Photorefractive Keratectomy
CHAPTER CONTENTS
PREOPERATIVE CONSIDERATIONS
Indications
- recurrent corneal erosion
- Reis-Bücklers dystrophy
- map-dot fingerprint or stromal corneal dystrophies (e.g., granular, macular, or lattice dystrophies)
- keratoconus (specific cases with stable refraction, limited astigmatism, and normal pachymetry)
- laser adjustment after keratoplasty, clear lens extraction, or cataract extraction
Inclusion Criteria
- realistic patient expectations
- clinically normal eyes
- patient age greater than 18 years (required for refractive stability)
- postoperative pachymetry not less than 300 μm (ablation depths calculated preoperatively)
- postoperative keratometry not less than 36 D (ablation depths calculated preoperatively)
- scotopic pupil size less than 8 mm for an excimer laser [provides an optical zone (OZ) <6 mm with no transition zone capability]
- good general health (patients with diabetes or HIV treatable if health permits but do not perform simultaneous bilateral surgery; see Contraindications)
Patient Evaluation
- manifest refraction
- measurement of best-corrected visual acuity (BCVA) using Snellen’s charts
- evaluation of pupil size in scotopic conditions using a Colvard pupillometer (model no. 0401; Oasis Medical)
- corneal topography
- slit-lamp examination of anterior segment
- fundus eye examination (for myopia >5 D)
- assessment of contrast sensitivity visual acuity under controlled luminance
- evaluation of halos or glare
Patient Preparation
- Instruct patient to remove soft contact lenses 2 to 3 days before the preoperative examination.
- For rigid gas-permeable contact lens wearers, verify the stability of topography before surgery (stability may take 4-6 weeks to occur).
SURGICAL CONSIDERATIONS
Contraindications
- relative contraindications
- scotopic pupil size greater than 8 mm (greater risk of having postoperative glare and halos)
- human immunodeficiency virus (HIV) (depends on general health status)
- diabetes (depends on general health status)
- infections that call for surgery to be delayed (e.g., a common cold, sinusitis, and other infectious illnesses)
- ocular pathology (especially autoimmune disease)
- dry-eye syndrome [often associated with systemic diseases such as sarcoidosis, mucin deficiency (e.g., Stevens-Johnson syndrome), avitaminose A, lipid deficiency (blepharitis), and decreased blinking (from contact lens use and post-herpes simplex keratitis)]
- scotopic pupil size greater than 8 mm (greater risk of having postoperative glare and halos)
- absolute contraindications
- unrealistic patient expectations about surgical “guarantees” (e.g., that surgery will not harm vision, the outcome will be piano, or spectacles or contacts will no longer be necessary)
- unstable or progressive myopia or hyperopia
- amblyopia or strabismus (inform patients with very low fusional amplitudes that the procedure will not improve their BCVA)
- unrealistic patient expectations about surgical “guarantees” (e.g., that surgery will not harm vision, the outcome will be piano, or spectacles or contacts will no longer be necessary)
General Methods
- Calibrate the laser for the exact ablation rate according to the excimer laser manufacturer’s directions.
- Give an oral analgesic to the patient 15 to 30 min before surgery.
- Instill 1 or 2 drops of ofloxacin 0.3% drops (Floxin or Ocuflox) 5 min before and 1 to 2 drops seconds before insertion of lid speculum.
- Instill 1 or 2 drops of 0.5% proparacaine hydrochloride (Alcaine or Ophthaine) 15 min before and a few more drops seconds before lid speculum insertion.
- Anesthetize the cornea and surrounding tissues, including the palpebral conjunctiva and edge of the eyelid, with a soaked cotton swab of 0.5% proparacaine hydrochloride.
- Place the head of the patient in the supine position with the chin higher than the forehead as you look into the microscope (rotate the patient’s head with your hands if needed).
- Insert lid speculum.
- Because the upper eyelid may push debris into the ablation zone, you should be able to see the upper part of the limbus under the speculum (debris and tear stream are evacuated toward the upper limbus by microblinking the eye).
- Prevent eyelashes from entering the path of the laser beam with tape, if necessary.
- Carefully dry the conjunctival sac (cul-de-sac) (a potential hazard for infection).
- Perform epithelial debridement with an Amoils epithelial scrubber or laser transepithelial removal.
- Gently wipe away any remaining epithelial debris with an eye spear.
- Absorb all excess fluid with a sponge.
- Determine the total amount of myopia and astigmatism to correct using a laser-specific nomogram and your own experience.
- Perform multipasses to divide the total treatment by specific nomograms (see Nomograms).
- Do not exceed 20 sec per division for laser treatment.
- Cool the cornea by adding chilled balanced saline solution (BSS).
- Administer medications immediately after the procedure.
- 2 drops of 0.5% ketorolac tromethamine (Acular)
- 2 drops of 0.3% ofloxacin
- Place a soft disposable contact lens on the eye (Fig. 9-1).
- Instruct the patient to wear sunglasses as protection from ultraviolet (UV) light.
- Ensure the quality of the sunglasses for blocking UV light.
- Perform multipasses to divide the total treatment by specific nomograms (see Nomograms).
Nomograms
THE MULTIZONE AND MULTIPASS TECHNIQUE The first multizone technique for PRK was developed to decrease the ablation depth for broad-beam excimer lasers. Later, the multizone/multipass technique was designed for use with these lasers to improve refractive results.
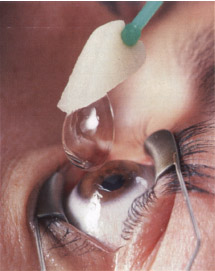
Figure 9-1 Contact lens insertion after PRK.
Methods
- initial pretreatment at a 2.5-mm diameter of 1.0 or 1.5 D (depending on the degree of myopia)
- division of remainder of treatment into several small corrections or passes between 3.5 and 6.0 mm in diameter
- correction of an equal amount of sphere at the same laser pass
- performance of cylinder correction at a diameter of 5.0 mm*
- stoppage of broad-beam laser and resetting of diaphragm to zero between sub-treatments
- performance of multiple passes over the same OZ
- a slight shifting of zones
- a smoother ablated surface with fewer ridges on the ablated surface
- smoother concentric steps
- less potential for tissue deposits
- a smoother ablated surface with fewer ridges on the ablated surface
- laser diaphragm (increases in size during the treatment and delimits zones)
- smaller treatments
- use of a specific algorithm titrated to the level of myopia (low to moderate)
- each ablative pass (≤ 7 total) lasts 10 to 30 sec
- use of a specific algorithm titrated to the level of myopia (low to moderate)
- treatment of astigmatism with an elliptical ablation combined with multizone/ multipass ablations
THE SCANNING MULTIPASS TECHNIQUE This technique is derived from the initial multizone/multipass technique and may be used with a scanning laser [e.g., Bausch and Lomb (Rochester, NY) 217 laser or Laser-Sight LSX].
* See Pop and Aras (1995) for a description of multizone/multipass algorithms.
Advantages
- use for low to high myopia or hyperopia (use laser- and surgeon-specific nomograms to adjust the total amount of myopia and astigmatism to correct by determining the fixed ratio that indicates the coefficient to apply to actual sphere and cylinder to obtain the desired sphere and cylinder corrections)
- no need for pretreatment for central island
- use of 6-mm OZ with a transition zone of 9 mm (large transitional zones are used to smooth the curve between the actual treatment zone and the remaining untouched corneal stroma)
- elimination of the need to perform multi-zones
- generation of multiple scanning of the corneal surface while removing stroma
- performance of equal passes so that the initial sphere is divided by equal but smaller treatments (sum of small treatments equals target correction)
- introduction of very small rest periods between subtreatments
- decreased haze for high myopes
- generation of multiple scanning of the corneal surface while removing stroma
- passes (≤6 total) should each last 20 to 30 sec (do not exceed 30 sec per pass)
- no need to wet the surface between passes
Disadvantages
- potential for calculation errors (if not automated)
- dehydration of corneal stromal (if excessive time between passes causing overcorrection)
Myopia
Equipment Preparation
- Configure the laser to treat all astigmatism (generally, 10-30% is removed from the computed sphere; may vary with different types and brands of lasers).
Nomogram
- patients less than 40 years old
- removal of 0.25 D of astigmatism to the computed sphere per diopter of cylinder [e.g., −6.00 − 1.00 × 45 degrees = 30% (2 D) and +0.25 D removed to the sphere; because of 1 D of cylinder; the computed ablation = –3.75 – 1.00 x 45 degrees)
- patients greater than 40 years old
- removal of an additional 0.25 D to the computed sphere
Results
- myopia less than 4 D
- 97% of eyes within ±1 D of emmetropia
- 4% needing retreatment
- no eyes with clinically significant haze
- no eyes with loss of more than 1 line of BCVA
- 97% of eyes within ±1 D of emmetropia
- myopia from 4 to 8 D
- 95% of eyes within ± 1 D of emmetropia
- 9% needing retreatment
- 0.2% of eyes with mild to moderate haze
- no eyes with loss of more than 1 line of BCVA
- 95% of eyes within ± 1 D of emmetropia
- myopia from 8 to 10 D
- 83% of eyes within ±1 D of emmetropia
- 12% needing retreatment
- 2% of eyes with mild to moderate haze
- 0.6% of eyes with loss of more than 1 line of BCVA
- 83% of eyes within ±1 D of emmetropia
- myopia greater than 10 D
- 80% of eyes within ± 1D of emmetropia
- 14% needing retreatment
- 2% of eyes with mild to moderate haze
- 1.4% of eyes with loss of more than 1 line of BCVA
- 80% of eyes within ± 1D of emmetropia
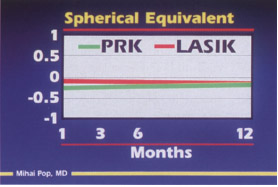
- equal results 1 month after surgery with photorefractive keratectomy (PRK) or laser in situ keratomileusis (LASIK) for myopia less than 10 D (Fig. 9-2)
Hyperopia
Equipment Preparation
- Set the slit-scanning laser to a repetition rate of 41 Hz with a polymethylmethacrylate/cornea ratio of 1.0 and a cylinder/sphere time rate of 0.45 (observe ~12 sec/D of correction).
Results
- hyperopia less than +3 D
- 92% of eyes within ±1 D of emmetropia
- 8% needing retreatment
- 3% of eyes with mild to moderate haze
- regression of +0.75 to +1 D
- 2% of eyes with loss of more than 1 line of BCVA
- 92% of eyes within ±1 D of emmetropia
Compound Astigmatism
Methods
- Perform a cross-cylinder technique using Vinciguerra’s nomogram for astigmatism greater than 3 D (see Vinciguerra et al., 1999).
- Dividing the astigmatism in two treatments.
- Perform half of the cylinder in minus cylinder and the other half in plus cylinder (e.g., the treatment of –4.00 – 6.00 X 180 degrees would be piano +3.00 × 90 degrees, piano −3.00 × 180 degrees, and −7.00 D total spherical ablation).
- Dividing the astigmatism in two treatments.
- removal of 0.25 D of astigmatism to the sphere per diopter of cylinder (see Myopia)
Mixed Astigmatism
Methods
- Use Chayet’s nomogram (Table 9-1).
- Use cylindrical correction with both myopic and hyperopic cylinder modes of the laser to split the amount of ablation made to the steep and flat meridians of an astigmatic cornea.
- Perform only a portion of the steep meridian correction and a portion of the flat meridian correction [elliptical ablation in the steep meridian (myopic astigmatism) results in a hyperopic shift].
- Assume that ablation in the steep meridian always induces, a 33% hyperopic shift, never to exceed 30% of the cylindrical component.
- For example, +2.00 − 4.00 × 0 degrees = 0.37% of –4 D is piano –1.5 × 0 degrees, which is corrected on the steep meridian ablation.
- After inducing 33% of hyperopic shift, the refraction is +2.50 − 2.50 X 0 degrees, which can be translated to piano +2.50 x 90 degrees that can be corrected on the flat meridian ablation.
- Do not confuse which axis is to be targeted for which ablation.
- Begin with the minus cylinder.
- Assume that ablation in the steep meridian always induces, a 33% hyperopic shift, never to exceed 30% of the cylindrical component.
| Meridians | ||
|---|---|---|
| Flat | Steep | |
| Simple myopic astigmatism | ||
-1.0 D spherical component | 7%-16% | 84%-93% |
0.0 D spherical component | 25% | 75% |
| Mixed astigmatism | ||
0.0 D spherical equivalent | 63% | 37% |
+1.0 D spherical equivalent | 80% | 20% |
| Simple hyperopic astigmatism | 100% | 0% |
Presbyopia
Patient Preparation
- Try monovision with contact lenses before proceeding with surgery (if the patient feels comfortable with this vision, proceed with the procedure).
Methods
- Although monovision is the only alternative to PRK for improvement of near vision, it is not considered real correction of accommodation.
- Correct one eye, usually the dominant one, toward emmetropia.
- Undercorrect the other eye with a slight level of myopia (−0.50 to −1.50 D).
- Correct one eye, usually the dominant one, toward emmetropia.
Alternative Treatments
LASER IN SITU KERATOMILEUSIS The upper limit for PRK may be between –7 and –12 D. For severe myopia (> –12 D), predictability decreases but the incidence of certain adverse effects (e.g., corneal haze) significantly increases to greater than 10% from less than 1% for low myopia corrections. Inform patients with myopia greater than 10 D and hyperopia greater than 3 D of the greater risks associated with the procedure (60% of patients experience excellent results).
- retreatments
- halos
- haze
- loss of BCVA
PHAKIC INTRAOCULAR LENS INSERTION Implant a phakic anterior chamber intraocular lens (IOL) in patients with myopia greater than 10 D or hyperopia greater than 3 D if the anterior chamber depth and axial length allow such an invasive procedure (see Chapters 14 and 16).
CLEAR LENSECTOMY Perform clear lensectomy with a posterior chamber IOL for patients with presbyopia. If the exact intended correction is not achieved after insertion of an IOL or a phakic IOL, retreatment can be performed with PRK or LASIK (see Retreatment).
HOLIUM LASER KERATOPLASTY This can be used to treat low hyperopia (<2.5 D).
POSTOPERATIVE CONSIDERATIONS
Medications
- corticosteroids [reverse inflammatory reaction but the mechanism of corticosteroids on keratocytes is still unclear and may cause increased intraocular pressure (IOP) or cataracts]
- antibiotics
- Prescribe a consistent regimen to reduce the risk of infection (start medication a few hours after surgery).
- Instill 0.3% ofloxacin every 4 hr for the first 72 hr and 3 times a day for the following week (the kill curve is ~ 3 hr, at which time 99.9% of bactericidal concentration is attained for Gram-positive bacteria, including Staphylococcus aureus, S. epidermidisy and S. pneumoniae and Gram-negative bacteria such as Haemophilus influenzae).
- Prescribe a consistent regimen to reduce the risk of infection (start medication a few hours after surgery).
- artificial tears
- Prescribe 4 times a day for 1 to 3 months after surgery (mandatory and greatly helps the healing process and patient comfort)
- oral analgesics (for pain management during the first 72 hr of re-epithelialization)
- Give nonsteroidal anti-inflammatory drugs (NSAIDs; 0.5% ketorolac tromethamine) every 4 hr for the first day and 3 times a day for the next 48 hr.
Complications
- loss of BCVA
- often the result of haze
- after prompt retreatment, lines of visual acuity regained
- often the result of haze
- undercorrection or overcorrection without haze
- Plan prompt retreatment as early as 1 month after surgery.
- Stop corticosteroids 2 to 3 days before retreatment.
- Plan prompt retreatment as early as 1 month after surgery.
- epithelial or stromal haze
- graded on a scale of 0 to 5 (Table 9-2)
- avoid by using an epithelial brush, chilled BSS after surgery, planned corticotherapy, and sunglasses to protect the eyes from UV light
- risk decreased by using the multizone/multipass technique (Fig. 9-3)
- generally increases near the second month after surgery and then decreases
- decreases with use of scanning excimer lasers
- graded on a scale of 0 to 5 (Table 9-2)
- halos, glare, and starburst (Fig. 9-4)
- caused by the pupil dilating to a greater diameter than the treatment optical zone (OZ)
- incidence greatly decreased by OZs greater than 4 mm and transition zones
- Stiles-Crawford effect (a spherical aberration of retinal directional sensitivity caused by the effect of light passing through the center of the pupil; insignificant for pupil size >4 mm and peaks for 8-mm pupil size)
- may be decreased someday by wavefront technology (accounts for the optical dry eye; see Chapter 12)
- usually resolves within 6 to 12 months of surgery
- may be decreased someday by wavefront technology (accounts for the optical dry eye; see Chapter 12)
- caused by the pupil dilating to a greater diameter than the treatment optical zone (OZ)
- dry eye
- Perform a Schirmer’s test and verify tear break-up time or ocular surface with dye staining pattern.
- Prescribe artificial tears without preservatives [newly developed artificial tears, such as TheraTears (Advanced Vision Research Inc., Woburn, MA) have balanced electrolytes that are more suitable for the eye].
- Use puncrum plugs for extreme cases.
- Perform a Schirmer’s test and verify tear break-up time or ocular surface with dye staining pattern.
- severe bilateral complication (incidence theoretically estimated at 0.01%)
- secondary effects from medications (check for increased IOP with corticosteroid use)
- sterile infiltrates (if present within the first few postoperative days, discontinue NSAIDs, remove the contact lens immediately, and patch the eye if re-epithelialization is not complete)
| Grade | Description |
|---|---|
| 0 | Clear cornea, with a possibility of a very light haze. There is no difference in texture between the central treated zone and the peripheral nontreated zone. The normal corneal stroma has a ground glass appearance. |
| 0.25 | Trace haze is defined as faint corneal haze just perceptible by broad oblique illumination. It is the minimal amount of corneal haze present in the grading scale and is characterized by a diffuse “cotton floss” appearance of the corneal stroma. |
| 0.5 | The haze is faint when seen by broad oblique illumination and is comparable to a light cottony cloud in the stroma. Although more perceptible than 0.25 trace haze, neither 0.25 nor 0.5 haze can be identified with broad direct illumination. |
| 1.0 | The haze is difficult to see by direct focal slit illumination. It is easier to see than the 0.5 faint haze by broad oblique illumination. The cottony cloud is more intense. |
| 2.0 | Haze affecting lightly the refraction: it is hardly seen by direct focal slit illumination. By broad oblique illumination, it has a more granular aspect than 1.0 haze. |
| 3.0 | Moderate haze: refraction is possible but with difficulty. It is considered as a moderately dense opacity that partially obscures the iris in direct illumination. Frequently, the haze is scattered in a series of small dots. |
| 4.0 | The opacity affects completely the refraction: the anterior chamber can be seen. The haze obscures any details of the iris. It is the first grade able to be seen without using any instrument. In direct slit illumination, it is a white-gray corneal opacity that may display thickness and elevation. Extremely rare. |
| 5.0 | The opacity makes the examination of the anterior chamber difficult or impossible. Almost never seen. |
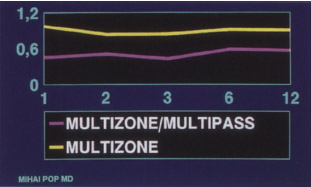

Figure 9–4 An artist’s depiction of halos.
RETREATMENT
Identification and classification of patients who require retreatment are subtle because some patients with mild corrections or haze are symptomatic whereas others are completely satisfied with their outcome.
Indications
- corneal haze
- Early diagnosis is mandatory.
- Retreatments are possible as early as 1 month after surgery before haze formation increases.
- Avoid severe haze by retreating promptly (advanced haze formation can activate mechanisms that create further haze even after retreatment).
- Retreatment of eyes with an aggressive healing pattern that is accompanied by a progressive myopic shift may create the same amount of haze when performed 6 to 12 months after the original treatment.
- Early diagnosis is mandatory.
- undercorrections, overcorrections, and regression
- These are correctable within 1 to 4 months after surgery.
- Stable refractive outcomes may result from prompt retreatment.
- Use holmium laser keratoplasty for overcorrections less than +1.50 D.
- These are correctable within 1 to 4 months after surgery.
Results
- up to 95% of eyes within ±1 D
- possibility of lost lines of BCVA after retreatment
Phototherapeuthic Keratectomy
Methods
- Perform phototherapeutic keratectomy (PTK) under low light intensity of the laser microscope to see the epithelial pseudofluorescence.
- Remove the epithelium by setting the laser to a depth of 55 μm for the optical and transition zone diameters.
- Stop the laser as soon as you see a black ring disturbing the pseudofluorescence of the epithelium.
- Remove any residual epithelial cell layer manually.
- very easy for minimal or no haze
- not recommended for moderate to severe haze (epithelial debris and cells remain between the island of scarred tissue that makes an irregular surface before retreatment; may result in unpredictable outcomes and increased chances of secondary scarring; use transepithelial PTK)
- very easy for minimal or no haze
- Proceed with usual algorithms for treatment of the refractive error as previously described.
- Retreat refraction of patients with haze (very rare in patients with <6 D of myopia).
- For advanced haze, treat only half the refraction because overcorrection may occur (initial manifest refraction may be imprecise; Fig. 9-5).
PHOTOTHERAPEUTIC KERATECTOMY AFTER RADIAL KERATECTOMY
Indications
- management of the small ridges usually found over the radial keratectomy (RK) incisions (found under the epithelium as the result of the stroma reaction following RK; retreatment is vital)
Methods
- Perform PTK at 6.0 mm diameter with a stromal depth of 70 μm to eliminate the small RK ridges.
- Stop PTK before reaching 70 μm (PTK may provide uniformity and homogeneity on the stromal surface).
- Maintain PTK until the loss of pseudofluorescence over the ridges is about 1.5 to 2.0 mm wide.
- When the nonfluorescent area on the incisions appears and is ± 1 mm wide, stop and treat the residual myopia (proceeding creates depressions along the RK incisions).
- After removing RK incision ridges, manually clean the remaining epithelium and perform PRK treatment as usual using the manifest refraction evaluated before surgery.
- Keep OZ at 5.0 to 5.5 mm (an OZ of 6.0 mm is not necessary because RK has already provided a relative transition zone for PRK).
- If inexperienced, stop the treatment every 10 sec and ask the patient to fixate on the aiming diode, recenter the pupil, and resume the procedure.
- Control patient eye movement by positioning your hands to recenter the treatment (one hand on the patient’s forehead and the other hand on the focusing mechanism of the laser or use an eye-tracker device to aim the laser).
- Indicate to the patient how many seconds of the treatment remain every 5 to 10 sec.
- Reassure the patient that everything is going well to ensure patient cooperation.
- Cool the cornea with chilled (4°C) BSS.
- After treatment, instill antibiotic drops to prevent infection and wash out debris.
- Do not wipe the cornea with a microsurgical sponge (small particles may catch under the contact lens and cause patient discomfort).
- Manipulate the outer surface of the contact lens carefully with a soaked microsurgical sponge to avoid contact with the inner surface and the introduction of foreign body material or debris.
- Do not use forceps because they may damage the surface of the contact lens.
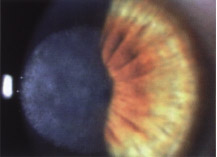
Figure 9-5 Advanced (grade 3) haze following PRK. BCVA decreased to 20/200.
Postoperative Care
- Examine the patient’s eyes 72 hr after surgery or daily until reepithelialization and at 1, 2, 3, 6, 12, and 24 months (for lower myopes, omit 2- and 6-month follow-ups if the patient’s condition does not change).
- Perform manifest refraction.
- Evaluate patient’s BCVA and uncorrected visual acuity using a Snellen’s chart.
- Use corneal topography to check for increased astigmatism or irregular astigmatism.
- Grade haze on a scale of 0 to 3 (clear to completely obscured), as proposed by some authors.
- Use corneal topography to check for increased astigmatism or irregular astigmatism.
- After correcting for astigmatism with PRK, perform a vector analysis on preoperative refraction rather than postoperative refraction.
- To estimate the quality of astigmatic correction, calculate Alpin’s index of success (defined as the proportion of the remaining astigmatism to treat on the corneal plane divided by the intended correction) using medical computer software.
- Analyze refractive outcomes and, if needed, adjust excimer laser nomograms (computer software packages, such as the ASSORT Eye Surgery Analysis Program from the Melbourne Excimer Group in Melbourne, Australia, may help in this task).
Suggested Readings
Griffith M, Jackson BW, Lafontaine MD, Mintsioulis G, Agapitos P, Hodge W. Evaluation of current techniques of corneal epithelial removal in hyperopic photorefractive keratectomy. J Cataract Refract Surg. 1998;24:1070-1078.
Jackson BW, Casson E, Hodge W, Mintsioulis G, Agapitos PJ. Laser vision correction for low hyperopia. Ophthalmology. 1998;105:1727-1738.
Kim JH, Hahn TW, Young CL. Photorefractive keratectomy in 202 myopic eyes: one year results. Refract Corneal Surg. 1993;9(suppl): S11-S16.
Kitazawa Y, Tokoro T, Ito S, Ishii Y. The efficacy of cooling on excimer laser photorefractive keratectomy in the rabbit eye. Survey Ophthalmol. 1997;42(Suppl 1):S82-S88.
O’Brart DP, Lohmann CP, Fitze FW, Smith SE, Kerr-Muir MG, Marshall J. Night vision after excimer laser photorefractive keratectomy: haze and halos. Eur J Ophthalmol. 1994;4:43-51.
Pop M. Prompt retreatment after photorefractive keratectomy. J Cataract Refract Surg. 1998; 24:320-326.
Pop M, Aras M. Multizone/multipass photorefractive keratectomy: six months results. J Cataract Refract Surg. 1995;21:633-643.
Pop M, Payette Y. Multipass versus single pass photorefractive keratectomy for high myopia using a scanning laser. J Refract Surg. 1999;15: 444-450.
Pop M, Payette Y. Results of bilateral photorefractive keratectomy. Ophthalmology. 2000; 107: 472-479.
Vinciguerra P, Epstein D, Azzolini M, Radice P, Sborgia M. Algorithm to correct hyperopic astigmatism with the Nidek EC-5000 excimer laser. J Refract Surg. 1999;15(suppl):Sl86-S187.
< div class='tao-gold-member'>



