Purpose
To evaluate the safety and efficacy of adding fixed-combination brinzolamide 1%/brimonidine 0.2% (BBFC) as adjunctive therapy to travoprost 0.004% (TRAV) in patients with open-angle glaucoma or ocular hypertension.
Design
Multicenter, randomized, double-masked, parallel-group phase 4 clinical trial.
Methods
setting : Multicenter; 32 sites in the United States. patient population : Total of 233 patients with open-angle glaucoma or ocular hypertension and with mean intraocular pressure (IOP) ≥21 mm Hg and <32 mm Hg while receiving once-daily TRAV monotherapy. intervention : Masked BBFC or vehicle (3 times daily) adjunctive to TRAV for 6 weeks. main outcome measure : Mean diurnal IOP averaged over 8 AM, 10 AM , 3 PM, and 5 PM time points at week 6. Superiority of BBFC+TRAV over vehicle+TRAV was based on statistical significance of a treatment difference favoring BBFC+TRAV.
Results
Mean diurnal IOP at week 6 (least squares mean ± standard error) was 17.6 ± 0.4 mm Hg and 20.7 ± 0.4 mm Hg in the BBFC+TRAV and vehicle+TRAV groups, respectively (between-group difference, −3.2 ± 0.5 mm Hg; P < .0001). Superiority of BBFC+TRAV over vehicle+TRAV was established. Mean and percent diurnal IOP change from baseline were significantly greater with BBFC+TRAV compared with vehicle+TRAV ( P < .0001 for both). Conjunctival hyperemia was the most common treatment-related adverse event in either group (BBFC+TRAV, 12.8%; vehicle+TRAV, 6.0%).
Conclusions
Adjunctive treatment with BBFC added to TRAV resulted in lower mean diurnal IOP after 6 weeks of treatment compared with vehicle added to TRAV; this difference was both statistically and clinically significant.
Glaucoma is a progressive optic neuropathy characterized by degeneration of retinal ganglion cells that results in loss of visual field and, potentially, leads to blindness. Although multiple factors (eg, elevated intraocular pressure [IOP], family history, ethnicity, corneal biomechanics) have been associated with the development and progression of primary open-angle glaucoma, large-scale clinical studies have demonstrated that reducing IOP decreases the risk of progression or conversion of ocular hypertension to glaucoma.
Many patients require multiple IOP-lowering agents to achieve and maintain their target IOP. In the Ocular Hypertension Treatment Study, approximately 40% of patients required ≥2 medications to meet their target IOPs. Initial therapy has been reported to be insufficient in the first 2 years of treatment for 50% of patients in the United States.
Combination therapies are often effective when taken as prescribed, but treatment adherence can decline with increasing numbers of individual medications and increasing treatment regimen complexity. Fixed-combination medications allow concomitant administration of multiple ocular hypotensive medications with a single-drop instillation, with the potential of additive IOP-lowering efficacy and a simplified dosing regimen.
A fixed combination recently approved by the US Food and Drug Administration (FDA) is Simbrinza, which contains the carbonic anhydrase inhibitor brinzolamide 1% and the α 2 -adrengeric agonist brimonidine 0.2% (BBFC; SIMBRINZA; Alcon Laboratories, Inc, Fort Worth, Texas, USA). Currently, BBFC is the only available FDA-approved fixed-combination glaucoma medication that does not contain a β-blocker. Two Phase 3 trials have demonstrated superior IOP-lowering efficacy of BBFC compared with its individual components (ie, brinzolamide 1% or brimonidine 0.2%).
Treatment guidelines for glaucoma have suggested a stepwise approach to adding agents for patients with insufficient IOP control. However, adding a fixed-combination therapy to monotherapy may be reasonable in patients needing to reach a target IOP lower than may be reasonably expected with addition of a single agent. Additive efficacy of BBFC adjunctive to branded prostaglandin analogues was recently described (unpublished data; Fechtner RD, et al. American Glaucoma Society, 2015). The purpose of this study was to evaluate the safety and efficacy of adding BBFC as adjunctive therapy to the prostaglandin analogue travoprost 0.004% (TRAV) in patients with open-angle glaucoma or ocular hypertension.
Methods
Study Design and Treatment
This was a prospective, multicenter, 1:1 randomized, double-masked, parallel-group phase 4 clinical trial conducted at 32 sites in the United States between October 2013 and April 2014 ( ClinicalTrials.gov identifier, NCT01937299 ; registered September 2013). The study was performed in compliance with the ethical principles of the Declaration of Helsinki, Good Clinical Practice, and the Health Insurance Portability and Accountability Act. The study protocol and consent forms were prospectively approved by The University of Texas Health Science Center Houston Committee for the Protection of Human Subjects, Chesapeake Institutional Review Board (IRB), West Virginia University IRB, and Sterling IRB. Informed consent was obtained from patients at enrollment.
Eligible patients were aged ≥18 years, were diagnosed with primary or secondary open-angle glaucoma or ocular hypertension, and had mean IOP ≥21 mm Hg and <32 mm Hg in at least 1 eye (the same eye[s]) at the 8 AM time point of both eligibility visits (ie, after washout of prior IOP-lowering medications and while receiving TRAV monotherapy). Exclusion criteria are provided in the Supplementary Table (available at AJO.com ).
The study consisted of 2 sequential phases: an open-label run-in phase and a randomized, double-masked treatment phase ( Figure 1 ). The open-label phase included a screening visit during which patients discontinued their prior IOP-lowering medication(s) and simultaneously initiated once-daily TRAV (TRAVATAN Z; Alcon Laboratories, Inc). TRAV therapy was continued throughout the duration of the study, and at no point during the study were patients without ocular hypotensive treatment. Eligibility visits were scheduled after the appropriate washout durations required based on patients’ prior ocular hypotensive therapies. Informed consent was obtained at the screening visit. After washout, eligibility IOP assessments were conducted at 8 AM, 10 AM, 3 PM, and 5 PM (all ±30 minutes), with the 2 visits scheduled 3–8 days apart to determine patients’ TRAV-treated baseline diurnal IOP and IOP at individual time points.
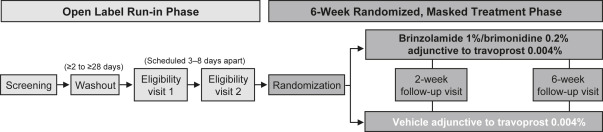
Patients who met eligibility criteria at both visits were randomized 1:1 to receive either BBFC+TRAV or vehicle+TRAV for the 6-week duration of the randomized, double-masked treatment phase. Randomization was centralized and blocked and was conducted using an electronic data collection system with interactive response technology. The random allocation sequence was generated using SAS 9.1 PROC PLAN software (SAS Institute, Inc., Cary, NC). Patients, investigators, investigational center staff, the study sponsor, and clinical monitors were masked to treatment assignments. Assigned treatments were provided in kits containing identical masked bottles, labels, and packaging.
Randomized patients were instructed to instill 1 drop of their masked treatment (BBFC or vehicle) in both eyes 3 times daily at approximately 8 AM, 3 PM, and 10 PM and to instill 1 drop of TRAV in both eyes once daily at bedtime. The treatment phase included 2 on-therapy follow-up visits conducted at week 2 and week 6. The schedule of study assessments is described in Table 1 .
| Assessment | Screen | Eligibility 1 and 2 | Week 2 | Week 6 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 8 AM | 10 AM | 3 PM | 5 PM | 8 AM | 10 AM | 3 PM | 5 PM | 8 AM | 10 AM | 3 PM | 5 PM | ||
| Best-corrected visual acuity | X | X | X | X | |||||||||
| Anterior segment examination | X | X | X | X | |||||||||
| Intraocular pressure a | X | X | X | X | X | X | X | X | X | X | X | X | X |
| Visual fields | X | X | |||||||||||
| Dilated eye examination | X | X | |||||||||||
| Adverse events | X | X | X | X | X | X | X | X | X | X | X | X | X |
a At scheduled study visits, the 8 AM and 3 PM intraocular pressure measurements were performed before administration of the 8 AM and 3 PM doses of study treatments.
Efficacy Outcomes
The primary efficacy endpoint was mean diurnal IOP averaged over 4 time points (8 AM, 10 AM, 3 PM, and 5 PM) at week 6. Secondary efficacy endpoints were mean diurnal IOP change and percent diurnal IOP change from baseline at week 6. Additional outcomes were mean diurnal IOP at week 2, mean and percent IOP change from baseline at week 2, IOP at each time point at week 2 and week 6, and mean and percent IOP change from baseline at each time point at week 2 and week 6.
To limit potential bias, IOP was measured using a masked IOP reader, and study personnel who administered eye drops during study visits did not perform IOP measurements. Intraocular pressure measurements were taken for both eyes at all study visits using a calibration-verified Goldmann applanation tonometer; 2 measurements were taken for each eye at every time point. The same operator and reader measured IOP for a given patient using the same method at all visits. On study visit days, the 8 AM and 3 PM treatment doses were administered by study personnel approximately 15 minutes after completion of IOP measurement.
Efficacy endpoints were analyzed in the intent-to-treat data set (ie, all patients who received study medication and completed at least 1 scheduled on-therapy visit). Data from 1 eye from each patient (ie, the study eye) were analyzed. If both eyes received study medication, the eye with higher IOP at 8 AM averaged across the 2 eligibility visits was selected. If IOP was equal between eyes at 8 AM assessments, the eye with higher IOP at 10 AM was selected. If both eyes had equal IOP at both the 8 AM and 10 AM time points, the right eye was selected as the study eye. Baseline IOP at each time point was calculated as the integer average of the IOP at the 2 eligibility visits at that time point.
Safety Outcomes
Safety was evaluated from adverse event (AE) reports and eye examination for all patients exposed to treatment; safety outcomes were summarized descriptively. Adverse events were coded using Medical Dictionary for Regulatory Activities version 16.0 Preferred Terms. Best-corrected visual acuity was assessed using a standardized ETDRS chart and was calculated as the number of letters read correctly + 30. Visual fields were evaluated using achromatic automated perimetry and were calculated as mean deviation from the Humphrey Field Analyzer device. If both eyes were treated, visual fields were averaged for the left and right eyes.
Statistical Analysis
The primary efficacy endpoint (mean diurnal IOP) was analyzed on an observed-case basis; consequently, there was no imputation for missing data. Treatment group differences in mean diurnal IOP at week 6, mean diurnal IOP change from baseline at week 6, and percent diurnal IOP change from baseline at week 6 were determined based on least squares (LS) means using a repeated-measures analysis of variance model that accounted for covariate baseline IOP, with fixed effect terms for treatment, visit, and treatment-by-visit interaction. The superiority of BBFC+TRAV relative to vehicle+TRAV was considered established if the mean reduction in diurnal IOP at week 6 was significantly greater for the adjunctive therapy relative to monotherapy (ie, lower mean diurnal IOP value with BBFC+TRAV), as determined using a 2-sided statistical test at the 5% significance level. To ensure that the statistical model and associated analyses were robust for any data points missing at random, a sensitivity analysis was performed using the last-observation-carried-forward (LOCF) method. In the sensitivity analysis, time-matched data were imputed for calculation and analysis of mean diurnal IOP at week 6. A post hoc analysis with study site as a fixed effect in the statistical model was conducted to confirm the results of the preplanned primary endpoint analysis. There was no preplanned statistical testing for treatment differences in IOP data for supportive endpoints; however, these endpoints were also analyzed post hoc using a model analogous to that described above. P values from this analysis were generated for descriptive purposes only.
A sample size of approximately 200 patients was determined to be sufficient to detect a between-group difference in mean diurnal IOP of 2.0 mm Hg, a common measure of clinical significance in IOP studies, with ≥90% power. This estimate was based on an assumed IOP standard deviation (SD) of 3.5–3.9 mm Hg and a 2-sided t test at a significance level of P < .05.
Results
Patients
Of the 233 patients randomized to treatment, 229 were included in the intent-to-treat data set (BBFC+TRAV, n = 113; vehicle+TRAV, n = 116; Figure 2 ). Mean ± SD patient age was 66.8 ± 10.5 years; most patients were female (59.4%; 136/229), white (78.6%; 180/229) and were diagnosed with open-angle glaucoma (72.1%; 165/229; Table 2 ). All demographic characteristics were comparable between groups. Overall, 7.7% of randomized patients (18/233) discontinued the study (BBFC+TRAV, 12.0%, 14/117; vehicle+TRAV, 3.4%, 4/116). The most common reason for discontinuation was an AE (BBFC+TRAV, 11.1%, 13/117; vehicle+TRAV, 0%). The remaining reasons for discontinuation were lack of efficacy (vehicle+TRAV, 3.4%, 4/116) and patient withdrawal (BBFC+TRAV, 0.9%, 1/117).
| Brinzolamide 1%/Brimonidine 0.2% + Travoprost 0.004% (n = 113) | Vehicle + Travoprost 0.004% (n = 116) | |
|---|---|---|
| Age, y | ||
| Mean ± standard deviation | 67.5 ± 10.3 | 66.0 ± 10.6 |
| Range | 31–91 | 30–90 |
| Age group, n (%) | ||
| <50 | 6 (5.3) | 10 (8.6) |
| 50–64 | 35 (31.0) | 38 (32.8) |
| ≥65 | 72 (63.7) | 68 (58.6) |
| Sex, n (%) | ||
| Female | 69 (61.1) | 67 (57.8) |
| Male | 44 (38.9) | 49 (42.2) |
| Race, n (%) | ||
| White | 91 (80.5) | 89 (76.7) |
| Black or African American | 17 (15.0) | 26 (22.4) |
| Asian | 5 (4.4) | 1 (0.9) |
| Ethnicity, n (%) | ||
| Not Hispanic or Latino | 102 (90.3) | 104 (89.7) |
| Hispanic or Latino | 11 (9.7) | 12 (10.3) |
| Diagnosis, n (%) | ||
| Open-angle glaucoma | 81 (71.7) | 84 (72.4) |
| Ocular hypertension | 24 (21.2) | 28 (24.1) |
| Open-angle glaucoma with pseudoexfoliation | 6 (5.3) | 2 (1.7) |
| Open-angle glaucoma with pigment dispersion | 2 (1.8) | 2 (1.7) |
| Diurnal intraocular pressure, mm Hg | ||
| Mean ± standard deviation | 22.5 ± 2.5 | 22.7 ± 2.4 |
| Range | 17.8–30.8 | 18.0–30.5 |
Efficacy Outcomes
Mean ± SD TRAV-treated baseline IOP was 22.5 ± 2.5 mm Hg in the BBFC+TRAV group and 22.7 ± 2.4 mm Hg in the vehicle+TRAV group ( Table 2 ). After 6 weeks of adjunctive therapy, LS mean ± standard error diurnal IOP was 17.6 ± 0.4 mm Hg for patients receiving BBFC+TRAV compared with 20.7 ± 0.4 mm Hg for patients receiving vehicle+TRAV (between-group difference, −3.2 ± 0.5 mm Hg; 95% confidence interval, −4.2 to −2.2 mm Hg; P < .0001; Figure 3 ). The criterion for superiority of BBFC+TRAV was met. At week 6, the mean diurnal IOP change from TRAV-treated baseline and the mean percent diurnal IOP change from TRAV-treated baseline were significantly greater with BBFC+TRAV compared with vehicle+TRAV, and mean IOP at individual time points was lower with BBFC+TRAV compared with vehicle+TRAV ( P < .0001; Table 3 ). At each time point (peak efficacy, 10 AM and 5 PM; trough efficacy, 8 AM and 3 PM) of the week 2 and week 6 visits, mean IOP was lower, and mean and mean percent IOP change from TRAV-treated baseline were greater in the BBFC+TRAV group compared with the vehicle+TRAV group ( P < .0001; Table 4 ). The largest IOP changes from baseline were observed in patients receiving BBFC+TRAV at the 10 AM peak efficacy time point. At the week 6, 10 AM time point, mean ± SD IOP was lower with BBFC+TRAV compared with vehicle+TRAV (16.7 ± 3.3 mm Hg vs 20.5 ± 4.3 mm Hg). Mean IOP change from TRAV-treated baseline and mean percent IOP change from TRAV-treated baseline at the week 6, 10 AM time point were −6.2 ± 3.3 mm Hg and −26.7% ± 13.5% with BBFC+TRAV compared with −2.2 ± 3.8 mm Hg and −9.7% ± 16.2% with vehicle+TRAV ( Table 4 ).
| Brinzolamide 1%/Brimonidine 0.2% + Travoprost 0.004% (n = 103) | Vehicle + Travoprost 0.004% (n = 112) | Between-Group Difference | 95% Confidence Interval | P Value a | |
|---|---|---|---|---|---|
| Mean intraocular pressure, mm Hg | |||||
| 8 AM | 19.7 ± 0.4 | 22.0 ± 0.4 | −2.3 ± 0.5 | −3.3 to −1.2 | <.0001 |
| 10 AM | 16.7 ± 0.4 | 20.8 ± 0.4 | −4.1 ± 0.5 | −5.2 to −3.1 | <.0001 |
| 3 PM | 17.6 ± 0.4 | 19.7 ± 0.4 | −2.1 ± 0.5 | −3.2 to −1.1 | <.0001 |
| 5 PM | 15.7 ± 0.4 | 20.1 ± 0.4 | −4.4 ± 0.5 | −5.5 to −3.4 | <.0001 |
| Diurnal intraocular pressure | |||||
| Mean change from travoprost 004%–treated baseline, mm Hg | −5.0 ± 0.3 | −2.0 ± 0.3 | −3.0 ± 0.4 | −3.8 to −2.2 | <.0001 |
| Mean percent change from travoprost 004%–treated baseline | −21.9 ± 1.2 | −8.8 ± 1.2 | −13.0 ± 1.7 | −16.4 to −9.7 | <.0001 |
a P values were generated using repeated-measures analysis of variance models that accounted for covariate baseline intraocular pressure with fixed effect terms for treatment, visit, and treatment-by-visit interaction; compound symmetry covariance matrix; and Kenward-Roger method for denominator degrees of freedom.
| Brinzolamide 1%/Brimonidine 0.2% + Travoprost 0.004% | Vehicle + Travoprost 0.004% | |||||||
|---|---|---|---|---|---|---|---|---|
| n | Mean Intraocular Pressure, mm Hg | Change From Baseline | n | Mean Intraocular Pressure, mm Hg | Change From Baseline | |||
| Mean | Percent | Mean | Percent | |||||
| Travoprost monotherapy | ||||||||
| Baseline | ||||||||
| 8 AM | 113 | 24.2 ± 2.3 | – | – | 116 | 24.2 ± 2.4 | – | – |
| 10 AM | 113 | 22.8 ± 2.9 | – | – | 116 | 22.8 ± 2.8 | – | – |
| 3 PM | 113 | 21.5 ± 3.0 | – | – | 116 | 21.9 ± 3.0 | – | – |
| 5 PM | 113 | 21.3 ± 3.0 | – | – | 116 | 21.6 ± 3.0 | – | – |
| Mean diurnal | 113 | 22.5 ± 2.5 | – | – | 116 | 22.7 ± 2.4 | – | – |
| Adjunctive therapy | ||||||||
| Week 2 | ||||||||
| 8 AM | 112 | 19.5 ± 3.5 | −4.7 ± 3.2 | −19.1 ± 12.9 | 115 | 21.6 ± 4.7 a | −2.6 ± 3.7 a | −10.9 ± 14.4 a |
| 10 AM | 107 | 16.5 ± 3.6 | −6.3 ± 3.2 | −27.6 ± 13.3 | 114 | 20.2 ± 3.7 a | −2.4 ± 3.5 a | −10.3 ± 14.9 a |
| 3 PM | 108 | 17.5 ± 3.1 | −4.0 ± 2.9 | −18.1 ± 12.5 | 113 | 19.6 ± 3.8 a | −2.3 ± 3.5 a | −9.9 ± 15.0 a |
| 5 PM | 107 | 15.3 ± 3.3 | −6.0 ± 3.3 | −27.8 ± 13.7 | 112 | 19.7 ± 3.7 a | −1.8 ± 3.3 a | −7.7 ± 14.5 a |
| Mean diurnal | 113 | 17.4 ± 3.0 | −5.1 ± 2.7 | −22.5 ± 11.3 | 115 | 20.5 ± 4.4 a | −2.1 ± 3.4 a | −9.2 ± 13.6 a |
| Week 6 | ||||||||
| 8 AM | 103 | 19.7 ± 3.7 | −4.6 ± 3.5 | −18.7 ± 13.7 | 112 | 21.6 ± 4.4 a | −2.5 ± 3.4 a | −10.6 ± 14.3 a |
| 10 AM | 103 | 16.7 ± 3.3 | −6.2 ± 3.3 | −26.7 ± 13.5 | 112 | 20.5 ± 4.3 a | −2.2 ± 3.8 a | −9.7 ± 16.2 a |
| 3 PM | 103 | 17.6 ± 3.2 | −3.9 ± 3.1 | −17.5 ± 13.5 | 112 | 19.4 ± 3.8 a | −2.4 ± 3.3 a | −10.6 ± 14.7 a |
| 5 PM | 103 | 15.7 ± 3.4 | −5.7 ± 3.3 | −26.2 ± 14.2 | 112 | 19.8 ± 4.0 a | −1.7 ± 3.6 a | −7.3 ± 16.4 a |
| Mean diurnal | 103 | 17.4 ± 2.8 | −5.1 ± 2.6 | −22.3 ± 10.8 | 112 | 20.3 ± 3.7 a | −2.2 ± 2.9 a | −9.6 ± 12.9 a |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


