Purpose
To investigate agreement in intraocular pressure (IOP) measurements among Icare rebound tonometry, Tonopen tonometry, and Goldmann applanation tonometry following vitreoretinal surgery.
Design
Reliability analysis of tonometers.
Methods
Fifty-eyes of 50 adults undergoing vitreoretinal surgery were enrolled. IOP was measured on first postoperative day using Icare (Tiolat, Helsinki, Finland), followed by Tonopen (Reichert, Depew, New York, USA) and Goldmann (Haag-Streit USA, Mason, Ohio, USA) in randomized order. Intraclass correlation coefficients (ICC) and Bland-Altman plots were calculated for all subcategories.
Results
Icare successfully measured IOP in all eyes, while Goldmann was unmeasurable in 6 eyes (12%) and Tonopen in 1 eye (2%). Mean IOP by Icare, Tonopen, and Goldmann was 15.9 ± 8.9, 16.9 ± 6.2, and 16.0 ± 7.3 mm Hg, respectively ( P = .76). Type of intraocular tamponade, status of lens, status of cornea, gauge of instrumentation, and history of prior vitrectomy did not result in significant differences among the 3 tonometers. ICC was excellent (>0.75) in all subgroups, except at IOP <10 and ≥23 mm Hg (based on Icare). In eyes with IOP <10 mm Hg, Icare underestimated IOP (mm Hg; P = .01) compared to Goldmann (2.0 ± 2.1) and Tonopen (3.5 ± 2.4), whereas at IOP ≥23 mm Hg Icare was overestimated ( P = .01) compared to Goldmann (3.77 ± 3.49) and Tonopen (4.97 ± 3.33). Overall, differences in IOP were ≤3 mm Hg in 58% of eyes for Icare-Tonopen, 72% for Tonopen-Goldmann, and 62% for Icare-Goldmann.
Conclusion
IOP measurements using Icare rebound tonometry, Tonopen, and Goldmann tonometry are in excellent agreement following vitreoretinal surgery. However, Icare overestimates at IOP ≥23 and underestimates at IOP <10 mm Hg.
Accurate measurement of intraocular pressure (IOP) after vitreoretinal surgery is important, as the incidence of elevated IOP postoperatively has been reported to range from 5% to 35%, and inaccurate detection of elevated IOP in the immediate postoperative period can result in irreversible optic nerve damage.
The Goldmann applanation tonometer (GAT) is currently the gold standard for IOP measurement and is widely used in clinical practice. Because of the relatively large size of the applanation tip, GAT has technical challenges with the potential for inaccuracy after vitreoretinal surgery owing to edema of the eyelid or conjunctiva, pain, discharge, and corneal irregularities such as corneal epithelial defects. The Tonopen applanation tonometer is a portable applanation tonometry device with a smaller, 1 mm tip, that is often used in place of GAT for measurement of IOP following vitreoretinal surgery. Both devices require topical anesthesia. The use of intraocular tamponade agents such as air, tamponade gases, or silicone oil may also introduce potential measurement difficulties owing to tamponade-related changes in the physical properties of the eye.
The Icare rebound tonometer is a hand-held portable tonometer based on the induction rebound principle—making a moving object collide with an eye while monitoring its motion parameters. IOP is calculated from the measurement of impact duration—the higher the IOP, the shorter the duration of the impact. Several studies have compared the Icare rebound tonometer to the Tonopen and/or GAT in healthy adults, pediatric patients, and those with glaucoma and have concluded there is reasonable correlation between them, particularly when accounting for central corneal thickness (CCT).
The Icare rebound tonometer has not been evaluated in the postoperative period following vitrectomy surgery, which differs from other ocular surgeries owing to the frequent use of intraocular tamponade agents, frequent postoperative corneal epithelial defects, corneal edema, and lid edema. This study was designed to prospectively compare the Icare rebound tonometer with the Tonopen applanation tonometer and GAT in the measurement of IOP in eyes of adult patients in the immediate postoperative period following vitreoretinal surgery.
Methods
This was a reliability analysis study approved by the Duke University Institutional Review Board that adhered to the tenets set forth in the Declaration of Helsinki. Written informed consent was obtained from all participants. Patients were recruited prospectively and the study was registered on clinicaltrials.gov with registration identifier NCT01786954 . Inclusion criteria consisted of all adult patients undergoing vitrectomy surgery either with no tamponade (ie, fluid) or with gas or silicone oil as postoperative tamponade. Patients who declined participation in the study and those unable to provide informed consent were excluded.
IOP was measured on the first postoperative day using the Icare rebound tonometer (Tiolat, Helsinki, Finland), followed by the Tonopen applanation tonometer (Reichert, Depew, New York, USA) and GAT (Haag-Streit USA, Mason, Ohio, USA) in randomized order. The study duration was a single clinical visit on the first postoperative day. Clinical data recorded included age; sex; racial group; diagnosis; ocular history; ophthalmic examination findings, including IOP measurements by Icare rebound tonometer, GAT, and Tonopen applanation tonometer; and CCT measurements by ultrasonic pachymetry. A complete postoperative ophthalmic examination was performed, including careful examination of the corneal surface, as is standard clinical care. If a bandage contact lens had been placed intraoperatively, the contact lens was temporarily removed for measurement of IOP, as is standard of practice for accurate IOP measurement.
All subjects underwent IOP measurements in the sitting position. Measurement with the Icare rebound tonometer was performed first, prior to administration of topical anesthetic. IOP measurements with the Tonopen applanation tonometer and GAT and CCT measurements required topical anesthesia and were carried out after Icare rebound tonometer measurement. All measurements were obtained by a fully trained ophthalmologist in the vitreoretinal service at the Duke Eye Center. To obtain Icare rebound tonometer measurements, single-use probes were fitted into the Icare rebound tonometer device. Subjects were instructed to fixate on a distant target while the probe of the Icare rebound tonometer was held at a distance of 4–8 mm perpendicular to the central cornea. Icare obtains 6 measurements from each eye on the central cornea, with the highest and lowest values excluded and the average calculated internally by the instrument, yielding a mean reading with a standard deviation (SD). Only good-quality measurements, defined by the manufacturer as those with the solid letter P or a flashing letter P and a horizontal line at the bottom of the screen, were accepted.
Topical anesthesia was then administered (proparacaine 0.5% eye drops; Alcaine; Alcon, Fort Worth, Texas, USA) and patients were randomized to undergo Tonopen tonometry prior to GAT, or GAT prior to Tonopen tonometery. All measurements with GAT were obtained with the patient sitting erect at the slit lamp in standard fashion. The dial was preset to a value of 20 mm Hg and reset to 20 mm Hg after each reading to minimize subjectivity in measurement. Two subsequent readings were obtained in this fashion. The Tonopen tonometer device was calibrated before each patient assessment. The Tonopen obtains 4 valid readings and then displays the averaged measurement along with a bar denoting statistical reliability. Only readings with displayed SD of ≤5% were used. The IOP was measured in duplicate with each instrument (Icare, GAT, and Tonopen) before moving to the next. Following completion of IOP measurements, CCT was measured in duplicate using ultrasonic pachymetry (Micropach; Sonomed, New Hyde Park, New York, USA). The corneal surface was re-examined for the presence of any new epithelial defects following measurement of IOP and CCT.
Statistical Analysis
IOP measured with the Icare rebound tonometer was compared to Tonopen applanation tonometer and GAT measures using a t test of equivalence of means. An IOP difference of ≥3 mm Hg was predefined as clinically significant in our study. A sample size of 30 was calculated to have 90% power at a 0.05 significance level to reject the null hypothesis that the test (Icare rebound tonometer) and standard (Tonopen applanation tonometer and GAT) were not equivalent, assuming the SD of the differences to be 3.9 mm Hg.
The Bland-Altman method was used to examine the level of agreement between each pair of tonometers. Linear regression was used to examine the influence of IOP on tonometer bias (difference from GAT) and the relationship between CCT and IOP for each tonometer. Statistical analyses were performed using SAS 9.3 (SAS Institute, Cary, North Carolina, USA). The Kolmogorov-Smirnov test was used to assess normality. Data not normally distributed were analyzed using nonparametric statistical techniques. Statistical analysis included descriptive statistics, Student t test, paired t test, Kruskal-Wallis test of difference among medians, and simple linear regression for univariate analyses. A multivariate analysis of variance, Wilks lambda multivariate statistic, was used to assess the significance of the difference among tonometers after adjusting for the influence of CCT on IOP measurements. All statistical tests were 2-tailed and intraclass correlation coefficient (ICC) was calculated to assess agreement. P values <.05 were considered statistically significant.
Results
Fifty eyes of 50 patients were included. Clinical and demographic characteristics of the study population are summarized in the Table . The patients included those with no intraocular tamponade (ie, fluid; n = 17), SF 6 or C 3 F 8 gas tamponade (n = 22), and silicone oil tamponade (n = 11). The indications for vitrectomy included rhegmatogenous retinal detachment (n = 25), macular hole (n = 7), epiretinal membrane (n = 4), nonclearing vitreous hemorrhage (n = 3), tractional retinal detachment (n = 4), retained lens material following cataract surgery (n = 3), and silicone oil removal (n = 4).
| Parameter | All Vitreoretinal Surgeries (n = 50) | No Tamponade/ Fluid (n = 17) | Gas Tamponade (n = 22) | Silicone Oil Tamponade (n = 11) |
|---|---|---|---|---|
| Eye | OD = 19 OS = 31 | OD = 5 OS =12 | OD = 6 OS = 16 | OD = 8 OS = 3 |
| Age, mean ± SD (y) | 62.9 ± 13.5 | 62.6 ± 13.4 | 66.8 ± 9.8 | 49.6 ± 16.7 |
| Sex, m / f | 27 / 23 | 8 / 9 | 15 / 7 | 4 / 7 |
| Prior PPV | 13 | 4 | 2 | 5 |
| Lens status | Phakic = 17 Pseudophakic = 27 Aphakic = 6 | Phakic = 7 Pseudophakic = 8 Aphakic = 2 | Phakic =7 Pseudophakic = 15 | Phakic = 3 Pseudophakic = 4 Aphakic = 4 |
| Corneal epithelial defect/edema | Epi defect = 27 Epi defect and corneal edema = 3 | Epi defect = 7 | Epi defect =12 Epi defect and corneal edema = 1 | Epi defect = 8 Epi defect and corneal edema =1 |
| Central corneal thickness, mean ± SD (range) (microns) | 639.5 ± 98.5 (444–1103) | 635.2 ± 72.6 (444–749) | 608.1 ± 58.9 (511–784.50) | 709.1 ± 155.8 (590–1103) |
| Icare rebound tonometry IOP, mean ± SD (range) (mm Hg) | 15.9 ± 8.9 (2–38) | 15.2 ± 8.2 (4–36) | 15.2 ± 8.1 (2–30) | 18.4 ± 11.6 (3–38) |
| Goldmann applanation tonometry IOP, mean ± SD (range) (mm Hg) | 16.0 ± 7.3 (1–39) | 17.2 ± 7.4 (6–39) | 14.1 ± 6.7 (1–27) | 17.8 ± 8.2 (7–32) |
| Tonopen applanation tonometry IOP, mean ± SD (range) (mm Hg) | 16.9 ± 6.2 (5–35) | 17.0 ± 6.4 (6–35) | 16.6 ± 5.8 (5–26) | 17.6 ± 7.2 (5–30) |
Twenty-four of 50 eyes (48%) had an epithelial defect without corneal edema on the first postoperative day, of which 4 eyes (16.7%) had unmeasurable IOP using the GAT. Two of fifty eyes (4%) had an epithelial defect with visible corneal edema, and both had unmeasurable IOP using the GAT. Tonopen readings could not be obtained in 1 eye (2%) with low IOP (2 mm Hg using Icare rebound tonometer). Reliable IOP readings were successfully obtained in all eyes using the Icare rebound tonometer.
IOP (mean ± SD, mm Hg) in eyes undergoing vitreoretinal surgery was 15.9 ± 8.9 using Icare rebound tonometer, 16.92 ± 6.22 using Tonopen, and 16.01 ± 7.27 using GAT, with no difference among tonometers ( P = .76). Post hoc analysis based on type of intraocular tamponade revealed there was no difference in IOP using the 3 instruments in either fluid-, gas-, or silicone oil–filled eyes ( Supplemental Table 1 , available at AJO.com ). Similarly there was no difference based on the status of the cornea ( Supplemental Table 2 , available at AJO.com ) or of the lens ( Supplemental Table 3 , available at AJO.com ). Stratification of eyes with IOP <10, 10–22, 23–29, and ≥30 mm Hg based on Icare rebound tonometer measurements ( Supplemental Table 4 , available at AJO.com ) demonstrated similar readings among the 3 tonometers for eyes with IOP 10–22 mm Hg, but there were significant differences for the subgroups with IOP <10 mm Hg ( P = .01), 23–29 mm Hg ( P = .01), and ≥30 mm Hg ( P = .007). Icare rebound tonometer measured lower than Tonopen and GAT for IOP <10 mm Hg and measured higher than Tonopen and GAT for the 23–29 mm Hg and ≥30 mm Hg subgroups.
The ICC (95% confidence interval) was 0.88 (0.79–0.93), 0.91 (0.84–0.95), and 0.88 (0.79–0.93) for the Icare-Tonopen, Icare-GAT, and Tonopen-GAT pairs, respectively. Sub-analysis based on the type of intraocular tamponade revealed excellent agreement among the tonometers for fluid, gas, and silicone oil tamponade, with ICC of 0.86–0.91 for Icare-Tonopen, 0.90–0.91 for Icare-Goldmann, and 0.84–0.91 for Tonopen-GAT ( Supplemental Table 5 , available at AJO.com ). In eyes with an epithelial defect with visible corneal edema, ICC (0.45) was lower compared to eyes with a corneal epithelial defect without edema (0.84–0.87) and eyes without an epithelial defect (0.88–0.89) ( Supplemental Table 6 , available at AJO.com ). In aphakic eyes, the ICCs were lower (range 0.68–0.79) when compared to pseudophakic eyes (0.85–0.91) and phakic eyes (0.85–0.94) ( Supplemental Table 7 , available at AJO.com ). ICCs were excellent for all vitrectomy gauges (0.90–0.98 for 20 gauge, 0.76–0.87 for 23 gauge, and 0.94–0.95 for 25 gauge). History of prior vitrectomy surgery did not impact the ICC (0.85–0.95 for eyes with previous vitrectomy and 0.86–0.91 for eyes without).
Sub-analysis based on IOP measurements (using the Icare rebound tonometer) demonstrated good correlation (ICC range 0.60–0.89) for eyes with low IOP (<10 mm Hg) and normal IOP (between 10 and 22 mm Hg) ( Supplemental Table 8 , available at AJO.com ). However, correlation between Icare rebound tonometer and Tonopen was poor at mildly elevated IOP (between 23 and 29 mm Hg; ICC 0.17) and elevated IOP (≥30 mm Hg; ICC 0.33).
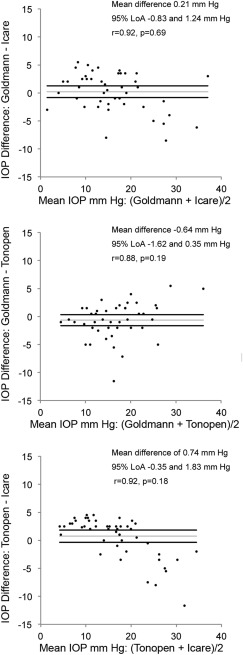
Bland-Altman plots, 95% limits of agreement (LoA), and the mean differences between the tonometers are provided in the Figure . These plots allowed us to determine the existence of any systematic differences between the measurements. The plots showed a trend that at higher IOPs, Icare read higher than the Goldmann and Tonopen, while the Tonopen read lower than Goldmann. This was confirmed using regression analysis, which showed a significant regression line for each tonometer pair. Bland-Altman sub-analysis in eyes with intraocular gas tamponade, with silicone oil tamponade, and with no tamponade ( Supplemental Figure 1 ) revealed that mean differences between tonometers were not different in all subgroups except for GAT-Icare rebound tonometer for the cohort without tamponade (r = 0.95, P = .008), with GAT measuring higher (1.97 mm Hg; 95% LoA 0.60–3.34 mm Hg). Bland-Altman sub-analysis was also performed based on the status of the cornea ( Supplemental Figure 2 ). The agreement among Tonopen and Icare rebound tonometer in eyes with epithelial defects and accompanying corneal edema (ICC 0.45) was lower compared to eyes with an epithelial defect but no corneal edema and without an epithelial defect (ICC range 0.84–0.87). In eyes with corneal edema, Icare (4.50 ± 2.12) under-read Tonopen (7.50 ± 2.83). Similarly, Bland-Altman sub-analysis was performed based on lens status ( Supplemental Figure 3 ). These showed that the agreement was lower in aphakic eyes (ICC range 0.68–0.79) compared to pseudophakic and phakic eyes (ICC range 0.85–0.94).
To evaluate the effect of CCT on measurements using the 3 tonometers, multivariate analysis of variance was performed, which demonstrated that there was no statistically significant effect of varying CCT on differences in IOP measurements between any of the 3 tonometers ( P = .39, Wilks lambda multivariate statistic).
Results
Fifty eyes of 50 patients were included. Clinical and demographic characteristics of the study population are summarized in the Table . The patients included those with no intraocular tamponade (ie, fluid; n = 17), SF 6 or C 3 F 8 gas tamponade (n = 22), and silicone oil tamponade (n = 11). The indications for vitrectomy included rhegmatogenous retinal detachment (n = 25), macular hole (n = 7), epiretinal membrane (n = 4), nonclearing vitreous hemorrhage (n = 3), tractional retinal detachment (n = 4), retained lens material following cataract surgery (n = 3), and silicone oil removal (n = 4).
| Parameter | All Vitreoretinal Surgeries (n = 50) | No Tamponade/ Fluid (n = 17) | Gas Tamponade (n = 22) | Silicone Oil Tamponade (n = 11) |
|---|---|---|---|---|
| Eye | OD = 19 OS = 31 | OD = 5 OS =12 | OD = 6 OS = 16 | OD = 8 OS = 3 |
| Age, mean ± SD (y) | 62.9 ± 13.5 | 62.6 ± 13.4 | 66.8 ± 9.8 | 49.6 ± 16.7 |
| Sex, m / f | 27 / 23 | 8 / 9 | 15 / 7 | 4 / 7 |
| Prior PPV | 13 | 4 | 2 | 5 |
| Lens status | Phakic = 17 Pseudophakic = 27 Aphakic = 6 | Phakic = 7 Pseudophakic = 8 Aphakic = 2 | Phakic =7 Pseudophakic = 15 | Phakic = 3 Pseudophakic = 4 Aphakic = 4 |
| Corneal epithelial defect/edema | Epi defect = 27 Epi defect and corneal edema = 3 | Epi defect = 7 | Epi defect =12 Epi defect and corneal edema = 1 | Epi defect = 8 Epi defect and corneal edema =1 |
| Central corneal thickness, mean ± SD (range) (microns) | 639.5 ± 98.5 (444–1103) | 635.2 ± 72.6 (444–749) | 608.1 ± 58.9 (511–784.50) | 709.1 ± 155.8 (590–1103) |
| Icare rebound tonometry IOP, mean ± SD (range) (mm Hg) | 15.9 ± 8.9 (2–38) | 15.2 ± 8.2 (4–36) | 15.2 ± 8.1 (2–30) | 18.4 ± 11.6 (3–38) |
| Goldmann applanation tonometry IOP, mean ± SD (range) (mm Hg) | 16.0 ± 7.3 (1–39) | 17.2 ± 7.4 (6–39) | 14.1 ± 6.7 (1–27) | 17.8 ± 8.2 (7–32) |
| Tonopen applanation tonometry IOP, mean ± SD (range) (mm Hg) | 16.9 ± 6.2 (5–35) | 17.0 ± 6.4 (6–35) | 16.6 ± 5.8 (5–26) | 17.6 ± 7.2 (5–30) |
Twenty-four of 50 eyes (48%) had an epithelial defect without corneal edema on the first postoperative day, of which 4 eyes (16.7%) had unmeasurable IOP using the GAT. Two of fifty eyes (4%) had an epithelial defect with visible corneal edema, and both had unmeasurable IOP using the GAT. Tonopen readings could not be obtained in 1 eye (2%) with low IOP (2 mm Hg using Icare rebound tonometer). Reliable IOP readings were successfully obtained in all eyes using the Icare rebound tonometer.
IOP (mean ± SD, mm Hg) in eyes undergoing vitreoretinal surgery was 15.9 ± 8.9 using Icare rebound tonometer, 16.92 ± 6.22 using Tonopen, and 16.01 ± 7.27 using GAT, with no difference among tonometers ( P = .76). Post hoc analysis based on type of intraocular tamponade revealed there was no difference in IOP using the 3 instruments in either fluid-, gas-, or silicone oil–filled eyes ( Supplemental Table 1 , available at AJO.com ). Similarly there was no difference based on the status of the cornea ( Supplemental Table 2 , available at AJO.com ) or of the lens ( Supplemental Table 3 , available at AJO.com ). Stratification of eyes with IOP <10, 10–22, 23–29, and ≥30 mm Hg based on Icare rebound tonometer measurements ( Supplemental Table 4 , available at AJO.com ) demonstrated similar readings among the 3 tonometers for eyes with IOP 10–22 mm Hg, but there were significant differences for the subgroups with IOP <10 mm Hg ( P = .01), 23–29 mm Hg ( P = .01), and ≥30 mm Hg ( P = .007). Icare rebound tonometer measured lower than Tonopen and GAT for IOP <10 mm Hg and measured higher than Tonopen and GAT for the 23–29 mm Hg and ≥30 mm Hg subgroups.
The ICC (95% confidence interval) was 0.88 (0.79–0.93), 0.91 (0.84–0.95), and 0.88 (0.79–0.93) for the Icare-Tonopen, Icare-GAT, and Tonopen-GAT pairs, respectively. Sub-analysis based on the type of intraocular tamponade revealed excellent agreement among the tonometers for fluid, gas, and silicone oil tamponade, with ICC of 0.86–0.91 for Icare-Tonopen, 0.90–0.91 for Icare-Goldmann, and 0.84–0.91 for Tonopen-GAT ( Supplemental Table 5 , available at AJO.com ). In eyes with an epithelial defect with visible corneal edema, ICC (0.45) was lower compared to eyes with a corneal epithelial defect without edema (0.84–0.87) and eyes without an epithelial defect (0.88–0.89) ( Supplemental Table 6 , available at AJO.com ). In aphakic eyes, the ICCs were lower (range 0.68–0.79) when compared to pseudophakic eyes (0.85–0.91) and phakic eyes (0.85–0.94) ( Supplemental Table 7 , available at AJO.com ). ICCs were excellent for all vitrectomy gauges (0.90–0.98 for 20 gauge, 0.76–0.87 for 23 gauge, and 0.94–0.95 for 25 gauge). History of prior vitrectomy surgery did not impact the ICC (0.85–0.95 for eyes with previous vitrectomy and 0.86–0.91 for eyes without).
Sub-analysis based on IOP measurements (using the Icare rebound tonometer) demonstrated good correlation (ICC range 0.60–0.89) for eyes with low IOP (<10 mm Hg) and normal IOP (between 10 and 22 mm Hg) ( Supplemental Table 8 , available at AJO.com ). However, correlation between Icare rebound tonometer and Tonopen was poor at mildly elevated IOP (between 23 and 29 mm Hg; ICC 0.17) and elevated IOP (≥30 mm Hg; ICC 0.33).
Bland-Altman plots, 95% limits of agreement (LoA), and the mean differences between the tonometers are provided in the Figure . These plots allowed us to determine the existence of any systematic differences between the measurements. The plots showed a trend that at higher IOPs, Icare read higher than the Goldmann and Tonopen, while the Tonopen read lower than Goldmann. This was confirmed using regression analysis, which showed a significant regression line for each tonometer pair. Bland-Altman sub-analysis in eyes with intraocular gas tamponade, with silicone oil tamponade, and with no tamponade ( Supplemental Figure 1 ) revealed that mean differences between tonometers were not different in all subgroups except for GAT-Icare rebound tonometer for the cohort without tamponade (r = 0.95, P = .008), with GAT measuring higher (1.97 mm Hg; 95% LoA 0.60–3.34 mm Hg). Bland-Altman sub-analysis was also performed based on the status of the cornea ( Supplemental Figure 2 ). The agreement among Tonopen and Icare rebound tonometer in eyes with epithelial defects and accompanying corneal edema (ICC 0.45) was lower compared to eyes with an epithelial defect but no corneal edema and without an epithelial defect (ICC range 0.84–0.87). In eyes with corneal edema, Icare (4.50 ± 2.12) under-read Tonopen (7.50 ± 2.83). Similarly, Bland-Altman sub-analysis was performed based on lens status ( Supplemental Figure 3 ). These showed that the agreement was lower in aphakic eyes (ICC range 0.68–0.79) compared to pseudophakic and phakic eyes (ICC range 0.85–0.94).




