National Health Interview Survey, over 29.3 million adults in the United States had been diagnosed with rhinosinusitis by a physician (5). This comprises approximately 12.6% of the United States population. It has been previously reported that the prevalence of rhinosinusitis is almost twice as common in women (20.9%) as in men (11.6%) (5,6). Other demographic factors, such as geography and race, may play a role as well. The frequency of rhinosinusitis is generally lower in the Western United States (12.1%), as compared to Southern regions (19.5%) (5). Non-Hispanic white persons (17.5%) and non-Hispanic black persons (15.7%) have approximately twice the prevalence of rhinosinusitis as Hispanics (8.6%) (5,6). While prior survey data have not demonstrated that poverty status is relevant to the frequency of rhinosinusitis, regional variation in the frequency of rhinosinusitis may potentially be attributable to differences in air quality, including presence of pollutants and allergens (5,6).
TABLE 33.1 PREDISPOSING FACTORS AND CONTRIBUTORS TO THE DEVELOPMENT OF RHINOSINUSITIS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
TABLE 33.2 TEMPORAL CLASSIFICATION OF RHINOSINUSITIS | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
and infection. Inflammation is a cascade of processes and signals via which mediators, such as leukocytes, are enabled to eliminate foreign agents and repair damaged tissues (4,11). Acute inflammation is characterized by exudation of fluid and plasma proteins from blood vessels with emigration of leukocytes, predominantly neutrophils, as these cells gather to combat the offending entity (4,11). It can be identified within minutes to hours of an insult or trigger. Chronic inflammation develops as the condition persists over weeks to months. The hallmarks of chronic inflammation are the presence of lymphocytes, macrophages, eosinophils, and basophils, along with increased vascularity, fibrosis, and tissue necrosis (4,11). Subacute inflammation is described as the interval period when overlapping patterns of inflammation are observed (4). Infection is traditionally diagnosed when microorganisms present in a host, directly interacting with the host tissue and replicating. This results in expression of disease in the host organism (4). Bacterial infection is characterized by the presence of one or more bacteria per high density field, which correlates to at least 1,000 colony forming units per milliliter (4).
TABLE 33.3 RELATIVE FREQUENCY OF INFECTIOUS ORGANISMS IN ACUTE RHINOSINUSITIS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
by inflammatory mediators in the setting of acute viral infection (4).
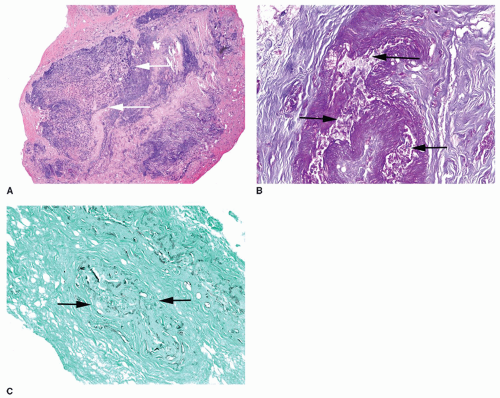
silver stain showing “hyphal forms within the submucosa with or without angiocentric invasion and tissue necrosis with minimal host inflammatory infiltrate” (40) (Fig. 33.1A-C). Because prompt diagnosis is imperative with this condition, frozen section with histopathologic evaluation of any suspicious lesions is necessary to facilitate surgical resection and initiation of antifungal therapy without delay. The potassium hydroxide-calcofluor white method is an alternative rapid diagnostic technique in which potassium hydroxide is used to dissolve human material, and the calcofluor white is used as an optic brightener that binds to the cell wall of the hyphae and fluoresces when viewed with a fluorescent microscope (40).
symptoms: purulent nasal discharge, nasal obstruction, and/or facial pain, pressure, or fullness (2,42) (Table 33.4). The logic behind this new slant in symptom interpretation for the diagnosis of ABRS is based upon the high sensitivity and relatively high specificity of these indicators for ABRS, especially when they are present for 10 days or longer (2,42,43,44). Presence of purulent nasal drainage, whether reported by the patient or seen on physical exam in the posterior pharynx or intranasally near the sinus ostia, correlates to the presence of bacteria on antral aspiration (2,45,46). Findings of purulence also correlate with radiographic evidence of ABRS (2,47). Nasal obstruction is associated with objective measures, such as rhinomanometry or nasal peak flow rate (2,48). Facial pain and dental pain are also predictive of ABRS according to culture results and to radiographic findings, but the location does not correlate with the specific sinuses involved (2,44,45,49).
TABLE 33.4 THREE CARDINAL SYMPTOMS TO DIAGNOSE ABRS | |
|---|---|
|
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree