Purpose
To compare a new insertion method using an EndoGlide (Angiotech/Network Medical Products) with the standard Busin glide (Moria USA) assisted insertion of the posterior lamellar corneal graft in Descemet stripping endothelial keratoplasty (DSEK).
Design
Prospective, consecutive, comparative, nonrandomized study.
Methods
Surgery was performed between October 2008 and October 2010 in 52 eyes of 52 consecutive patients with endothelial dysfunction suitable for DSEK. Twenty-two consecutive eyes underwent the new EndoGlide-assisted insertion of donor lenticule and 30 eyes underwent the surgery using the Busin glide. Six-month follow-up data are available for all patients. Clinical details, best-corrected visual acuity, manifest refraction, intraoperative and postoperative complications, and corneal endothelial cell loss were assessed at 6 months after DSEK.
Results
At 6 months after surgery, the best-corrected visual acuity was similar in both groups (0.13 logarithm of the minimal angle of resolution in the EndoGlide eyes as compared with 0.15 logarithm of the minimal angle of resolution in the Busin group; P = .34). Mean spherical equivalent was 0.65 diopter (D) and 0.51 D, and mean refractory cylinder was 1.39 D and 1.08 D, respectively ( P = .40). The endothelial cell loss was much lower in the EndoGlide group (25.76%) as compared with the Busin group (47.46%; P < .0001).
Conclusions
In conclusion, the new EndoGlide results in significantly less endothelial cell loss than Busin glide donor insertion in DSEK. The visual outcomes and refractive changes were similar in both groups.
From its inception more than a decade ago, Descemet stripping endothelial keratoplasty (DSEK) has become the gold standard technique for eyes requiring corneal transplantation because of endothelial dysfunction. Although DSEK now reliably provides good visual outcomes with low risk of complications, the procedure continues to evolve as surgeons develop new techniques and instrumentation to facilitate the surgery and further improve safety and efficacy.
Although DSEK offers the advantage of a tectonically stronger cornea, reduced astigmatism, and quicker rehabilitation as compared with penetrating keratoplasty, the main concern relating to this operation is intraoperative donor endothelial damage. There has been significant interest in strategies for minimizing surgical DSEK graft manipulation to reduce endothelial cell loss (ECL). At 6 to 12 months, this has been reported to be in the range of 13% to 61%.
Various methods of inserting the donor graft into the anterior chamber (AC) have been described in DSEK. One of the most common techniques involves folding the donor corneal lenticule in a so-called taco fold and inserting it into the AC using various types of noncompressing forceps and suture techniques. However, with these techniques, the corneal endothelium is subjected to trauma as a result of the initial folding and later during the unfolding of the graft in the AC. Other insertion techniques involve the use of glides, including the Busin glide (Moria USA, Doylestown, Pennsylvania, USA; Figure ). The Busin glide was designed to avoid folding of the donor endothelium and, as the lenticule usually unfolds spontaneously within the AC, manipulation is reduced, resulting in a less traumatic insertion and greater endothelial cell survival. Atraumatic insertion of the donor lenticule by this method still is technically challenging. The lenticule easily can be pushed backward and occasionally off the Busin glide, either when attempting to grasp the edge with forceps or by irrigating fluid from the AC. This results in additional graft manipulation and trauma. In addition, because many surgeons position the glide slightly outside the incision to avoid AC collapse, endothelial cell damage may occur when the lenticule is compressed as it is pulled through the incision. Alternatively, if the Busin glide is advanced through the incision to avoid this compression, the AC can collapse, which may result in touch between the graft and AC structures on insertion.
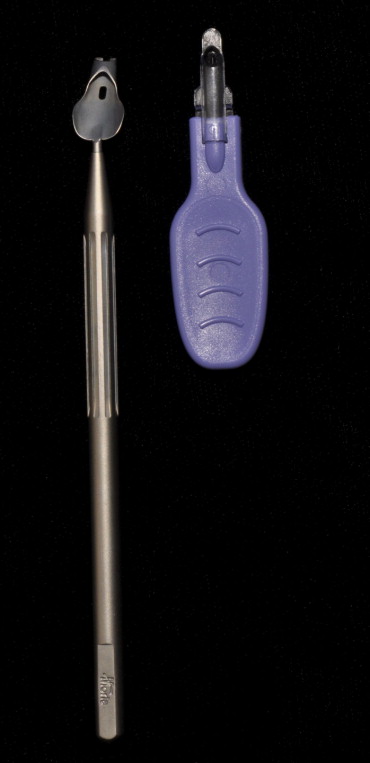
Recently, an EndoGlide (Angiotech, Reading, Pennsylvania, USA/Network Medical Products, North Yorkshire, UK; Figure ) has been introduced in an attempt to reduce the endothelial damage during donor lenticule insertion in DSEK surgery. The tip of this device is inserted into the AC to create a closed system, which prevents fluid flowing out through the glide or the incision. This addresses the concerns of graft compression by the corneoscleral wound and prevents AC collapse. The graft is delivered in the correct orientation with little need of manipulation. These advantages are proposed to reduce the endothelial cell damage. The aim of this prospective study is to compare, for the first time, the ECL with the new EndoGlide with the Busin glide for the insertion of donor lenticule during DSEK in a consecutive series of patients.
Methods
This prospective, comparative, nonrandomized study included 52 consecutive DSEK patients with at least 6 months follow-up. All surgeries were performed as day cases under local or general anesthesia at 1 institution by 1 surgeon (E.J.H.).
We included patients with Fuchs endothelial dystrophy, pseudophakic bullous keratopathy, and failed grafts resulting from endothelial dysfunction. We excluded any patients with iridocorneal endothelial syndrome or glaucoma tubes and eyes with severely disrupted anterior segment morphologic features, because these complex patients were unevenly distributed, with more in the Busin group. We also excluded any patients for whom confocal microscopy could not be performed.
Before surgery, all patients had visually significant corneal edema secondary to endothelial dysfunction. They underwent a complete evaluation including measurement of best-corrected visual acuity (BCVA), slit-lamp examination, intraocular pressure (IOP) measurement, and dilated funduscopy. The preoperative endothelial cell density was provided by the eye bank using specular microscopy and the corneal button was stored in Eagles medium with 2% bovine calf serum. This then was transferred to Dextran medium (Cornea transplant service eye bank, Bristol, United Kingdom) for transportation. The endothelial cell count was performed in the eye bank before transfer to Dextran medium and 3 days before transportation to the hospital.
Surgical Technique
Donor preparation
A deep manual dissection of the corneoscleral donor tissue using the Melles technique on a disposable artificial anterior chamber (Katena Products Inc, Denville, NJ, USA) was performed to dissect the donor endothelial graft. A corneal trephine with a diameter of 8.5 or 8.75 mm was used to punch the donor from the endothelial side down. This was stored under culture medium until required.
Recipient
The same corneal trephine was used to make a circular mark on the central epithelial surface of the recipient cornea with gentian violet ink. A temporal clear corneal tunnel was made using a 3.2-mm keratome (Alcon, Fort Worth, Texas, USA). An AC maintainer (M2656; Sterimedix, Redditch, UK) connected to a balanced salt solution (BSS) infusion line was inserted via a paracentesis at the inferonasal limbus. A long limbal tunnel was made nasally 180 degrees opposite the corneal tunnel with a microvitreoretinal blade (Alcon) and a further paracentesis was made at the limbus at the 8-o’clock position for the right eye and at the 2-o’clock position for the left eye using a 15-degree blade. Descemet membrane was stained with 0.01% trypan blue dye (Visiblue; Dutch Ophthalmic research centre, VN Zuidland, Netherlands) and scored using a blunt-tip reverse Sinskey hook (Bausch & Lomb, St. Louis, Missouri, USA) using the epithelial mark as a guide. Descemet membrane was stripped off using Melles Descemet strippers and was removed with a Macpherson forceps. The incision then was enlarged to 4.2 mm for the Busin technique and to 5 mm for the EndoGlide. The first 30 patients had the donor inserted using the Busin glide, then the EndoGlide was used for 22 cases.
Donor insertion using the new EndoGlide (n = 22)
The donor lenticule was separated from the anterior lamella by gentle irrigation of BSS using a Rycroft canula and was transferred together onto the donor well of the glide, endothelial side facing up. The internal lumen of the glide capsule was filled with BSS. A small amount of dispersive viscoelastic was placed on the endothelial surface. The graft was pulled into the glide capsule by introducing the straight-loading forceps (Angiotech; Network Medical Products, Reading, Pennsylvania, USA) by holding the leading stromal edge of the posterior donor lenticule. As the graft was drawn slowly into the glide capsule, the sides coiled upward to adopt a double-coil configuration, which was facilitated by using a Sinskey hook on the stromal side of the lenticule. The graft was drawn forward, fully coiled, until the leading edge reached the anterior opening of the glide capsule. The glide introducer was locked into the posterior opening of the glide capsule, and the entire complex was removed from the preparation base and inverted right side up for insertion into the AC. The EndoGlide was inserted smoothly until the anterior edge was seen through the cornea to be fully in the AC. The contralateral hand inserted the angled forceps (Angiotech, Network Medical Products) through the nasal paracentesis to pull the graft into the AC. The leading edge of the graft was held after it uncoiled and a small bubble of air was inserted before releasing the graft.
Donor insertion with the Busin glide (n = 30)
The donor endothelial lenticule was inserted using the Busin glide and Busin forceps (Moria, Antony, France). The donor lenticule was separated from the anterior lamellar surface and transferred onto the glide endothelial side up, with the aid of Macpherson forceps or BSS in a Rycroft cannula, and a small amount of viscoelastic was placed on the endothelial surface. The Busin forceps was inserted into the glide up to the slot to grasp the donor button, which then was pulled into the glide opening. The glide then was inverted and positioned at the entrance of the corneal tunnel. The Busin forceps were passed via the nasal incision to grasp the edge of donor lenticule and to pull it into the AC.
Donor positioning
The donor lenticule was positioned using BSS. The corneal tunnel was sutured with 3 interrupted 10–0 nylon sutures and the AC maintainer site was closed with 1 suture in all cases in both the groups. Filtered air was injected into the AC, and if necessary, the endothelial graft was manipulated into a central position using stroking motions with a Simcoe canula (attached to the infusion line) across the anterior epithelial surface. After good centration was achieved, the AC was filled completely with filtered air to achieve a high IOP for 10 minutes. During this period, any fluid was squeezed out from the graft–host interface with the Simcoe canula by massaging the epithelial corneal surface. The size of the air bubble then was reduced to approximately 8.5 mm in diameter with a physiologic IOP.
The patient was left supine in the postoperative recovery area for 2 hours. The patient was checked after surgery for pupil block. After surgery, all patients were given Chloramphenicol eye drops 4 times daily for 4 weeks and a tapering dose of Dexamethasone 0.1% eye drops starting hourly and reducing over the first 2 weeks. Thereafter, topical steroids were used 4 times daily for 2 months, 3 times daily for 2 months, twice daily for 2 months, and then once daily.
After surgery, all patients were followed up at 1 week and 1, 3, and 6 months. Key postoperative measurements included visual acuities (Snellen uncorrected visual acuity and BCVA), slit-lamp examination, ultrasonic pachymetry, IOP measurement, and confocal microscopy (Confoscan 4; Nidek Technologies Srl, Albignasego, Padova, Italy). The confocal microscopy was performed by an independent and experienced ophthalmic technician, masked to the graft insertion technique.
Statistical Analysis
The unpaired t test was used to compare the mean values of different groups, unless assumption tests showed that the groups had significantly different standard deviations or that they did not have a Gaussian distribution, in which case a nonparametric test (Mann–Whitney) was selected. A P value of less than .05 was considered statically significant. All data analysis was carried out using InStat software (GraphPad, La Jolla, California, USA).
Results
Fifty-two eyes of 52 patients were included in this study. Twenty-two eyes underwent the new EndoGlide glide-assisted DSEK and 30 eyes underwent the Busin glide-assisted DSEK. Table 1 presents the preoperative data, and Table 2 shows the operative data. Data analysis of visual outcome, endothelial cell count, and complication rates were performed at 6 months of follow-up.
| EndoGlide (n = 22) | Busin Glide (n = 30) | |
|---|---|---|
| Average age (SD), yrs | 74.27 (10.78) | 73.60 (9.8) |
| Sex | ||
| Female | 43% | 63.6% |
| Male | 57% | 36.36% |
| Reason for transplantation | ||
| Pseudophakic bullous keratopathy | 6 (27%) | 9 (30%) |
| Fuchs endothelial dystrophy | 15 (68.1%) | 21 (70%) |
| Failed DSEK | 1 (4.54%) | 0 |
| Other comorbidity | ||
| Cataract | 9 (40.9%) | 10 (33.3%) |
| Glaucoma | 2 | 3 |
| Amblyopia | 1 | 1 |
| Diabetic retinopathy | 0 | 1 |
| Retinal vein occlusion | 1 | 1 |
| Epiretinal membrane | 0 | 1 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


