It can present as chronic or recurrent nasal discharge, snoring, SDB, recurrent otitis media, or Eustachian tube dysfunction (
18). Physical examination often elicits a history of mouth breathing, hyponasal voice, and the classic “adenoid facies,” characterized by an incompetent lip seal, a narrow upper dental arch, increased anterior face height, a steep mandibular plane angle, and a retrognathic mandible. This development occurs as the result in the changes in head and tongue position and muscular balance secondary to the open mouth breathing that accompanies nasopharyngeal obstruction (
29). Recent studies confirm that there are changes in facial growth and development among children with adenoid hypertrophy. These changes are characterized by increased total and inferior anterior heights of the face, as well as more anterior and inferior position of the hyoid bone (
30).
Tonsillar hypertrophy can present with dysphagia, SDB at night with gasping for breath, voice changes, and dental malocclusion. Children may even provide a subjective experience of having difficulty “getting food down” or the sensation of oropharyngeal fullness.
The presentation of new onset unilateral tonsillar hypertrophy is most likely due to infection, but the differential should include malignancy. Sudden enlargement of Waldeyer ring should raise suspicion for lymphoma, but actual use of asymmetric hypertrophy without concomitant symptomatology is often of poor predictive value (
31). Unusual infections such as atypical mycobacteria, actinomycosis, and lymphoproliferative disease in solid organ transplant children need to be considered with asymmetric enlargement.
CLINICAL EVALUATION
Adenoids
Obstructive adenoid hyperplasia is diagnosed by clinical evaluation. Mouth breathing, snoring, and hyponasal speech are common presenting complaints. Rhinorrhea, postnasal drip, and chronic cough are common and nonspecific findings. The aforementioned adenoid facies may be present. Assessment of hyponasality is accomplished by having the child repeat words that emphasize nasal emission, which are phonemes using the “m,” “n,” or “ng” sound, such as milkman or mickey mouse and comparing them to words that do not, such as baseball or phrases such as “patty ate apple pie” (
32).
Differentiating acute and chronic infections from sinonasal disease can be challenging. Often, acute and chronic adenoiditis can be confused and misdiagnosed as “sinusitis.” Most children under the age of 6 will have effective resolution of these symptoms with adenoidectomy (
33). Parents should be made aware that if symptoms persist after adenoidectomy, further investigation and treatment of sinonasal disease may be necessary to fully address the appropriate etiology.
A thorough physical exam includes at a minimum anterior rhinoscopy, which is easily facilitated in the cooperative child, with the use of an otoscope and large ear speculum. This allows the clinician to distinguish possible sources of nasal obstruction within the anterior nasal cavity, such as turbinate hypertrophy, edematous mucosa, or foreign body and to differentiate these from sources within the nasopharynx.
Palatal evaluation should also be undertaken, to ascertain the presence of submucous clefting, bifid uvula, or history of nasal regurgitation. This diminishes the possible complication of postoperative velopharyngeal insufficiency. Children with neuromuscular and central nervous system problems are also at increased risk for velopharyngeal insufficiency following adenoidectomy (
32,
34).
Evaluation of the adenoids in an uncooperative child can be difficult and can be accomplished using lateral neck radiographs, although these are often superfluous compliments to physical exam and history. More recently, an increasing number of children have been evaluated using office nasopharyngoscopy. This technique allows for direct visualization without radiation exposure. Nasopharyngoscopy offers an excellent view of the adenoids and adjacent structures. Parikh et al. (
35) have proposed a grading system for endoscopic examination of adenoid hypertrophy that is validated and user-friendly. Other methods such as video fluoroscopy have been shown to be effective at assessing adenoid size but are less commonly used and carry unnecessary radiation exposure (
36).
Tonsils
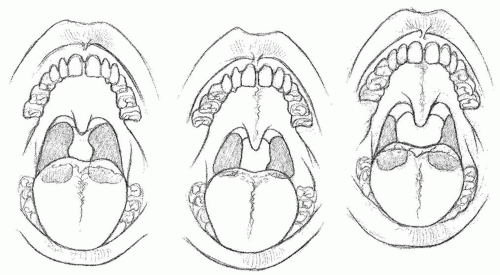
Physical examination should be performed with the child’s mouth open and the tongue on the floor of the mouth, within the oral cavity. A tongue depressor should be used to depress the tongue with gentle and firm pressure along the oral tongue. Gagging and tongue extrusion will cause the tonsils to medialize and can produce the appearance of false enlargement. Having the child say “aah” will allow assessment of palatal integrity and motion. Tonsillar size may be measured on a scale of 1 to 4 with 1 (0% to 25% airway obstruction), 2 (25% to 50% airway obstruction), 3 (50% to 75% airway obstruction), and 4 (75% to 100% airway obstruction) (
37). By convention, this grading scale is denoted as 1+ to 4+ (
Fig. 95.2). Tonsillar hypertrophy is usually defined as falling within the 3+ to 4+ range.
The otolaryngologist must be cognizant of signs of chronic disease, such as peritonsillar erythema, tender
cervical lymphadenopathy, tonsilloliths, smooth glistening tonsils, or excessively cryptic tonsils.
Of note, children at particular risk for the development of SDB secondary to tonsillar hypertrophy include those with craniofacial anomalies, Down syndrome, obesity, and neuromuscular or CNS abnormalities. These children may have symptoms not necessarily predicted by clinical presentation or tonsillar size.