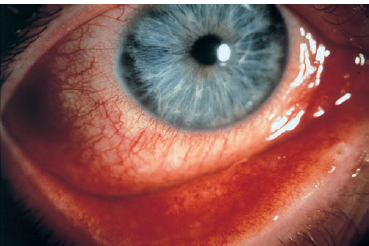
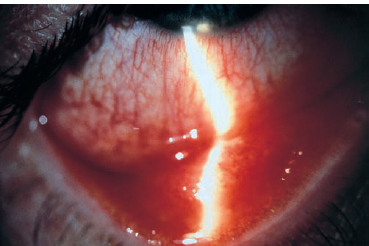
Chapter 6 Acute Conjunctivitis Acute conjunctivitis is an inflammatory conditon of the conjunctiva of less than 4 weeks duration. It is a very common and often mild disorder, but some forms can leave functional sequelae in the absence of specific treatment. Most cases of acute conjunctivitis are of infectious or allergic origin. The diagnosis of infectious conjunctivitis is often based on a combination of signs and symptoms; laboratory investigations are rarely necessary. Fig. 6.1 Epidemic keratoconjunctivitis Fig. 6.2 Epidemic keratoconjunctivitis: follicular conjunctivitis Conjunctival defenses against infectious organisms are ensured by the mechanical palpebral and epithelial barriers, the flushing action of the tears, the commensal conjunctival flora, the immune system, and the antibacterial components of the tear film, such as mucus (which reduces bacterial adhesion to the conjunctival epithelium), lysozyme, (3-lysin, lactoferrin, and secretory IgA1. The commensal bacterial flora also limits colonization of the conjunctiva by pathogenic microorganisms. It is mainly composed of Staphylococcus epidermidis or S. aureus, corynebacteria, Propionibacterium acnes, viridans streptococci, and occasionally pneumococci or hemolytic streptococci2. These bacteria are rarely responsible for ocular surface infections. The patient’s history is very helpful for making the diagnosis. Acute conjunctivitis starts with a foreign body sensation, tingling, burning, pruritus, photophobia, tearing, and discharge. Ocular pain and decreased vision point to a corneal complication. The bilateral or unilateral nature of acute conjunctivitis has little diagnostic value. Nonetheless, bacterial conjunctivitis usually starts as a bilateral condition, while viral conjunctivitis starts as a unilateral condition. Discharge is a consistent feature. The secretions are serous, mucopurulent, or purulent. They sometimes lead to the formation of pseudomembranes. The nature of the secretions is not sufficient to establish a diagnosis. Conjunctival hyperemia is another consistent feature. It is sometimes diffuse, but never predominates around the limbus. It is blanched by topical application of vasoconstrictors such as phenylephrine. It can be masked by chemosis, eyelid edema, or ptosis. Conjunctival inflammation is characterized by the presence of papillae and/or follicles. Papillae are small bumps (« 0.1 mm) centered by a dilated terminal vessel. Their hyperplasia reflects a nonspecific inflammatory reaction with edema and cellular infiltration. They range in size from a pinhead to a cobblestone (> 1 mm). In most cases of bacterial and allergic conjunctivitis they can be seen on the superior tarsal conjunctiva after flipping the lid. Follicles are whitish nodules with vascularized edges that range in size from 0.5-2.0 mm. They are seen on the medial canthus and lower fornix, where the conjunctiva is less adherent. They correspond to a hyperplasia of lymphoid follicles of the conjunctival chorion. They are often associated with papillae and are frequent in viral conjunctivitis (adenovirus, herpesvirus, enterovirus 70, coxsackie A24, poxvirus), toxic conjunctivitis, chlamydiosis, and Parinaud’s oculoglandular syndrome (cat-scratch disease). Membranous conjunctivitis formation is now rare since the virtual eradication of diphtheria (Table 6.1). After severe bacterial conjunctivitis (streptococci, pneumococci) or viral conjunctivitis (adenovirus, herpesviruses), conjunctival epithelial necrosis leads to the formation of a yellowish avascular fibrin plaque or membrane firmly adherent to the chorion of the tarsal conjunctiva. These membranes are difficult to remove and leave an irregular hemorrhagic surface. They can progress to conjunctival fibrosis and are sometimes associated with corneal infiltration. They differ from pseudomembranes, which readily detach from the conjunctival epithelium. Subconjunctival hemorrhages can occur in acute conjunctivitis. They are petechial or diffuse, and point to a viral cause (enterovirus 70, coxsackie A24, adenovirus, herpesvirus). Conjunctival granuloma is mainly a sign of sarcoidosis, but can also be found in Parinaud’s oculoglandular syndrome (Table 6.2). Preauricular adenopathies are frequent, but nonspecific, in ad-enoviral conjunctivitis (Table 6.3) Table 6.1 Causes of membranous and pseudomembranous conjunctivitides
Bacteria: diphtheria, beta-hemolytic streptococci (+++), pneumococci, gonococci, neonatal chlamydial conjunctivitis Viruses: Haemophilus influenzae subtype aegyptius, adenovirus (+++), herpesvirus Mycosis: Candida albicans Ligneous conjunctivitis Stevens-Johnson syndrome Chemical burns |
Table 6.2 Parinaud’s oculoglandular syndrome
| Cause Cat-scratch disease +++ (Bartonella henselae) | Clinical Manifestations Child, contact with a cat. Inoculation is usually conjunctival. Incubation lasts 1-3 weeks. Conjunctival nodules 3-4 mm in diameter with little or no purulent discharge. Occasional lymph node suppuration. Possible systemic manifestations: fever, headache, maculopapular eruption, abdominal pain, rarely encephalitis. |
| Tularemia (rabbit fever or deerfly fever) (Franscisella tular-ensis) | Contact with rabbits. Ulcerated conjunctival nodules, corneal ulceration, endophthalmitis. Systemic manifestations +++: pneumonia, pharyngitis, fever, prostration. |
| Tuberculosis (Mycobacterium tuberculosis) | One or several conjunctival nodules, sometimes ulcerated Occasional lymph node suppuration. |
| Syphilis (Treponema pal-dum) | All stages Associated mucocutaneous lesions. |
| Lymphogranuloma venereum (Chlamydia tracho-matis) | Lid edema, chemosis, corneal opacity. Genital infection. |
| Pasteurellosis Listeriosis Actinomycosis Herpes simplex Rickettsiosis (Mediterranean fever) Sarcoidosis |
Table 6.3 Conjunctivitis associated with preauricular adenopathy
| Chlamydia trachomatis Virus: adenovirus Toxic conjunctivitis Superacute bacterial meningococcal or gonococcal conjunctivitis Parinaud’s oculoglandular syndrome |
Bacterial Conjunctivitis
Bacterial conjunctivitis is favored by defective protection of the eyelids and ocular surface constituents (tear film, conjunctival epithelium). It is more frequent in winter and spring, while viral conjunctivitis usually occurs in summer.3 Transmission usually occurs by hand contact, but the conjunctiva can also be inoculated from the nasopharynx (Haemophilus influenzae, Staphylococcus aureus) or lids (Staphylococcus aureus), as well as via the bloodstream (meningococci), the genitalia (neonatal conjunctivitis), and infected eyedrops or contact lenses. Bacterial conjunctivitis is bilateral and accompanied by abundant discharge, which is initially serous then mucopurulent or purulent. Superacute conjunctivitis necessitates a rapid diagnosis and systemic therapy because it is usually caused by Neisseria gonorrhoeae or occasionally Neisseria meningitidis.
Superacute Bacterial Conjunctivitis
Causes
Superacute conjunctivitis is associated with bilateral papillary hyperplasia and thick purulent secretions, chemosis, palpebral edema, and sometimes preauricular adenopathies. It can be complicated by corneal infiltration, which can lead to abscess formation. It is usually caused by gonococci, and more occasionally by meningococci, staphylococci, or streptococci. A laboratory diagnosis must be obtained before starting systemic antibiotic therapy.
Gonococcal Conjunctivitis
Gonococcal conjunctivitis is usually associated with a genital infection, which may be asymptomatic. It is characterized by the frequency and severity of corneal complications. Indeed, gonococci can invade the intact corneal epithelium, provoking severe corneal ulcer and sometimes leading to perforation. Anterior uveitis with hypopion can occur.
Meningococcal Conjunctivitis
Meningococcal conjunctivitis is more frequent in children. It is transmitted by respiratory secretions and spreads via the bloodstream.4 It causes bilateral conjunctivitis, sometimes associated with keratitis or anterior uveitis. Systemic antibiotic therapy is recommended, as it cuts the risk of meningeal involvement by a factor of 20.5
Laboratory Investigations
Direct examination of conjunctival scrapings shows Gram-negative diplococci. Isolates belonging to the genus Neisseria are identified by culture on chocolate agar incubated under CO2-enriched atmosphere. Gonococci and meningococci are distinguished on the basis of carbohydrate fermentation tests.
Therapy
Systemic therapy is required for purulent meningococcal and gonococcal conjunctivitis, in order to prevent corneal involvement and dissemination (Table 6.4).
The development of gonococcal resistance to penicillin and cyclines calls for first-line use of third-generation cephalos-porins (a single dose of 1 g ceftriaxone intramuscularly) or quin-olones.6 Intercurrent Chlamydia trachomatis infection should also be sought and treated (with cyclines or erythromycin).
Intravenous therapy with penicillin G is recommended for meningococcal conjunctivitis. Contacts should receive preventive treatment with oral rifamycin.
Acute Bacterial Conjunctivitis
Acute conjunctivitis is characterized by bilateral papillary hyperplasia accompanied by mucopurulent discharge; adenopathies are absent. These forms often resolve spontaneously within 15 days. The patient’s history helps in determining the cause. In adults, Staphylococcus aureus is the most frequent isolate,3 while Hemophilus influenzae is often found in children under 5.7 Immunocompromised patients have a high incidence of conjunctivitis due to Moraxella or Gram-negative bacilli.8 There are seasonal variations: Hemophilus influenzae is often isolated in the warm season, while pneumococci are more frequent in winter.
Table 6.4 Conjunctivitis warranting systemic antibiotic therapy8
| Gonococci Meningococci Chlamydia trachomatis Hemophilus influenzae subtype aegyptius Pseudomonas aeruginosa in immunocompromised patients Diphtheria Membranous streptococcal conjunctivitis |
Causes
Staphylococcal Conjunctivitis
Staphylococcus aureus colonizes the lid margins and the base of the lashes. It can cause acute recurrent or chronic conjunctivitis, associated with ulcerative blepharitis. Corneal involvement is frequent. Superficial punctate keratopathy is due to the release of toxins, while inferior marginal infiltrates and phlyc-tenular limbal keratitis are secondary to a hypersensitivity reaction to staphylococci.9 Staphylococcus epidermidis rarely causes conjunctivitis.
Streptococcal Conjunctivitis
Streptococcal conjunctivitis is sometimes complicated by membranes and pseudomembranes, but corneal involvement is rare.
Pneumococcal Conjunctivitis
Pneumococcal conjunctivitis is more frequent in cold climates and often occurs in outbreaks. It is often associated with petechial subconjunctival hemorrhage and respiratory signs.
Hemophilus influenzae Conjunctivitis
Hemophilus influenzae conjunctivitis usually occurs in winter, and is characterized by its prolonged course. It is sometimes associated with subconjunctival hemorrhage and early marginal corneal infiltrates. It can be recurrent in patients with intercurrent otitis media10 or respiratory tract infection.11 It is sometimes responsible for bluish coloration of the periorbital skin (in children) or orbital cellulitis.12
The aegyptius subtype is involved in Brazilian purpuric fever, which combines acute conjunctivitis, fever, abdominal signs, and hemorrhagic skin lesions. The mortality rate is 70% in the absence of diagnosis and early treatment.13
Corynebacterium diphtheriae Conjunctivitis
Corynebacterium diphtheriae conjunctivitis has become very rare since the advent of widespread vaccination. It is characterized by severe membranous conjunctivitis, associated with membranous pharyngitis, myocarditis, and muscle palsies. The bacteria can penetrate the intact corneal epithelium, with a high risk of keratitis and corneal perforation.
Moraxella lacunata Conjunctivitis
This form of acute recurrent or chronic follicular conjunctivitis usually occurs in a population at risk (chronic alcoholism, malnutrition, etc.). It is sometimes associated with preauricular adenopathies. The medial and lateral canthi are often involved (angular blepharoconjunctivitis). It can be complicated by corneal ulceration.14
Actinomyces israelii Conjunctivitis
Actinomyces israelii conjunctivitis causes chronic unilateral conjunctivitis predominating at the medial canthus and associated with infection of the lacrimal excretory system. Pressure on the canalicular area may express a whitish granular material.
Bacteriological studies are rarely done, and the diagnosis is suggested by the patient’s history and clinical characteristics. When conjunctivitis is resistant to topical empiric antibiotic therapy (Table 6.5), a treatment-free period of 36 hours is required before performing conjunctival cultures and eventually scrapings for smear examination. Cultures should be inoculated directly on blood agar (streptococci), chocolate agar (Neisseria, Haemophilus), and thioglycolate-enriched medium (anaerobes).15
Therapy
In more than 85% of cases the conjunctivitis is self-limited or resolves after topical antibiotic therapy, one drop every 2-4 hours for 7-10 days16 (Table 6.6). Several single-agent or combined broad-spectrum topical antibiotics (such as polymyxin B-neomycin-gramicidine or polymyxin B-trimethoprim) are commercially available. They are chosen empirically in most settings, but neonates, immunocompromised subjects, and patients with severe conjunctivitis must first undergo a thorough microbiological evaluation. Topical fluoroquinolones should be reserved for severe conjunctivitis. Systemic therapy is usually not necessary. Lid hygiene and oral cyclines are often beneficial in chronic staphylococcal conjunctivitis.
Parinaud’s Oculoglandular Syndrome
This is a form of unilateral chronic follicular conjunctivitis associated with one or several yellowish conjunctival granulomas and preauricular, submandibular, or cervical ade-nopathies. The most frequent cause is cat-scratch disease17 (Table 6.2).
Table 6.5 Indication of conjunctival smear in conjunctivitis
| Neonatal conjunctivitis Hyperacute conjunctivitis Membranous or pseudomembranous conjunctivitis Refractory conjunctivitis Chronic or acute recurrent conjunctivitis Immunodepression |
Table 6.6 Topical (single or combined) antibiotics available in the United States
| Antibiotic | Category | Spectrum |
|---|---|---|
| Norfloxacin Ofloxacin Ciprofloxacin | Fluoroquinolone Fluoroquinolone Fluoroquinolone | Sensitive: staphylococci, Hemophilus, Neisseria, Chlamydia trachomatis, Gram-negative rod infections (Pseudomonas) Resistant: anaerobes and streptococci |
| Bacitracin | Cyclopeptide | Sensitive: Gram-positive bacteria |
| Gentamicin Tobramycin Neomycin Polymyxin | Aminoglycoside Aminoglycoside Aminoglycoside Cyclopeptide | Sensitive: staphylococci, gonococci, Pseudomonas (tobramycin, polymyxin B), Haemophilus (polymyxin B) Resistant: meningococci, streptococci |
| Oxytetracycline | Cycline | Sensitive: broad spectrum (Gram-positive and negative bacteria), chlamydia Resistant: some Gram-negative bacteria |
| Erythromycin | Macrolide | Sensitive: methicillin-susceptible staphylococci, Neisseria, Chlamydia trachomatis, Haemophilus Resistant: some staphylococci, streptococci (pneumoniae), Pseudomonas |
| Chloramphenicol | Phenicol | Sensitive: streptococci, Neisseria, Chlamydia trachomatis Resistant: some Gram-negative bacteria |
Neonatal Conjunctivitis
Neonatal conjunctivitis is defined as conjunctival inflammation diagnosed in the first months of life. It can be bacterial, viral, or toxic. It necessitates specific therapy based on the results of the conjunctival smear (Table 6.5). Its incidence has fallen with the treatment of genital infections in pregnant women and routine prevention in newborns. Three quarters of the normal neonatal commensal bacterial flora is derived from the vaginal flora (lactobacilli, corynebacteria, and bifidobacteria).18
Epidemiology
The estimated incidence of neonatal conjunctivitis in the United States is 0.6 %.19 Chlamydia trachomatis remains the most frequent cause,20 but gonococci (less than 3% of cases)21 are responsible for the most severe cases. Concomitant infections are far from rare. There is a risk of transmission to infants born through the vaginal route, and also by Cesarean section after the membranes have ruptured (Table 6.7). The risk is increased by all forms of ocular trauma during delivery or post-partum.
Table 6.7 Risks of neonatal conjunctivitis
| Active Genital Infection in the Mother | Risk of Neonatal Conjunctivitis22,52 | Incidence of Neonatal Conjunctivitis in the United States24,26 |
|---|---|---|
| Gonococci | 42% | 0.3/1000 births |
| Chlamydia trachomatis | 31-60% | 5-60/1000 births |
| Herpes simplex | 40-60% | 1/3500 births |
Gonococcal Conjunctivitis
After an incubation period of 2-7 days, the newborn develops bilateral purulent conjunctivitis22 with lid edema and chemosis. It is sometimes complicated by conjunctival membranes, corneal ulceration or edema, ocular perforation, or endoph-thalmitis. It is sometimes associated with extraocular signs such as arthritis, meningitis, or septicemia.
Chlamydia Trachomatis Conjunctivitis
Chlamydia trachomatis (serotypes D-K) is responsible for 40% of cases of neonatal conjunctivitis.23 The conjunctivitis usually appears around the eighth day (incubation period of 4-15 days) and is of variable severity. It is generally mucopurulent (sometimes purulent), pseudomembranous, hemorrhagic, or associated with moderate chemosis or lid edema.22,24 It is sometimes poorly symptomatic. It is never follicular, because conjunctival lymphoid tissue is nonexistent before the age of 6-8 weeks.25 In the absence of treatment there is a risk of chronic conjunctivitis, corneal opacification, and neovascularization. The main systemic complication is interstitial pneumonitis.
Herpetic Conjunctivitis
In the United States, one in 3500 newborns is infected by herpes simplex virus.26 Transmission occurs during delivery in 85% of cases,26 and more rarely during pregnancy or post-partum. It is favored by prematurity and by fetal monitoring with scalp electrodes.26 It should be suspected if the mother has a history of genital herpes, even if she is asymptomatic at the time of delivery.26
Neonatal herpetic conjunctivitis can lead to disseminated infection, particularly after maternofetal transmission. Clinical onset occurs 7-14 days after birth. It is unilateral or bilateral, and is characterized in 80% of cases by lid edema and cutaneous vesicles.26 The conjunctivitis is not follicular before 6-8 weeks of life, and can be accompanied by chemosis and microdendritic or geographic keratitis. The stromal edema often resolves and rarely progresses to corneal opacification with neovascularization as long as the immune system is immature. The ocular morbidity is far from negligible, with corneal scarring in 6% of cases,27 together with a risk of cataract, chori-oretinal scars, or optic nerve atrophy. The mortality rate of disseminated herpes is about 57 %.26 A diagnosis of herpes should be considered in all forms of neonatal nonpurulent conjunctivitis, because the prognosis depends on early treatment.
Opportunistic Conjunctivitis
Neonatal conjunctivitis can be caused by Staphylococcus aureus, and more rarely by pneumococci, Hemophilus influenzae, Streptococcus viridans, and Gram-negative bacilli. Infection can occur during delivery or post-partum. The clinical manifestations are delayed (second week).28 Recurrent conjunctivitis in the first month of life may point to nasolacrimal duct stenosis. It is associated with chronic tearing, and pressure applied on the lacrimal sac area can sometimes express purulent material.
Candida Conjunctivitis
Candida conjunctivitis is rare. Onset occurs after the fifth day of life, in the form of pseudomembranous conjunctivitis accompanied by white plaques.
Toxic Conjunctivitis
Toxic conjunctivitis occurs a few hours after birth, in the form of moderate conjunctivitis with nonpurulent secretions. It is usually secondary to instillation of 1 % silver nitrate, which is toxic for conjunctival epithelial cells in 90% of cases.29 It resolves spontaneously within 48 hours and does not necessitate treatment.
Laboratory Investigations
Laboratory tests must be done in all forms of neonatal conjunctivitis, before starting specific treatment (Table 6.5).
Cytological examination of conjunctival smears may show cells with basophilic inclusions (pointing to chlamydiosis), lymphocytes, or eosinophilic intracytoplasmic inclusions (suggestive of herpes simplex infection).
Direct examination may identify gonococcal conjunctivitis (Gram-negative diplococci) pending confirmation by culture on chocolate agar in a CO2-enriched atmosphere, and carbohydrate fermentation tests.
Chlamydia trachomatis can be identified using monoclonal antibodies and immunolabeling methods. Growth in cell culture (McCoy or Hela 229 cells) takes 2-3 days. Gene amplification by the polymerase chain reaction also can be used to detect the microbial genome.
Identification of herpes simplex antigen using direct immunofluorescence is a more rapid diagnostic method than cell culture.
Therapy
Gonococcal Conjunctivitis
Antibiotic therapy must always be given systemically. Since the development of gonococcal resistance to penicillin through penicillinase production, third-generation cephalosporins (ceftriaxone 50 mg/kg per day i. v. or i.m. for 7 days, or a single dose of 50 mg/kg)30 have been recommended for first-line treatment. Resistant gonococci account for about one quarter of cases in children.31
Topical antibiotic therapy is not necessary. Some authors advocate using normal saline to irrigate the eyes to prevent corneal complications induced by purulent discharge and proteo-lytic enzymes under the lids.
Prophylactic therapy consists in instillating 1 % silver nitrate, 0.5 % erythromycin, or 1 % tetracycline into the eye of every neonate immediately after delivery.
Chlamydial Conjunctivitis
Antibiotic therapy with oral erythromycin (50 mg/kg per day in four daily intakes, for 14 days) or roxithromycin (for 10 days) is recommended.32 Topical treatment is not crucial.33
Herpetic Conjunctivitis
Treatment is based on i. v. aciclovir at a dose of 10 mg/kg every 8 hours for 10 days, combined with a topical antiviral agent (3% aciclovir ointment). Systemic prevention of neonatal conjunctivitis is mainly directed against gonococci. It must be administered from the first hour of life.34 Silver nitrate is rarely used. Antibiotics indicated in this setting include rifamycin, cyclines, and erythromycin. Polyvidone iodine can also be used.
Viral conjunctivitis is suggested by follicular conjunctivitis, frequently associated with corneal involvement, preauricular adenopathy, and systemic signs of viral infection. The type of discharge and the presence of papillae are both nonspecific.
Adenoviral Keratoconjunctivitis
Adenoviruses are nonenveloped, double-stranded DNA viruses. The genome is contained in an icosahedral capsid 70-90 nm in diameter. The virus replicates within the nucleus of the infected host cell. It is very stable and resistant to many solvents (alcohol, ether, etc.) and detergents. Some serotypes, such as 5, 8, and 19, can be recovered viable on inert materials after 4-5 weeks.35 There are 47 serotypes of adenovirus36 which are distinguished on the basis of genomic variations. There is no known animal reservoir. Adenovirus is the leading cause of viral conjunctivitis and is responsible for 5-20% of cases of acute conjunctivitis.37
Epidemiology
Adenoviral conjunctivitis is often acute and occasionally chronic38. It is transmitted by the hands or indirectly via contaminated ophthalmic equipment or eyedrops. There is a winter peak concomitant with respiratory tract infections, and also a summer peak associated with swimming pools. Infants are protected by maternal antibodies up to age 6 months. After a mean incubation period of 7 days (± 3 days), viral replication on the ocular surface can last 21 days. The conjunctivitis is often bilateral, although one eye is often less severely involved than the other.
Causes
The clinical manifestations result from both viral replication and the immune response with cellular infiltration of Bowman’s layer and the underlying stroma by lymphocytes, histiocytes, and fibroblasts.39 The clinical presentations depend on the serotype involved: epidemic keratoconjunctivitis, pharyn-geal conjunctival fever, nonspecific acute follicular conjunctivitis, and chronic papillary conjunctivitis.
Epidemic Keratoconjunctivitis
Epidemic keratoconjunctivitis (EKC) is due to adenovirus serotypes 8,19, and 37,40,41 that are transmitted by the hands, ophthalmic intruments (e.g., applanation tonometer), or infected eyedrops. It is highly contagious and mainly affects adults.
It is characterized by follicular conjunctivitis (Figs. 6.1 and 6.2), sometimes associated with preauricular adenopathy; systemic signs are absent. Papillary hypertrophy, conjunctival hemorrhage, chemosis, and lid edema are frequent. In severe forms, membranes or pseudomembranes may develop on the tarsal conjunctiva. They can mask the follicles and rapidly progress to conjunctival fibrosis. Anterior uveitis, increased intraocular pressure, and disciform keratitis are rare.
In the acute phase, 3-4 days following the onset of the first conjunctival symptoms, diffuse superficial punctate keratitis occurs with gradual confluence. The conjunctivitis resolves in 2-3 weeks, while the corneal involvement peaks at the fourth week. Between the second and third weeks, nummular fluorescein-negative subepithelial infiltrates 1-2 mm in diameter develop, mainly in the center of the cornea, but sometimes over the entire corneal surface. These infiltrates disappear spontaneously, but often very slowly (sometimes after several years). They can cause permanent photophobia and glare, and decreased visual acuity secondary to irregular astigmatism or opacities located in the visual axis.
Pharyngeal Conjunctival Fever
Pharyngeal conjunctival fever (PCF) is caused by the transmission, via respiratory secretions, of adenovirus serotypes 3, 4, or 7.42 It is sometimes contracted in swimming pools. It is more frequent in children.
Bilateral follicular conjunctivitis is associated with fever, pharyngitis, and submaxillary and preauricular adenopathies. The secretions are serous, and corneal involvement is limited to superficial punctate keratopathy; corneal subepithelial infiltrates are much less common than in EKC. Palpebral ecchymosis and petechial subconjunctival hemorrhages can occur. The disease usually resolves without complications after two weeks.
Chronic Papillary Conjunctivitis
More rarely, adenoviruses cause chronic nonspecific papillary conjunctivitis.43 An erronious diagnosis of allergic conjunctivitis in patients with undiagnosed adenoviral conjunctivitis, and concomitant use of topical corticosteroids may alter the course of the disease and favor its spread. The diagnosis can only be established once the virus has been isolated.
Differential Diagnosis
Thygeson superficial punctate keratitis is distinguished from adenoviral keratopathy by a negative history of conjunctivitis, the absence of follicles, its recurrent nature, the topography of the lesions which are predominant in the center of the cornea, the strictly epithelial involvement, and fluorescein uptake by some lesions.
Laboratory Investigations
Cytological examination of the conjunctival smear shows a lymphocytic and polymorphonuclear infiltrate, and no intracy-toplasmic inclusions within the conjunctival epithelial cells.
The virus can be isolated during the acute phase of conjunctivitis or keratitis, but is no longer detectable at the stage of nummular subepithelial keratitis. The virus is identified by its cytopathogenic effect in cell culture, after 2-7 days. The viral antigens can be identified by immunoenzymatic or direct immunofluorescence methods in 1-2 hours. Pharyngeal conjunctival fever is rarely necessary to establish the diagnosis.
Antibody detection, with a rise in titer between two samples, confirms the diagnosis at the early stage of subepithelial opacities. The serotype diagnosis is based on hemag-glutination or neutralization.
Therapy
There are no antiviral treatments with proved efficacy in adenoviral conjunctivitis.44–46 Trials of trifluridine have been inconclusive, despite adenoviral sensitivity in vitro.46 Cidofovir reduces viral replication in rabbits but is not used in humans.47
Treatment of the acute phase is therefore symptomatic, and is based on cold compresses, irrigation with normal saline, and sometimes topical vasoconstrictors. Smoked glasses attenuate photophobia and glare. Topical antibiotics only prevent super-infection. Topical corticosteroids are used only for severe forms with conjunctival membranes or anterior uveitis (Table 6.8). Indeed, although they have an antiviral action in vitro, they carry a risk of prolonging viral shedding and facilitating viral spread.48
Table 6.8 Indications of topical corticosteroid therapy in adenoviral keratoconjunctivitis
| Early Stage | Nummular Keratitis | |
|---|---|---|
| Advantages | Improves symptoms Antiviral action in vitro | Resorption of opacities in the visual axis |
| Disadvantages | Favors viral replication Prolongs carriage | Steroid dependence Relapse of opacities on treatment cessation |
| Reduces immune defenses No long-term advantage | Steroid-induced complications No long-term advantage | |
| Indications | Anterior uveitis Membranes, pseudomembranes | Must be avoided |
The use of topical corticosteroids in nummular keratitis is controversial because it carries a risk of steroid dependence and complications (cataract and glaucoma). Corticosteroids aid the resolution of subepithelial opacities, but these reappear on treatment cessation49 (see Table 6.8).
One cause of decreased vision during the cicatricial period (after 2 years) is irregular astigmatism due to subepithelial scarring. Irregular astigmatism can be attenuated by the use of rigid contact lenses. Excimer laser phototherapeutic kera-tectomy can be indicated for superficial scars located in the visual axis. However, it induces hyperopic shift and is inadequate for deep opacities.
The patient must be informed of the risk of transmission and of preventive measures. Regular hand washing and strictly personal use of linen and glasses are recommended. There is a real risk of transmission for at least 1 week after symptom onset.43 Patients should be exempted from work or school for 15 days after involvement of the second eye.
In the ophthalmologist’s office, preventive measures include the use of gloves, hand washing, careful cleansing of equipment (e.g., slitlamp), and the use of single-dose eyedrop preparations.
Herpetic Conjunctivitis
Herpes simplex virus is a double-stranded DNA virus with an icosahedral capsid and an envelope derived from the host cell nuclear membrane. The virus can remain viable for 2 hours on a dry surface and for 8 hours on a wet surface. It is inactivated by many disinfectants, including 70% alcohol, quaternary ammonia, glutaraldehyde, iodinated compounds, and phenols.50 It is transmitted by contact with an infected mucocutaneous lesion, secretions, or contaminated equipment (e.g., a tonometer). It is the leading cause of sight-threatening ocular infections.51 Humans are the only viral reservoir.
Ocular herpes in adults is usually due to herpes simplex virus type 1 (HSV1), while neonatal herpes is mainly due to type 2 (HSV2). Primary infection takes place in childhood, and is followed by a period of latency in neuronal cell bodies situated in ganglia. Reactivation occurs following events such as stress, exposure to ultraviolet radiation, trauma, surgery, and menstruation.
Epidemiology
Primary infection often occurs between 1 and 5 years of age. It takes place earlier in poor socioeconomic conditions. At age 5, 60% of children have encountered HSV1, but symptomatic forms and ocular involvement only affect 6% and 1 %, respectively, of infected subjects.52 Ninety per cent of adults are seropositive. The recurrence rate after the primary infection is 24% during the first year53 and 40% after 5 years.54 It is multiplied by two if corneal lesions were associated with palpebro-conjunctival lesions during the primary infection.55 The risk of stromal keratitis after ocular herpes is about 20%.
Clinical Aspects
Clinical severity is variable and depends on the viral strain and the immune response. Thus, atopic subjects frequently have bilateral lesions, and HIV-infected subjects have prolonged and frequently recurrent exacerbations. Herpetic conjunctivitis usually occurs during the primary infection, while corneal involvement is most frequent during recurrences.
Primary herpetic infection is rarely symptomatic, manifesting as gingivostomatitis or ocular involvement. After an mean incubation period of 7 days, vesiculous palpebral lesions occur, associated with conjunctivitis (54%),55 superficial keratitis (63%), and, more rarely, stromal keratitis or uveitis. The conjunctivitis is frequently unilateral, follicular, and associated with a preauricular adenopathy. Conjunctival ulceration can occur, while pseudomembranes are rare. The corneal involvement is often atypical, consisting of epithelial punctate keratitis, multiple dendrites, or geographic keratitis. Stromal involvement is rare.
Recurrent herpetic conjunctivitis is rare and can mimic adenoviral conjunctivitis with follicles and corneal subepithelial opacities. Corticosteroids are contraindicated in such cases.
Laboratory Investigations
The clinical picture is often highly evocative. Cytological examination of conjunctival scrapings shows giant syncytial balloon cells, reflecting the cytopathogenic action of the virus. The use of monoclonal antibodies and direct immunofluorescence yields a rapid diagnosis (1-2 hours). The virus can be isolated by inoculation on cell cultures (10-15 days). Electron microscopy is not routinely used.
During primary infection, seroconversion or a rise in the antibody titer supports the diagnosis. Serological tests have no diagnostic value during recurrences.
Therapy
Herpetic conjunctivitis occurring during the primary infection requires topical antiviral treatment (aciclovir ointment). Corticosteroids are contraindicated.
Epstein-Barr Virus Conjunctivitis
Epstein-Barr virus (EBV) is a herpesvirus with an icosahedral capsid and double-stranded DNA. It is responsible for infectious mononucleosis (glandular fever) and is also involved in Burkitt’s lymphoma and nasopharyngeal cancer.
Epstein-Barr virus is transmitted by the saliva. Ninety per cent of individuals have detectable anti-EBV antibodies by the time they reach their third decade.56
Clinical Aspects
Infectious mononucleosis is associated with sore throat, poly-adenopathy, hepatitis and hyperlymphocytosis, and fatigue. Conjunctivitis is the most frequent ocular manifestation (up to 38% of cases).57 It sometimes occurs 1-4 weeks after the acute episode. It is follicular and/or papillary, often unilateral, and sometimes associated with subconjunctival hemorrhage or membranes. It is one of the manifestations of Parinaud’s oculo-glandular syndrome.58 It can be complicated by corneal involvement, consisting of epithelial punctate keratitis, micro-dendrites,59 and, more rarely, diffuse subepithelial opacities of 0.1-2 mm in diameter,60 with or without neovascularization. The other forms of ocular involvement, such as scleritis, uveitis, chorioretinitis, papilledema, and oculomotor palsies, are rarer.
Laboratory Investigations
Serological tests are only contributory two weeks after clinical onset. The diagnosis of acute infectious mononucleosis is based on the demonstration of early antigens (EA), IgM antibodies and a rise in IgG antibodies against the viral capsid antigen (anti-VCA); antibodies against the Epstein-Barr nuclear antigen (anti-EBNA) are undetectable. Anti-EBNA antibodies appear late and remain positive for years.
Epstein-Barr virus can be detected by immunoenzymatic methods on corneal scrapings.
Therapy
There is no specific therapy for the ocular manifestations of infectious mononucleosis. Aciclovir has not proved its efficacy. Topical corticosteroid therapy is not usually necessary.
Varicella-Zoster Virus Conjunctivitis
Varicella-zoster virus (VZV) is a double-stranded DNA virus 150-200 nm in diameter, with an icosahedral protein capsid and an envelope derived from the host outer cell membrane.
Epidemiology
It is transmitted by direct contact with skin lesions, or the saliva (chickenpox).61 Chickenpox corresponds to the primary infection and usually affects young children, while zoster is due to viral reactivation in neurosensory ganglia and affects adults.
Clinical Aspects
Chickenpox
The disseminated vesicular eruption can affect the lids and ocular surface. It sometimes involves the conjunctiva, where vesicles close to the limbus take on the appearance of phlyc-tenules that progress to ulceration.61 Papillary conjunctivitis is frequent in uncomplicated chickenpox. More rarely, inflammatory conjunctival papules can develop and become ulcerated. Corneal involvement is limited, and includes epithelial punctate keratitis, pseudodendrites, and, rarely, recurrent disciform keratitis.62
Herpes Zoster
Ocular involvement is frequent (76% of cases) when the eruption affects the side of the tip of the nose (alae nasae), which is innervated by the nasociliary branch of the ophthalmic division of the trigeminal nerve.63 In the acute phase, papillary or follicular conjunctivitis can be associated with conjunctival vesicles, petechial subconjunctival hemorrhages and, rarely, pseudomembranes or membranes.64,65 Bacterial superinfection is frequent. Viral replication in the corneal epithelium results in epithelial punctate keratitis, or multiple peripheral pseudodendrites that stain weakly with fluorescein.66,67 Unilateral preauricular adenopathy is frequent.
Laboratory Investigations
Direct immunofluorescence and viral neutralization techniques offer rapid confirmation of the diagnosis. The virus can be isolated by cell culture.
Therapy
Oral or intravenous aciclovir is recommended for complicated forms of chickenpox. Conjunctivitis alone does not warrant this treatment. The efficacy of 3 % aciclovir ointment has not been demonstrated.
Treatment of zoster ophthalmicus with oral aciclovir (800 mg, five times a day for 7 days) or oral valaciclovir (1 g, three times a day for 7 days) must be started within 72 hours after the eruption. It reduces the incidence of severe ocular complications and postherpetic neuralgia.68–70 Topical aciclovir may be indicated when conjunctivitis is associated with corneal involvement.
Molluscum Contagiosum Conjunctivitis
Molluscum contagiosum is due to poxvirus infection.71 Human beings are the exclusive reservoir host of this 230-330 nm-large double-stranded DNA virus. It replicates in the epidermis and induces hypertrophy and hyperplasia of epithelial cells. The virus is poorly immunogenic and relapses are possible.
Epidemiology
Transmission occurs through direct contact or contaminated water, such as swimming pools. Scratching of the lesions is a source of autoinoculation. Children are frequently affected. The lesions are often disseminated in atopic and immunocom-promised (in particular HIV-infected) subjects.71a
Clinical Aspects
After a variable incubation period (1 week to 3 months), painless translucent cutaneous nodules appear more likely on the face, abdomen, and genitalia. The elementary lesion consists of a round, smooth papule with an umbilicated center, on an ery-thematous base. Its color is flesh or pearly-white, and its size generally ranges from 2-4 mm, but it also may be larger. Expressed fluid material from its core contains the infectious agent. Ocular involvement is associated with palpebral lesions (Fig. 6.3). It consists of frequently chronic follicular conjunctivitis, sometimes complicated by epithelial superficial keratitis, pseudodendrites, or a corneal pannus.71b The molluscum is rarely located on the conjunctiva or caruncle. The skin lesions may regress slowly in immunocompetent subjects, and can become superinfected.
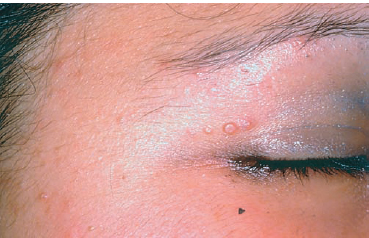
Fig. 6.3 Molluscum contagiosum on the lid margins (courtesy of Prof. S. Belaich, Dermatology, Hâpital Bichat, Paris)
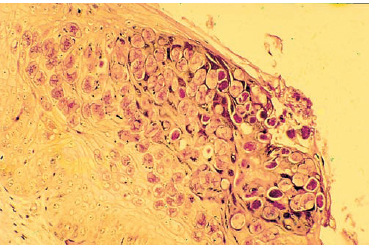
Fig. 6.4 Molluscum contagiosum inclusions
Laboratory Investigations
The diagnosis is generally easy, as the clinical aspect is highly evocative. Histopathological examination of skin lesions shows characteristic intracytoplasmic eosinophilic inclusions (molluscum bodies) (Fig. 6.4). Cell culture is not possible, and there are no serological diagnostic tests. The virus can be identified by electron microscopy.
Therapy
There is no medical therapy. Treatment of the lesions located on the lid margins is somewhat delicate. Excision and curet-tage of these lesions are preferred to cryotherapy and caustic chemical applications which are too toxic in this region.71c
Papillomavirus Conjunctivitis
Papillomavirus is a double-stranded DNA, icosahedral, nonenveloped virus 52-55 nm in diameter belonging to the papovavirus family. There are 58 types of human papillomavirus, identified on the basis of genomic variations.
Epidemiology
Papillomavirus infection is more frequent in children and young adults. Transmission occurs by direct contact or via soiled linen. It is favored by poor hygiene. Autoinfection is frequent. Asymptomatic carriage is possible.
Clinical Aspects
Papillomavirus has a propensity to reside and infect cutaneous and mucosal epithelial cells. It causes hypervascularized, pe-donculated raspberry-colored lesions on the bulbar, tarsal, and caruncular conjunctiva, or sessile lesions on the limbus. It rarely invades the canaliculi.72 Papilloma of the lid margins is associated with papillary conjunctivitis, sometimes with epithelial punctate keratitis, and rarely with a corneal pannus. The relapse rate after treatment is 40 %.73 Papillomavirus is also isolated in 85% of cases of unilateral conjunctival dysplasia or epithelioma.74
Laboratory Investigations
Histopathological conjunctival examination of the lesions shows koilocytosis75 with basophilic cytoplasmic inclusions. Cell culture and serological diagnosis are not available. The viral DNA can be detected by in situ hybridization75 or poly-merase chain reaction (PCR).76 The virus itself can be identified by electron microscopy.
Therapy
The lesions may regress spontaneously. Their treatment is based on surgery, which combines resection with cauterization or cryotherapy of the base of the lesion. It carries a risk of autoinoculation. The CO2 laser has been used to treat papilloma.77 There is no specific treatment for conjunctival involvement.
Acute Hemorrhagic Conjunctivitis
Enterovirus 70 and coxsackie A24 virus have been incriminated in several outbreaks of acute hemorrhagic conjunctivitis.78 These two viruses, which belong to the picornavirus family, have a single-stranded RNA genome and four structural proteins that determine the serotype. They replicate most efficiently at 33-35°C in a moist environment.
Epidemiology
Transmission to the eye occurs via the hands or soiled linen. The mean incubation period is 24 hours (12-48 hours).
Clinical Aspects
Enterovirus 70 and coxsackie A24 infection produces the same clinical picture.79 The follicular conjunctivitis is characterized by subconjunctival hemorrhage that mainly affects the supero-temporal quadrant of the bulbar conjunctiva, but that can be diffuse. Chemosis, papillary hypertrophy, preauricular ade-nopathies, and lid edema, with or without ecchymosis, can occur. Pseudomembranes are rare. Corneal involvement is often limited to the epithelium (epithelial punctate keratitis); corneal infiltrates and scars are rare. Recovery occurs spontaneously in 3-5 days.
Hemorrhagic conjunctivitis is associated with extraocular signs such as fatigue, fever, myalgia, headache, rhinorrhea, and cough. Neurological complications have been reported in patients with enterovirus 70 conjunctivitis.80 They are rare (1/10000-1/20000) and are due to motoneuronal injury in the anterior horn of the spinal cord. They consist of palsies of the peripheral nerves or cranial nerves (mainly the facial nerve), preceded by radicular or muscle pain. They appear 1-5 weeks after the conjunctivitis and last about 3 weeks on average. Recovery is sometimes incomplete.
Laboratory Investigations
Enterovirus 70 and coxsackie A24 viruses are difficult to isolate because of the rapid resolution of hemorrhagic conjunctivitis. They can only be recovered if the sample is taken during the first two days and inoculated on cell culture.81 Monoclonal antibodies and immunoenzymatic, direct immunofluorescence, or viral neutralization techniques permit rapid diagnosis.82
Serological tests (viral neutralization or hemagglutination) can assist with the diagnosis in patients with late manifestations (neurological complications). A titer above 1/16 and a four-fold rise in titer between two samples is necessary to establish the diagnosis. The rise in antibody titers occurs late, however (after the seventh day).
Therapy
Treatment is symptomatic, based on the notion that application of a hot compress to the lids hinders viral replication.83
Other Forms of Viral Conjunctivitis
Measles is frequently associated with acute conjunctivitis and epithelial superficial keratitis. It is responsible for corneal scarring and systemic complications in immunocompromised patients.
The follicular conjunctivitis and epithelial superficial keratitis sometimes seen in patients with rubella resolve rapidly in immunocompetent subjects and do not require specific therapy. Bacterial superinfection and corneal complications are frequent in immunocompromised patients.
Cytomegalovirus (CMV) rarely infects the ocular surface, although follicular conjunctivitis can occur in patients with AIDS. Cells with CMV inclusions have been found in the vessel walls of the conjunctiva.84
References
20. Schachter J. Chlamydia infections. West J Med 1990; 153: 523-34.
25. Grosskreutz C, Smith LBH: Neonatal conjunctivitis, Int Ophthalmol Clin 1992; 32:71.
28. Rapoza PA. Epidemiology of neonatal conjunctivitis. Ophthalmology 1986; 93: 456-461.
29. Friendly DS. Ophthalmia neonatorum. Pediatr Clin North Am 1983; 30: 1033.
37. Gigliotti F. Acute conjunctivitis of childhood. Pediatr Ann 1993; 22: 353-6.
42. Gigliotti F, Williams WT, Hayden FG, Hendley JO, Benjamin J, Dickens M, Gleason C, Perriello VA, Wood J. Etiology of acute conjunctivitis in children. J Pediatr 1981; 98: 531-6.
49. Murrah WF. Epidemic keratoconjunctivitis. Ann Ophthalmol 1988; 20: 36-8.
57. Ash HH, Arbogast JL Infectious mononucleosis. J Indiana Med Assoc 1942; 35: 562-4.
59. Wilhelmus KR. Ocular involvement in infectious mononucleosis. AmJ Ophthalmol 1981; 91: 117-8.
63. Harding SP. Management of ophthalmic zoster. J Med Vir 1993; 1: 97-101.
65. Marsh RJ, Cooper M. Ophthalmic zoster. Eye 1993; 7: 350-70.
78. Yin-Murphy M. Viruses of acute hemorrhagic conjunctivitis. Lancet 1973; 1: 545-6.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree