INDICATIONS
- Intravitreal injections are an increasingly common therapeutic tool for addressing various retinal pathologies. Examples include:
- Antivascular endothelial growth factor (VEGF) agents for neovascular (“wet”) age-related macular degeneration (AMD) or proliferative diabetic retinopathy (PDR)
- Corticosteroids for cystoid macular edema (CME) due to diabetes, central retinal vein occlusion (CRVO), or branch retinal vein occlusion (BRVO)
- Antimicrobials for bacterial or fungal endophthalmitis
- Antivirals for retinal necrosis in immunocompromised patients
- Antivascular endothelial growth factor (VEGF) agents for neovascular (“wet”) age-related macular degeneration (AMD) or proliferative diabetic retinopathy (PDR)
CONTRAINDICATIONS
- Active blepharitis: This increases the risk of inoculating the vitreous with bacteria and should be treated with lid scrubs and topical antibiotics prior to injection.
ALTERNATIVES
Medical Therapy
- Macular degeneration: Nutritional supplements (antioxidants plus zinc) and smoking cessation have been shown to slow the progression from nonexudative (“dry”) macular degeneration to the “wet” neovascular form.1
Lasers/Surgery
- Macular degeneration: Prior to the development of anti-VEGF agents, various laser treatments were commonly used for AMD. These include:
- Laser photocoagulation: Laser spots are applied to areas of choroidal neovascularization (CNV). This technique is only effective with well-demarcated CNV. It also has high rates of recurrence, and results in laser-induced scotoma when burns are placed near the macula.
- Photodynamic therapy (PDT): A photosensitizing agent (verteporfin) is administered intravenously. The retina is then exposed to a low-energy 689 nm laser for 83 seconds, which activates the agent and results in neovascular closure. Patients must be cautioned to avoid direct sunlight for several days following each treatment because of the risk of photosensitivity reactions.
- Laser photocoagulation: Laser spots are applied to areas of choroidal neovascularization (CNV). This technique is only effective with well-demarcated CNV. It also has high rates of recurrence, and results in laser-induced scotoma when burns are placed near the macula.
- Bacterial endophthalmitis: Pars plana vitrectomy permits physical removal of intraocular organisms and inflammatory mediators, as well as direct administration of antibiotics. Sufficient vitreous specimens are easily obtained. In post-cataract extraction patients with visual acuity that is better than light perception upon presentation, however, needle biopsy with intravitreal antibiotic injection produces equivalent outcomes.2
RELEVANT PATHOPHYSIOLOGY
Age-Related Macular Degeneration (AMD)
- AMD is the leading cause of irreversible vision loss in the elderly.
- Progressive deterioration of the macula results in central vision loss.
- It occurs in two forms:
- Nonexudative (“dry”): Subacute degeneration of the macula characterized by retinal pigment epithelium atrophy and the accumulation of subretinal deposits, or drusen. This condition may progress to “wet” AMD.
- Neovascular (“wet”): Development of choroidal neovascularization (CNV) leads to fluid extravasation and serous detachment of the retinal pigment epithelium due to breaks in Bruch’s membrane. This produces severely blurred central vision and metamorphopsia.
- Nonexudative (“dry”): Subacute degeneration of the macula characterized by retinal pigment epithelium atrophy and the accumulation of subretinal deposits, or drusen. This condition may progress to “wet” AMD.
Anti-VEGF Agents
- VEGF-A: Vascular endothelial growth factor-A stimulates choroidal/retinal neovascularization and increases vessel permeability.
- Three anti-VEGF agents have been approved by the Food and Drug Administration (FDA).
- Bevacizumab (Avastin): Bevacizumab was FDA approved in 2004 for intravenous treatment of metastatic colon cancer, and used off-label for intravitreal injections. It contains a full-length monoclonal antibody that inhibits all active VEGF-A isoforms.
- Ranibizumab (Lucentis): Ranibizumab was FDA approved in 2006 for exudative AMD. It contains a monoclonal antibody–binding fragment that inhibits all active VEGF-A isoforms. Lucentis costs significantly more per dose than Avastin and trials comparing efficacy are currently underway.
- Pegaptanib sodium (Macugen): Pegaptanib sodium was FDA approved in 2004 for classic exudative AMD. It contains an RNA oligonucleotide (aptamer) linked to polyethylene glycol that specifically binds to and inhibits VEGF 165. It has been superseded by the development of Lucentis and Avastin.
- Bevacizumab (Avastin): Bevacizumab was FDA approved in 2004 for intravenous treatment of metastatic colon cancer, and used off-label for intravitreal injections. It contains a full-length monoclonal antibody that inhibits all active VEGF-A isoforms.
- Monthly injections are required in many patients, but it is possible to vary the frequency of administration based on macular ocular coherence tomography (OCT) findings.
Cystoid Macular Edema (CME)
- Cytokine release from activated inflammatory cells is thought to trigger breakdown of the blood-retinal barrier, leading to accumulation of fluid in the outer plexiform layer of the macula.
- This distorts central vision and produces cystic spaces visible on OCT.
- It occurs in a various disorders, inducing diabetes, central retinal vein occlusion (CRVO), and branch retinal vein occlusion (BRVO).
- It is also a potential complication of cataract extraction and other intraocular surgery.
- Initial treatment is with topical steroids or nonsteroidal anti-inflammatory drugs (NSAIDS). If this fails, sub-Tenon’s or intravitreal steroid injections (e.g., triamcinolone) may be given.
Infectious Endophthalmitis
- Infection of intraocular compartments and adjacent layers can result from direct inoculation (e.g., open globe injury, intraocular surgery) or hematogenous spread.
- Potential signs and symptoms include ocular pain, decreased visual acuity, conjunctival injection, chemosis, hypopyon, and the presence of anterior chamber “cell and flare” on slit lamp examination.
- If endophthalmitis is suspected, specimens of the aqueous and/or vitreous should be obtained for culture and sensitivity using needle biopsy or vitrectomy.
- Most antibiotics have limited ability to penetrate the vitreous if administered topically, systemically, or as a sub-Tenon’s injection. Intravitreal injection is therefore the best strategy for achieving therapeutic drug levels. Direct contact with intraocular tissues, however, increases the risk of retinal toxicity.
- Bacterial endophthalmitis: Onset is usually acute. Empiric treatment must be instituted pending culture results. Vancomycin is generally used for gram-positive coverage, whereas amikacin (an aminoglycoside) or ceftazidime (a third generation cephalosporin) is added for gram-negative coverage. Antibiotic coverage is narrowed and focused once an etiologic agent has been identified.
- Fungal endophthalmitis: Progression is usually more indolent. Amphotericin B was the traditional therapy of choice, but azole antifungals are less toxic to the retina and have gained popularity.
- The addition of intravitreal corticosteroids helps to reduce inflammation, but does not appear to influence long-term visual outcomes.
PREOPERATIVE ASSESSMENT
- Slit lamp exam: Check the anterior chamber for “cell and flare” as well as for hypopyon, which layers in the inferior angle.
- Dilated fundus exam: Assess for posterior segment pathology, including neovascularization, CRVO, BRVO, macular drusen, or vitreous stranding and cells.
- Macular OCT: Examine images for evidence of drusen or cystoid edema.
- Fluorescein/indocyanine green angiography: Assess for dye diffusion patterns suggestive of retinal or choroidal neovascularization. Normal retinal vessels retain fluorescein dye, but it leaks from neovascular beds, which have increased permeability. Fluorescein diffusely enhances the choroidal circulation because its fenestrated capillaries are permeable to the dye. Indocyanine green, however, is normally retained and leakage helps to pinpoint areas of occult choroidal neovascularization.
ANESTHESIA
- Topical anesthetic drops are applied to the inferior fornix.
- The injection site can be further anesthetized with subconjunctival infiltration (e.g., lidocaine) or with an application of topical anesthetic jelly.
PROCEDURE
- Preparation: Topical antibiotic drops may be administered for several days preceding the injection. The lids, lashes, and ocular surface are carefully painted with Betadine prior to lid speculum insertion.
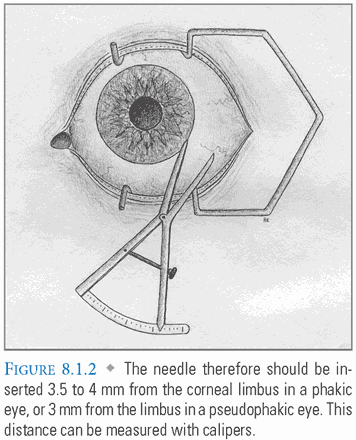
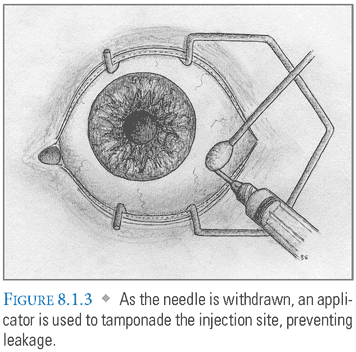
- Injection: A 0.5 inch 27- to 30-gauge needle is passed through the pars plana to avoid damage to the highly vascular ciliary body anteriorly and the sensory retina posteriorly. It should be inserted 3.5 to 4 mm from the limbus in a phakic eye, or 3 mm from the limbus in a pseudophakic or aphakic eye (FIG. 8.1.2). The inferotemporal quadrant is selected most frequently due to ease of access. The globe can be stabilized with a cotton-tipped applicator during injection. The needle, directed toward the center of the globe, is steadily advanced over 1 second to a depth of half the needle length. As the needle is withdrawn, this applicator is used to tamponade the injection site, preventing leakage (FIG. 8.1.3).
COMPLICATIONS
- Sudden decreased visual acuity: This most commonly occurs as a result of a transient increase in intraocular pressure (IOP) with rapid and spontaneous resolution. If symptoms do not resolve in 1 minute, however, an anterior chamber paracentesis should be performed.
- Floaters: These are due to injected medication or air bubbles in the visual axis and should resolve rapidly.
- Secondary cataract or glaucoma: These are frequent complications of intravitreal steroids. Careful monitoring is warranted while receiving such injections.
- Endophthalmitis: Infection is very rare with proper adherence to sterile technique.
- Vitreous hemorrhage: This is also a rare complication with appropriate needle insertion.
POSTOPERATIVE CARE
- After injection, view the fundus with an indirect ophthalmoscope to rule out complications.
- It is not necessary to check the IOP with applanation tonometry if the optic nerve appears well-perfused following injection.
- Instruct the patient to apply topical antibiotic drops for several days after the injection.
- Counsel the patient to report severe ocular discomfort or reduction in visual acuity, but reassure him or her that transient blurring or dark patches in the visual fields (bubbles) are to be anticipated.
Further Reading
Text
Andreoli CM, Miller JW. Anti-vascular endothelial growth factor therapy for ocular neovascular disease. Curr Opin Ophthalmol. 2007;18(6):502–508.
Fu A, Bui A, Rae R, et al. Cystoid muscular edema. In: Yanoff M, Duker J, eds. Ophthalmology. 3rd ed. Philadelphia, PA: Mosby; 2009:696–701.
Lang GK. Vitreous body. In: Ophthalmology: A Pocket Textbook Atlas. 2nd ed. New York, NY: Georg Thieme Verlag; 2007:285–304.
Read RW. Endophthalmitis. In: Yanoff M, Duker J, eds. Ophthalmology. 3rd ed. Philadelphia, PA: Mosby; 2009: 815–819.
Rosenfeld PJ, Martidis A, Tennant MTS. Age-related mucular degeneration. In: Yanoff M, Duker J, eds. Ophthalmology. 3rd ed. Philadelphia, PA: Mosby; 2009:558–673.
Primary Sources
Age-Related Eye Disease Study Research Group. A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, beta carotene, and zinc for age-related macular degeneration and vision loss: AREDS report no. 8. Arch Ophthalmol. 2001; 119(10):1417–1436.
Arevalo JF, Fromow-Guerra J, Sanchez, et al.; Pan-American Collaborative Retina Study Group. Primary intravitreal bevacizumab for subfoveal choroidal neovascularization in age-related macular degeneration: results of the Pan-American Collaborative Retina Study Group at 12 months follow-up. Retina. 2008;28(10):1387–1394.
Brown DM, Kaiser PK, Michels M, et al.; ANCHOR Study Group. Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med. 2006;355(14):1432–1444.
Endophthalmitis Vitrectomy Study Group. Results of the Endophthalmitis Vitrectomy Study. A randomized trial of immediate vitrectomy and of intravenous antibiotics for the treatment of postoperative bacterial endophthalmitis. Arch Ophthalmol. 1995;13(12): 1479–1496.
Ferrara N, Damico L, Shams N, et al. Development of ranibizumab, an anti-vascular endothelial growth factor antigen binding fragment, as therapy for neovascular age-related macular degeneration. Retina. 2006;26(8): 859–870.
Rosenfeld PJ, Brown DM, Heier JS, et al.; MARINA Study Group. Ranibizumab for neovascular age-related macular degeneration. N Engl J Med. 2006;355(14):1419–1431.
VEGF Inhibition Study in Ocular Neovascularization (V.I.S.I.O.N.) Clinical Trial Group; Chakravarthy U, Adamis AP, Cunningham ET Jr, Goldbaum M, Guyer DR, Katz B, Patel M. Year 2 efficacy results of 2 randomized controlled clinical trials of pegaptanib for neovascular age-related macular degeneration. Ophthalmology. 2006;113(9):1508.e1–25.
8.2PARS PLANA VITRECTOMY (PPV)
Small instruments, inserted through the pars plana, are used to cut and remove vitreous, peel membranes, and laser photocoagulate providing treatment to various posterior segment pathologies.
INDICATIONS
Common indications include (but are not limited to):
- Proliferative diabetic retinopathy (PDR)
- Proliferative vitreoretinopathy (PVR)
- Retinal detachment with or without breaks
- Macular hole
- Macular pucker (epiretinal membrane)
- Persistent or recurring vitreous hemorrhage
- Vitreoretinal traction
- Complications of anterior segment surgery (e.g., dislocated lens, choroidal hemorrhage)
- Infection (e.g., endophthalmitis, viral retinitis) and inflammatory conditions (e.g., sarcoidosis, Behçet’s syndrome)
- Persistent macular edema
- Subretinal hemorrhage
- Trauma and intraocular foreign bodies (IOFB)
- Retinopathy of prematurity
- Choroidal neovascularization (CNV)
- Tumors (e.g., lymphoma, diagnostic vitrectomy)
RELEVANT PHYSIOLOGY/ANATOMY
Vitreous
- 98% water; hyaluronic acid and collagen fibrils account for gel-like consistency.
- The outer cortex adheres to the internal-limiting membrane (ILM) of the retina.
- The outer membrane (the hyaloid membrane) adheres firmly to the ora serrata (vitreous base), posterior lens capsule, pars plana epithelium, optic nerve head, blood vessels, and perifoveal region.
- With aging, the vitreous body contracts and can pull away from the ILM leading to a posterior vitreous detachment (PVD).
Posterior Hyaloid Face
After the vitreous cortex detaches from the ILM, this thin layer remains adherent to the ILM in the macular region, providing a persistent source of retinal traction.
Retina
Nine layers (from inside to outside)
- Internal limiting membrane: glial cell fibers separate from vitreous
- Nerve fiber layer: from third-order neurons, converge on optic disc
- Ganglion cell layer: third-order neurons
- Inner plexiform layer: synapses of second and third-order neurons
- Inner nuclear layer: second-order neurons, bipolar cells (horizontal and amacrine cells)
- Outer plexiform layer: synapses of first and second-order neurons
- Outer nuclear layer: first-order neurons, nuclei of rods and cones
- Outer limiting membrane: separates rod and cone nuclei from photoreceptive elements
- Rod and cone photoreceptors
- Retinal pigment epithelium (RPE)
- Subretinal space: lies between photoreceptors and RPE
- Bruch’s membrane: choroid basement membrane (Note: CNV is thought to arise via breaks in Bruch’s membrane.)
Pars Plana (FIG. 8.2.1)
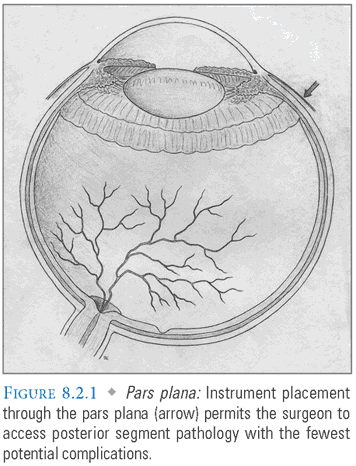
- Entrance site (sclerostomy) is approximately 3.5 mm (pseudophakic eyes) to 4 mm (phakic eyes) posterior to the limbus.
- Sclerostomy allows entrance into the posterior segment without damaging the retina (anterior to the ora serrata) or causing hemorrhage (posterior to the highly vascularized pars plicata).
RELEVANT PATHOPHYSIOLOGY
Retinal Detachment: Three types
- Rhegmatogenous: Full-thickness break (e.g., hole or tear) in the sensory retina with extravasation of vitreous fluid into the subretinal space; usually preceded by a PVD.
- Tractional: Fibrovascular tissue (e.g., vitreal, epiretinal, or subretinal membranes) leads to tractional forces that separate the sensory retina from the RPE.
- Serous/Hemorrhagic: Fluid accumulates in the subretinal space without traction or retinal breakage; seen with degenerative, infectious, and inflammatory diseases.
Vitreous Hemorrhage
- Causes include PDR, branch or central retinal vein occlusion, retinal tears or detachment, or trauma.
- Mechanism generally involves vitreoretinal traction leading to rupture of small vessels.
Epiretinal Membranes
- Membranes are composed of glial cells, fibroblasts, RPE cells, and inflammatory cells; they become adherent to the retina.
- PVR: Scar tissue formation is commonly seen after retinal detachment. RPE and glial cells enter the vitreous cavity after a retinal tear, and despite adequate repair, these cells can proliferate on the retinal surface leading to retinal traction upon membrane contraction.
- When tractional forces from epiretinal membranes cause wrinkling of the retinal surface overlying the macula, this is referred to as macular pucker.
Macular Hole
- A macular hole involves a partial or full-thickness break in the sensory region overlying the macula. Although the exact etiology remains unknown, it is believed that vitreoretinal traction over the macula is responsible for creating these holes.
- Staging (determines treatment outcomes):
- Stage 1: Loss of normal foveal depression; presence of yellow ring or pigmentary spot
- Stage 2: Full-thickness hole with diameter <400 microns
- Stage 3: Full-thickness hole with diameter >400 microns
- Stage 4: Full-thickness hole with diameter >400 microns plus posterior hyaloid detachment
- Stage 1: Loss of normal foveal depression; presence of yellow ring or pigmentary spot
- Watzke-Allen Test: Classic clinical test used to differentiate macular holes from pseudoholes/ epiretinal membranes. A narrow beam of light is placed over the macula. If the patient describes a break in the line, this is a positive test for a macular hole. This test is not always reliable, and optical coherence tomography (OCT) is often used to confirm the diagnosis.
ALTERNATIVE TREATMENTS FOR COMMON PATHOLOGIES
Diabetic Vitreous Hemorrhage
- Observation followed by laser therapy: A large percentage of diabetic bleeds clear spontaneously.
- Laser photocoagulation: This is done when any part of the retina is visible after initial hemorrhage. If hemorrhage is excessive, this is not a viable option.
- Intravitreal bevacizumab (Avastin) or ranibizumab (Lucentis)
- Anterior retinal cryotherapy (ARC): This is done if portions of the retina are visible; it is infrequently performed compared to laser photocoagulation.
The Diabetic Retinopathy Vitrectomy Study (DRVS) suggests early vitrectomy for vitreous hemorrhage; although recently, many retinal specialists are trying bevacizumab first.
Retinal Break/Detachment
- Laser/cryotherapy: Creates adhesions between the retinal pigment epithelium (RPE) and sensory retina.
- Pneumatic retinopexy: Air or gas is injected into the vitreous to keep the retina in place in conjunction with laser treatment or cryotherapy to create anchoring adhesions.
- Scleral buckle: External compression of the globe over the site of a retinal break reapposes the retina with the RPE; it is often coupled with laser or cryotherapy.
Significant Vitreoretinal Traction or Preretinal Membranes
- Close observation followed by surgical intervention (e.g., PPV and/or membrane removal)
Macular Hole
Observation: About half of stage 1 holes resolve spontaneously along with a smaller percentage of stage 2 holes.
PREOPERATIVE SCREENING
Screening involves complete history and physical (H&P), which is very important in diabetic/hypertensive patients; VA measurement and pupillary function; visual fields; and Amsler grid test for metamorphopsia (indicates macular involvement).
- Anterior segment assessment: Observe carefully for iris neovascularization as this may require panretinal photocoagulation (PRP) prior to vitrectomy.
- Lens assessment (phakia vs. pseudophakia vs. aphakia): In pseudophakic eyes, the type of intraocular lens (IOL) may influence the operative strategy (e.g., intravitreal silicone oil may adhere to a hydrophobic lens).
- Indirect biomicroscopy: Used to assess relationship and characteristics of the vitreous and retina; it is important to determine if PVD is present. If so, the extent of the posterior hyaloid surface is noted. The location of retinal breaks and the severity of membrane proliferation should also be noted.
- Ultrasound: Essential for eyes with opaque vitreous (e.g., hemorrhage). Can identify retinal detachments, membranes, intraocular foreign bodies (IOFBs), choroidal detachments, subretinal fluid, and tumor location.
- Electroretinography (ERG): ERG can be used to evaluate the functionality of the retina.
- Imaging: For trauma cases, CT scans are very helpful for localizing IOFBs and assessing bony damage. A magnetic resonance imaging (MRI) is contraindicated as metallic IOFBs may shift during scanning.
ANESTHESIA
Local anesthesia (e.g., retrobulbar, peribulbar, or sub-Tenon’s block) is most commonly administered. Infrequently, topical anesthesia is sufficient.
General anesthesia is only considered in patients who cannot tolerate local anesthesia or who present with recent open globe.
Nitrous oxide is contraindicated in patients who are receiving intraocular gas.
PROCEDURE
The following narrative describes the introductory steps in a PPV with the intention to clear opacified media and relieve vitreoretinal traction. Various case-dependent surgical techniques are discussed under “Further Surgical Considerations.”
 SEE TABLES 8.2.1 Rapid Review and 8.2.2 Surgical Pearls, and WEB TABLES 8.2.1 to 8.2.4 for review of suggested instrumentation and supplies.
SEE TABLES 8.2.1 Rapid Review and 8.2.2 Surgical Pearls, and WEB TABLES 8.2.1 to 8.2.4 for review of suggested instrumentation and supplies.


General Vitrectomy Techniques:
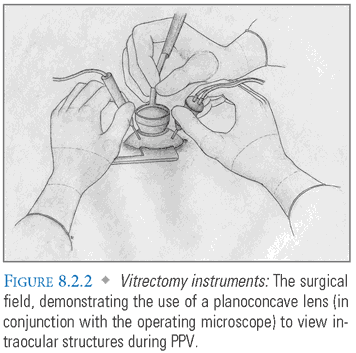
- Microscope and Lens Setup: After the operating microscope is adjusted, the surgeon must choose an appropriate lens to visualize the posterior segment. If an assistant is present, a handheld lens can be used (FIG. 8.2.2). If no assistant is present, a lens can be sutured to the sclera (e.g., Landers lens) or a noncontact system (e.g., BIOM, Oculus, Washington) can be used. For evaluation of the peripheral retina, scleral depression is combined with the use of a prismatic or wide-field viewing lens with an inverter on the microscope. A binocular indirect ophthalmoscopy may also be used.
- Vitrectomy Instruments: There are two basic types of vitrectors used to cut and remove vitreous. A guillotine cutter is a blunt instrument in which a small piece of vitreous is aspirated through a side port and cut by an inner blade moving up and down. A rotary cutter is a blunt instrument in which the inner blade rotates inside the outer probe. The aspiration port is closer to the tip allowing for closer cutting to the retina. Of note, the higher the cutting speed, the less traction on the vitreous and retina (i.e., decreased risk of iatrogenic retinal tear).
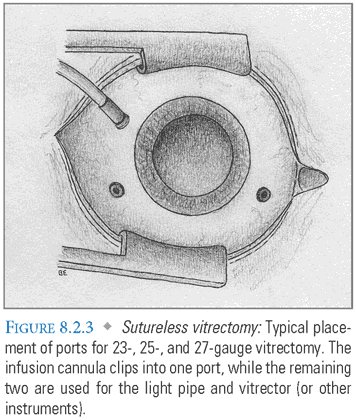
The surgeon may also decide between a sutureless 23-, 25-, or 27-gauge vitrector versus a 20-gauge vitrector. Use of the 20-gauge vitrectomy instrument necessitates opening the conjunctiva and subsequent suturing to the sclera. The 23-gauge to 27-gauge vitrector ports are placed transconjunctivally and do not require suturing to the sclera.
- Description of sutureless (23-gauge to 27-gauge) vitrector port: The insertion system consists of a sharp blade (trocar) surrounded by a sleeve through which the vitrector and other instruments will be inserted. Trocars are most commonly placed in the inferotemporal, superonasal, and supertemporal regions, through which the infusion cannula, fiberoptic light, and vitrector will be placed (FIG. 8.2.3).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


