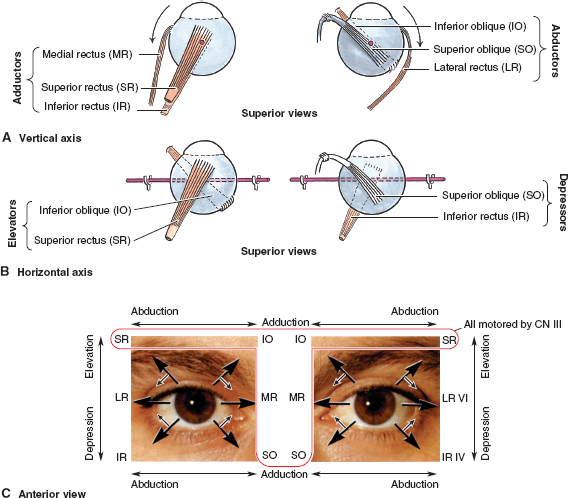
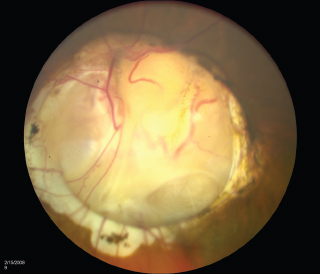
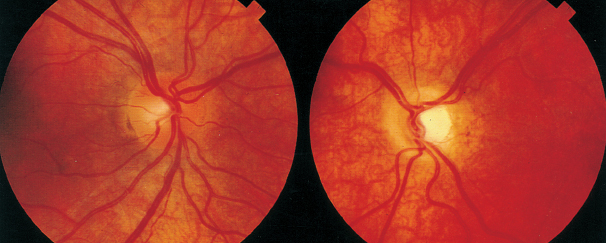
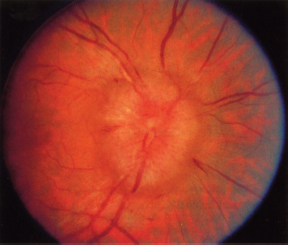
NEURO-OPHTHALMOLOGIC HISTORY AND KEY TESTS Three major categories of complaint in neuro-ophthalmology: Afferent (e.g., visual loss, field or acuity, visual processing). Efferent (e.g., diplopia, pupil abnormalities, eye lid abnormalities, jumping eyes). Pain (e.g., headache, eye pain). Questioning About Afferent System Specifically define the following: Onset (e.g., upon waking, following a headache). Course (e.g., sudden, gradually progressive). Duration (e.g., seconds, minutes, hours, days). Severity of visual loss. Q: “Could you see faces?” Q: “Could you read?” Q: “Could you see motion?” Timing and onset of visual loss. Laterality (and did the patient cover one eye to check?). Monocular. Binocular. Progression. Visual field defect versus acuity loss. Associated pain (with eye movements?), headaches, nausea, or aura. Associated motor or sensory dysfunction. Associated scintillations or other visual phenomena. Efferent Localization Q: “Where is the lesion?” Monocular or binocular (diplopia). Vertical or horizontal diplopia. Worse in fields of gaze (comitance) ? Worse throughout day or fatigable? Pain Q: “When, where, how much?” Palliative and precipitating factors. Associated symptoms. Motility Examination Supraduction or elevation: Eye moves up. Infraduction or depression: down. Abduction: away from midline. Adduction: toward midline Intorsion: inward rotation of the upper pole of the vertical meridian of each eye toward the midline of the face Extorsion: outward rotation of the upper pole of the vertical meridian of each eye away from the midline of the face Table 6.1 Muscle actions Accommodative Amplitude (Right Eye and Left Eye) Deviations Distance with or without correction. Near with or without correction. Primary position and diagnostic positions. Primary and secondary deviation. Primary: fixing with nonparetic eye. Secondary: fixing with paretic eye. Secondary deviation greater than primary. More innervational effort to keep paretic eye fixating on target. Innervation drives fellow eye to greater deviation: Esodeviation: turns in. Exodeviation: turns out. Hyperdeviation: eye turned up. Hypodeviation: eye turned down. Torsion (incyclotorsion or excyclotorsion). Stereoacuity Testing 40 arc seconds: 20/20 acuity. 60 arc seconds: 20/40 acuity. 100 arc seconds: 20/200 acuity. Titmus testing (i.e., fly, animals, circles). Double Maddox rod testing for torsion. Distance and Near Testing Check primary and downgaze positions. Especially useful for superior oblique palsy. Can be compared to funduscopic torsion. Saccades and pursuit (horizontal and vertical). Latency. Amplitude. Accuracy. Speed (velocity). Motility: Special Tests Cover-Uncover Test for Tropia or Manifest Deviation Method Have the patient fixate on target. Cover one eye and look for shift in noncovered eye. Uncover eyes. Repeat test covering other eye. Absence of shift: straight (orthotropia). Shift of noncovered eye indicates tropia. Requires vision good enough to fixate on target. Alternate Cover Test for Phoria or Latent Deviation Method Hirschberg Test For patients unable to fixate. See Figure 6.2. Method Hold fixation light at 33 cm. Measure deviation of light reflex from pupil center, indicative of tropia. Decentration of 1 mm = 7 degrees = 15 prism diopters. Pupil edge margin = 15 degrees = 30 prism diopters. Iris = 30 degrees = 60 prism diopters. Limbus = 45 degrees = 90 prism diopters Krimsky Method For patients unable to fixate. Hold fixation light at 33 cm. Add prism in front of one eye until corneal light reflex is centered in both eyes. Semiquantitative assessment of deviation. FIGURE 6.2. Left esotropia. Note lateral displacement of Hirschberg light reflex in the LE. Pupils are pharmacologically dilated. Photograph demonstrates right ptosis and asymmetry of the red reflex, which is caused by misalignment of the eyes. Three-Step Test for Hypertropia (Bielschowsky/Parks Test) Step 1: Which eye is higher? Right eye (RE) or left eye (LE)? Step 2: Is the deviation worse in right or left gaze? Step 3: Is the deviation worse in right head tilt (RHT) or left head tilt (LHT)? Example Right depressors (R-IR, right superior oblique muscle [R-SO]). Left elevators (L-SR, left inferior oblique muscle [L-IO]). Only R-SO and L-SR act in gaze to the left (deviation worse in gaze toward paretic muscle). In tilt right, RE intorts and LE extorts to maintain position. Of the R-SO and L-SR, only the R-SO is an intorter of RE (L-SR is intorter of LE). Intorsion deficit is made up by the following: RE intorter = R-SR; therefore, secondary elevation by R-SR makes the RHT worse. Step 4: Measure ocular torsion (if torsion mismatch consider alternative diagnosis of hyperdeviation such as skew/ocular tilt reaction). Mnemonic: SINRAD = Superiors INtort Recti ADduct. Summary Nonorganic visual loss may manifest as loss of visual acuity or visual field. No organic lesion can be identified by examination, and nonphysiologic responses confirm the diagnosis. May be unilateral or bilateral. May be any form and severity of visual loss. Evaluation of Nonorganic Visual Loss Optokinetic Nystagmus Drum for Severe Loss (Not as Useful for Mild or Moderate Loss) Horizontal jerk nystagmus. One-inch-wide stripes indicate vision of at least 20/400. If “loss” occurs in only one eye: Rotate drum before both eyes. Once good nystagmus elicited, cover unaffected eye. Nonorganic loss continues jerk. Optokinetic nystagmus (OKN) may be suppressed by inattention. Mirror Test for No Light Perception Move mirror back and forth. Rotate and twist mirror. If acuity greater than LP, then nystagmoid movement. Difficult to suppress response. Proprioception Testing for Severe Visual Loss Ask the patient to touch the left and right fingertips together. A patient with organic disease performs the task easily; one with nonorganic disease cannot. This is a test of proprioception, not vision. Forced Acuity Testing Start with smallest line (20/10). Tell the patient that you are showing a line of “doubled” size. Show next larger line (20/15). Works best with computerized projections of multiple Snellen lines of same size. Repeat until the patient is able to read line. Express incredulously, “You can’t see these ‘big’ letters?” Near Vision Testing A discrepancy of near versus distance vision, if not refractive error or media, is usually evidence of nonorganic loss. Potential Acuity Meter Measure best corrected acuity. Dilate pupil. Tell the patient that the test “bypasses” visual problem. Improved PAM vision is evidence for ocular media opacity or nonorganic loss. Red-Green Glasses Method Project red/green duochrome slide. Red lens over abnormal eye. If the patient is able to read the entire line, then nonorganic visual loss. Red sees letters on both sides of the screen. Green lens, only green side. Mnemonic: “green over good eye.” May use Ishihara color plates. Polarized lenses. American Optical polarizing test. Polarized glasses. Project-O-Chart slide. Color testing. Optic neuropathy often causes color defect. Stereopsis Test binocularly first. RE 20/20 and LE 20/200 = stereo of 180 seconds of arc. OU 20/20 has 40 seconds of arc. Normal stereopsis = nonorganic acuity loss. Pupil Responses Eye with no light perception (NLP) should not react to light if from anterior visual pathway disease (patients will have normal pupil responses if cortical visual loss). Evaluation of Nonorganic Visual Field Defects Constricted Field Test at two different distances (e.g., 1 and 2 m). Increasing the distance from the screen should increase the mapped field proportionately. Size of object varied (i.e., if double test distance, double test object). A field that fails to expand at any distance (“tunnel”) is nonorganic. Test monocular and binocular field. If field loss persists under binocular conditions, then nonorganic. Saccadic Testing Saccades in supposedly blind peripheral field are a nonorganic finding. Tell the patient that it is a test of eye movements. Visual Field Defects Suggestive of Nonorganic Disease Bilateral constriction without expansion at increasing distance (tunnel field). Overlapping or inversion of isopters. Spiraling of isopters. Nasal hemianopias when tested monocularly that persist on binocular testing. Types of Visual Field Defects Etiology and evaluation based on pattern of visual field loss. Unilateral Nerve Fiber Bundle Defect Arcuate or altitudinal in shape. Scotoma extends to blind spot. Etiology Ipsilateral optic nerve or retinal lesion. Field defect size, density, and shape correspond to retinal or optic nerve lesion. Central or paracentral scotomas suggest macular or optic nerve disease. Bilateral nerve fiber layer or retinal defects may occur (e.g., glaucoma, retinal detachment). Compressive lesions may cause nerve fiber defects (optic neuropathy). Evaluation Careful ophthalmoscopy. Optic nerve head examination. Exclude artifact of testing. Exclude retinal disease. Exclude glaucoma. Exclude optic neuropathy. Bilateral Cecocentral or Central Scotomas May be bilateral optic neuropathy of any cause (e.g., compressive), but more common causes are listed below. Etiology Toxic or nutritional amblyopia. Bilateral demyelinating (optic neuritis). Syphilis. Bilateral macula-involving occipital lesions. Bilateral macular disease. Leber hereditary optic neuropathy. Evaluation Exclude toxic optic neuropathies (e.g., heavy metals, ethambutol). Consider nutritional amblyopia (vitamin B12, folate, complete blood cell count [CBC]). Neuroimaging. Testing for Leber hereditary optic neuropathy. Junctional Visual Field Defects Etiology Ophthalmic Findings Although purported to be from contralateral crossing fibers extending into ipsilateral optic nerve (Willbrand knee), it is unlikely to exist anatomically, but the clinical localizing finding of the junctional scotoma to the junction of the optic nerve and chiasm remains valid. Evaluation Neuroimaging (preferably magnetic resonance imaging [MRI]) of junction of the optic nerve and chiasm. Bitemporal Step or “Flavor” Visual Field Defect See Figure 6.3. FIGURE 6.3. Schematic illustration of the visual pathway and visual field defects produced by lesions in various areas of the pathway. (ON, optic nerve; CH, chiasm; OT, optic tract; LGB, lateral geniculate body; ML, Meyer loop; OR, optic radiations) (Reproduced from Tasman W, Jaeger E. The wills eye hospital atlas of clinical ophthalmology, 2nd ed. Lippincott Williams & Wilkins, 2001, with permission.) Chiasmal lesion until proven otherwise. Pseudochiasmal fields (not respecting midline). Tilted disk. Colobomas. Bilateral nasal retinal disease (e.g., schisis). Glaucoma. Bilateral optic neuropathy. Lesions affecting the chiasm: Pituitary adenoma. Meningioma. Craniopharyngioma. Internal carotid artery aneurysm. Other tumors. Demyelinating disease. Trauma. Evaluation Neuroimaging with attention to the chiasm. Homonymous Hemianopsia See Figure 6.3. Etiology Signs and Symptoms Signs and symptoms to help localize homonymous hemianopsia: Optic tract: Contralateral APD. Band or bow-tie optic disk atrophy (contralateral). Generally incongruous homonymous hemianopsia. Lateral geniculate body: Incongruous wedge defects “spears to fixation” (anterior choroidal branch of internal carotid artery). May spare or selectively involve horizontal strip of field (lateral choroidal branch). Temporal lobe: Defect denser superiorly (Meyer loop); “pie in the sky” scotoma (Figure 6.3). Parietal lobe: Incongruous defect, denser inferiorly (Figure 6.3). More likely complete with macular splitting. Other clinical signs: Right-sided lesions. Constructional apraxias. More severe neglect. Left-sided lesions. Gerstmann syndrome: Finger agnosia. Right-left confusion. Agraphia. Acalculia. Aphasia. Neglect. Occipital: Often isolated neurologically. Usually very congruous. May be paracentral. Macular sparing (central 5 degrees) due to dual circulation from middle and posterior cerebral arteries. May spare or selectively involve a temporal monocular crescent at anterior tip of calcarine cortex (Figure 6.3) supplied by anterior cerebral artery. Optic Nerve Head Disorders Coloboma Summary A coloboma is an excavated defect in the optic nerve that is due to incomplete closure of the embryonic fissure associated with a PAX6 mutation. Etiology Congenital: incomplete or abnormal closure of proximal end of embryonic fissure. Signs and Symptoms May be asymptomatic. May have visual loss. Ophthalmic Findings May be isolated. May be associated with iris, ciliary, or choroidal colobomas. May be associated with other ocular abnormalities (microphthalmia, choroidal colobomas). May be associated with other systemic abnormalities (e.g., Aicardi syndrome, Goldenhar syndrome, and CHARGE syndrome). Treatment None. Prognosis Usually static. Reference Brodsky MC. Congenital optic disc anomalies. Surv Ophthalmol 1994;39:89–112. Optic Pit Summary An optic pit is a congenital excavated defect or depression in the optic nerve due to a defect in the lamina cribrosa. Etiology Congenital. Signs and Symptoms May be asymptomatic. May have visual loss. May have metamorphopsia. Ophthalmic Findings Small excavated defect (pit) in optic nerve rim (Figure 6.5). Usually temporal (may be nasal or central). Bilateral in 15% of cases. May be associated with serous retinal detachment. Adjacent to optic pit. May extend into macula. FIGURE 6.5. Optic pit temporally on the optic nerve. May have an associated serous retinal detachment. (Reproduced from Tasman W, Jaeger E. The Wills eye hospital atlas of clinical ophthalmology, 2nd ed. Lippincott Williams & Wilkins, 2001, with permission.) Special Tests Fluorescein angiography for serous detachment. Treatment No treatment unless serous detachment. Laser treatment if serous detachment though often unsuccessful. Vitrectomy in unresponsive cases. Prognosis Often, self-limited serous detachment. May recur. Reference Brodsky MC. Congenital optic disc anomalies. Surv Ophthalmol 1994;39:89–112. Optic Nerve Hypoplasia and Dysplasia Summary Optic nerve hypoplasia is a congenitally small optic nerve. Etiology Congenital. Aplasia (rare). Hypoplasia. Dysplasia. Signs and Symptoms Sometimes asymptomatic. May have decreased acuity. Range: 20/20 to NLP. Usually nonprogressive. Cannot judge level of acuity from appearance of disk. Visual field defects. Ophthalmic Findings Unilateral or bilateral. Strabismus. Nystagmus (including see-saw). May have superimposed amblyopia. Strong association with astigmatism. Fundus signs. Small optic disk. One third to half the normal size. With or without pallor. Normal optic nerve dimension: horizontal, 1.38 mm; vertical, 1.46 mm. “Double ring sign” (Figure 6.6). May be confused with peripapillary atrophy. Outer ring: junction of sclera and lamina cribrosa. Inner ring: abnormal extension of retina and RPE. Decreased nerve fiber layer thickness. Normal-sized retinal vessels that may be tortuous. Associated Signs Developmental delay, most common. Hypothalamic or pituitary dysfunction. Endocrine dysfunction. Facial abnormalities. Skeletal abnormalities. Ocular abnormalities (e.g., coloboma). Risk Factors (Usually Idiopathic) Young maternal age. Use of medicines in pregnancy (e.g., alcohol, phenytoin). Maternal diabetes (superior segmental hypoplasia). Trisomies 13, 18, and 21. Intrauterine cytomegalovirus and hepatitis. Evaluation Pediatric endocrinology consultation. Hypothalamic hypopituitarism. Neonatal hypoglycemia. Diabetes insipidus. Growth hormone deficiency. Thyroid hormones. Neuroimaging. Special Tests Stereo disk photos. Formal visual field tests when the patient is old enough. Treatment Underlying endocrine disease. Trial of patching for amblyopia. Correct refractive errors (astigmatism). Safety spectacles. Developmental Abnormalities of Midline Brain Structures (De Morsier Syndrome—Septooptic Dysplasia) Agenesis of anterior commissure. Absent septum pellucidum and corpus callosum. Central nervous system (CNS) basal encephalocele. Testing for pituitary endocrine dysfunction is warranted. Reference Brodsky MC. Congenital optic disc anomalies. Surv Ophthalmol 1994;39:89–112. Other names: Fuchs coloboma. Inferior conus. Inversion or dysversion of disk. Summary The optic disk may be congenitally tilted in one or both eyes, and this may result in a visual field defect. Signs and Symptoms Eighty percent of cases bilateral. Ophthalmic Findings See Figure 6.7. Oval disk (with reduction in disk diameter). Depression usually inferiorly. Elevation usually superiorly. Crescent, usually inferior (in direction of tilt). Oblique direction of retinal vessels (emerging from superotemporal disk, sweep nasally before turning temporally). Hypopigmentation (wedge-shaped area, opposite defective portion of the disc). Visual field defects (usually superotemporal, may have bitemporal field defects simulating a chiasmal lesion, but defects do not respect vertical midline). Evaluation Optic disk photos. Attention of field testing to vertical meridian. Ectasia should match field defect location. If vertical step in field, MRI scan. Prognosis Usually static. FIGURE 6.7. The congenital tilted optic disk is apparent as the oval of nerve tissue superiorly. There is no apparent optic cup. Treatment None. Reference Giuffe G. The spectrum of the visual field defects in the tilted disk syndrome: clinical study and review. Neuroophthalmology 1986;6:239–246. Papilledema Summary Papilledema is a term that should be reserved for optic disk edema attributable to increased intracranial pressure. All other forms of optic disk swelling should be referred to as optic disk edema. Etiology Any cause of increased intracranial pressure: Neoplastic (e.g., glioblastoma multiforme). Infectious (e.g., abscess, meningitis). Inflammatory (e.g., sarcoid). Traumatic. Infiltrative. Vascular (e.g., arteriovenous malformation [AVM]). Subarachnoid hemorrhage. Venous sinus thrombosis. Hydrocephalus. Pseudotumor cerebri. Signs and Symptoms Usually bilateral but may be unilateral or asymmetric. Usually preserved visual acuity and color discrimination early. May have transient visual loss (TVL) lasting for a few seconds resulting in obscurations of vision. Ophthalmic Findings Disk hyperemia: congestion of fine optic nerve capillaries. Opacification of nerve fiber layer. Peripapillary hemorrhages. Cotton-wool spots. Loss of the physiologic cup is a late sign. Venous dilation: elevation of optic nerve head. Retinal exudates. Circumferential retinal folds (Paton lines) in peripapillary region. Glaucomalike defects. Evaluation Neuroimaging (to exclude mass lesion). Cranial MRI with contrast MR venogram (MRV) to exclude cerebral venous sinus thrombosis is recommended. Lumbar puncture. Opening pressure. Cell count. Glucose, protein, cytologic testing, cultures. Differential Diagnosis Pseudopapilledema. Optic disk drusen. Hyperopic disks. Other causes of bilateral optic disk edema. Anterior ischemic optic neuropathy (ION). Diabetic papillitis. Optic neuritis. Inflammatory or infiltrative optic disk edema. Treatment Treat underlying cause. Acetazolamide. Corticosteroids (but may worsen weight gain or pseudotumor cerebri). Surgical, if the patient fails medical therapy. Optic nerve sheath fenestration. Ventriculoperitoneal or lumboperitoneal shunt. Bariatric surgery for weight loss in pseudotumor cerebri. Pseudotumor Cerebri Summary Pseudotumor cerebri is defined as increased intracranial pressure with normal neuroimaging and normal cerebrospinal fluid (CSF) content. Etiology Usually idiopathic. Multiple associations: Sagittal sinus thrombosis. Radical neck surgery. Hypoparathyroidism. Addison disease. Renal disease. Systemic lupus erythematosus. Behçet disease. Vitamin A. Tetracycline. Steroid withdrawal. Nalidixic acid. Anabolic steroids. Lithium. Epidemiology Usually young, obese women. Beware of the diagnosis of pseudotumor cerebri in men, thin patients, or elderly individuals. Signs and Symptoms Papilledema (does not have to be present for diagnosis) (Figure 6.9). Nonlocalizing sixth-nerve palsy, but no other cranial nerves involved or focal neurologic signs. Headache. Transient visual obscurations (seconds). Pulsatile tinnitus. Evaluation Neuroimaging. Preferably MRI with contrast MRV. Lumbar puncture, check opening pressure. FIGURE 6.9. Papilledema secondary to pseudotumor cerebri. Both optic nerves had a similar appearance. Indications: Severe intractable headache unresponsive to headache medications. Progressive visual loss. Treat underlying associated conditions. Eliminate exogenous factors. Weight loss. Medications: Acetazolamide. Less commonly, other medicines (e.g., digoxin, furosemide, topiramate). Corticosteroids (see warning above). Surgical therapy: Optic nerve sheath fenestration. Ventriculoperitoneal or lumboperitoneal shunt. Special Testing Visual field testing. Optic disk photography. References Corbett JJ, Thompson HS. The rational management of idiopathic intracranial hypertension. Arch Neurol 1989;46: 1049–1051. Wall M, George D. Idiopathic intracranial hypertension (pseudotumor cerebri): a prospective study of 50 patients. Brain 1991; 114:155–180. Optic Nerve Head Drusen Summary Optic nerve head drusen are calcified hyaline bodies within the optic disk substance that may cause visual field loss. Etiology Congenital. Autosomal dominant in some cases. Histopathology Hyaline bodies; may be calcified. Acellular laminated concretions in optic nerve substance. Demographics Usually white patients. Uncommon in black patients. Signs and Symptoms Usually asymptomatic. May have visual field defects (slow progression). (often arcuate, an enlarged blind spot, or visual field constriction). Decreased visual acuity is rare, usually not due to drusen alone. May have transient visual obscurations. Differential Diagnosis Arteritic ischemic optic neuropathy (AION). Retinal artery or vein occlusion. Subretinal neovascular membrane. Subretinal exudation or heme. Systemic Diseases Associated conditions: Retinitis pigmentosa. Pseudoxanthoma elasticum. Angioid streaks. Retinal hemorrhages. Chorioretinal folds. Migraine. Ophthalmic Findings Usually bilateral (66%). Discrete, amorphous deposits in optic nerve (Figure 6.10). More often nasal than temporal. Absent cup, small disk. Anomalous disk vasculature. No exudates or disk telangiectasias. Tend to become more apparent with age. Buried disk drusen in children. Drusen transilluminate within disk head. Subfoveal neovascularization. Evaluation A clinical diagnosis. Examine family members. FIGURE 6.10. Optic nerve drusen cause the surface of the optic nerve to appear bumpy. These drusen can become calcified and are visible on ultrasonography. Ultrasound of nerve head (for buried drusen). Computed tomography (CT) scan (rarely needed) may show calcific drusen. Preinjection fluorescein angiography. Autofluorescence. Probably not helpful in buried drusen. May show delay in filling of peripapillary choroid. Stereo disk photos. Reference Rosenberg MA, Savino PJ, Glaser JS. A clinical analysis of pseudopapilledema, I: population, laterality, acuity, refractive error, ophthalmoscopic characteristics, and co-incident disease. Arch Ophthalmol 1979;97:65–70. Melanocytoma Summary A melanocytoma is a congenital pigmented abnormality of the optic disk head. Etiology Congenital. Signs and Symptoms Usually asymptomatic. Ophthalmic Signs Black optic nerve head lesion (Figure 6.11). May be slightly elevated. FIGURE 6.11. Melanocytomas are black lesions frequently found off the optic nerve. They represent benign melanocytic proliferation. Special Tests Usually, none needed. Prognosis May grow slowly. Treatment None. Differential Diagnosis Melanoma. Combined hamartoma of the retina and RPE. Reference Joffe L, Shields JA, Osher RH, et al. Clinical and follow-up studies of melanocytomas of the optic disc. Trans Am Acad Ophthalmol Otolaryngol 1979;86:S1067–S1078. Myelinated Nerve Fiber Layer Summary Myelination normally terminates at the lamina cribrosa but may occur in the nerve fiber layer of the retina. Etiology Congenital. Signs and Symptoms Usually asymptomatic. Ophthalmic Findings White myelin in distribution of nerve fiber layer, often radiating from the optic nerve, but may occur only in the peripheral retina (Figure 6.12). Evaluation Clinical diagnosis. Prognosis Static. Treatment None. Differential Diagnosis Cotton-wool patch. Retinal infiltrate. Arteriole occlusion. Optic Nerve and Chiasm Disorders Ischemic Optic Neuropathy Summary ION is a clinical syndrome characterized by acute loss of vision, evidence for an optic neuropathy, and usually a swollen optic nerve in an elderly patient. Etiology Arteritic ION: giant cell arteritis (GCA). Nonarteritic ION: other vasculopathic risk factors (e.g., diabetes, atherosclerosis). Signs and Symptoms Acute loss of visual acuity and/or visual field (usually altitudinal defect) (Figure 6.13). Dyschromatopsia. Usually unilateral. Usually painless. Usually more severe visual loss. May be bilateral. Constitutional symptoms and signs (headache, jaw claudication, scalp tenderness, malaise, fever, weight loss, anorexia, etc.). Demographics Usually occurs in older patients (>40 years). Elderly patients evaluate for GCA (>50 years). Patients often have vasculopathic risk factors. Ophthalmic Findings Usually swollen optic nerve (anterior ION). Relative afferent pupillary defect in unilateral or bilateral asymmetric cases. Absent anterior or posterior segment inflammation. FIGURE 6.13. Inferior altitudinal defect in a patient with anterior ION. Notice that the defect respects the horizontal midline. (From Miller N, Newman N. Walsh and Hoyt’s clinical neuro-ophthalmology, 4th ed., vol. 1. Baltimore, MD: Williams & Wilkins, 1995:213, with permission.) Systemic Findings Diabetes. Hypertension (left ventricular hypertrophy). Smoking. Hypercholesterolemia. Hypotension (e.g., postsurgical, nocturnal, other) or anemia. Collagen-vascular disease. GCA. Special Tests Erythrocyte sedimentation rate (ESR) and/or C-reactive protein test for GCA. Complete blood count with platelet count. Temporal artery biopsy for GCA. Pathology Temporal artery inflammation with or without giant cells for GCA. Disruption of internal elastic lamina. Intimal hyperplasia. Disease Course Often, static visual loss. May have progressive visual loss. Small cup-to-disk ratio predisposing factor (“disk at risk”). Often, severe visual loss. Untreated disease leads to bilateral severe visual loss. Treatment Treat underlying vasculopathic risk factors. Aspirin (one per day) therapy may reduce risk in fellow eye (conflicting data). No role for optic nerve sheath decompression (see Ischemic Optic Neuropathy Decompression Trial study). Oral prednisone (1.0 to 1.5 mg/kg/day). Consider intravenous (i.v.) steroids for monocular patients, bilateral, or severe visual loss. Follow-Up Nonarteritic: as needed. Arteritic: 65% of patients develop arteric ION in fellow eye if untreated. Follow ESR and clinical symptoms. Taper steroids slowly. Differential Diagnosis Infectious optic neuropathy. Inflammatory optic neuropathy. Infiltrative optic neuropathy. Compressive optic neuropathy. Toxic optic neuropathy. Hereditary optic neuropathy (Leber hereditary optic neuropathy). References Hayreh SS, Podhajsky PA, Raman R, et al. Giant cell arteritis: validity and reliability of various diagnostic criteria. Am J Ophthalmol 1997;123:285–296. The Ischemic Optic Neuropathy Decompression Trial Research Group. Optic nerve decompression surgery for nonarteritic anterior ischemic optic neuropathy (NAION) is not effective and may be harmful. JAMA 1995;273:625–632. The Ischemic Optic Neuropathy Decompression Trial Research Group. Characteristics of patients with nonarteritic anterior ischemic optic neuropathy eligible for the Ischemic Optic Neuropathy Decompression Trial. Arch Ophthalmol 1996;114:1355–1374. Optic Neuritis Summary Optic neuritis is a general term that is used to describe idiopathic or demyelinating optic neuropathy. Etiology Idiopathic: most frequent. Demyelinating diseases: multiple sclerosis (MS). Infectious diseases: viral disease, syphilis, cat scratch disease, Lyme disease, etc. Inflammatory diseases: collagen-vascular disease, sarcoid, etc. Signs and Symptoms Unilateral, acute visual loss. Pain worse with eye movement. Bilateral simultaneous onset in adults. Anterior or posterior segment inflammation. Macular star figure. Lack of pain. Male. NLP vision. Significant retinal hemorrhages or cotton wool patches. Lack of significant improvement. History or examination findings suggestive of underlying inflammatory or infectious process. Demographics Usually young adults, but may occur in any age group. Often female patients, but may be either gender. Ophthalmic Findings Decreased visual acuity and/or visual field. Relative afferent pupillary defect in unilateral or bilateral but asymmetric cases. Absence of anterior or posterior uveitis. Normal (65%) or swollen (35%) optic nerve (most cases are retrobulbar). Systemic Findings Associated with MS. Special Tests MRI of the brain and orbits to look for demyelinating white matter disease (Figure 6.14). Pathology Optic nerve demyelination. Disease Course Ninety percent of cases improve to normal or near-normal vision with or without treatment. Evaluation Typical optic neuritis: MRI to evaluate for demyelinating disease. No further laboratory evaluation necessary. Atypical optic neuritis MRI head and orbit with fat suppression and contrast. Laboratory evaluation directed by history and physical examination (e.g., syphilis serologic testing, antinuclear antibody testing, chest radiography, and lumbar puncture). Treatment Can observe for improvement. Consider i.v. steroids (methylprednisolone 1,000 mg/day for 3 days followed by oral steroid taper [See Optic Neuritis Treatment Trial study]). I.V. steroids hasten visual recovery but do not change ultimate visual outcome. Consider i.v. steroids. Consider further evaluation for MS. Neurologic consultation for consideration of immunomodulatory treatments (interferon beta). Follow-Up Visual recovery over 4 to 6 weeks. Risk of MS. Differential Diagnosis Infectious optic neuropathy. Inflammatory optic neuropathy. ION. Infiltrative optic neuropathy. Compressive optic neuropathy. Toxic optic neuropathy. Hereditary optic neuropathy (Leber hereditary optic neuropathy). References Beck RW, Cleary PA, Trobe JD, et al. The effect of corticosteroids for acute optic neuritis on the subsequent development of multiple sclerosis. N Engl J Med 1992;326:581–588. Beck RW, Trobe JD, the Optic Neuritis Treatment Group. What we have learned from the Optic Neuritis Treatment Trial. Ophthalmology 1995:102;1504–1508. Toxic or Nutritional Optic Neuropathy Summary Patients with toxic or nutritional optic neuropathies usually present with bilaterally symmetric, slowly progressive visual loss. Signs and Symptoms Visual field defect: typically central or cecocentral scotomas. Visual acuity loss. Bilateral and symmetric visual loss. Slowly progressive. Optic nerve may appear normal until late in course. Optic atrophy usually develops eventually. Etiology Pernicious anemia. Dietary deficiency (e.g., veganism, alcoholism). Vitamin B12 deficiency. Folate. Amantadine. Amiodarone. Amoproxan. Aniline dyes. Barbiturates. Cafergot. Cabromal. Carbon disulfide. Carbon tetrachloride. Cephaloridine. Chloramphenicol. Chlorodinitrobenzene. Clioquinol. Dichlorodiphenyltrichloroethane. Digitalis. Dinitrotoluene. Disulfiram (Antabuse). D-Penicillamine. Elcatonin. Emetine. Ethambutol. Ethchlorvynol (Placidyl). Ethylene glycol. 5-Fluorouracil. Glue sniffing. Halogenated hydroxyquinolone. Heavy metals (e.g., arsenic, lead, mercury). Hexachlorophene. Iodoform. Iodopyracet (Diodrast). Isoniazid. Lysol. Methanol. Methyl acetate. Methyl bromide. Octamoxin. Organophosphate pesticides. Penicillamine. Phenazone (antipyren). Pheniprazine (Catron). Plasmocid. Quinine. Sodium fluoride. Streptomycin. Styrene (vinyl benzyl). Sulfonamides. Thallium. Thioglycolates. Tolbutamide (Orinase). Trichloroethylene. Vincristine. Evaluation MRI of the head. Vitamin B12 level. Folate level (serum and erythrocyte). CBC. Urine heavy metal screen (mercury, lead, arsenic) if history is suggestive. Leber Hereditary Optic Neuropathy Summary Leber hereditary optic neuropathy is a rare inherited mitochondrial optic neuropathy that affects young male patients. Etiology Hereditary defect in mitochondrial DNA. Demographics Affects men, usually in their twenties. Rarely affects women (10% to 20% of cases). Mitochondrial inheritance. Signs and Symptoms Usually acute, rapid, unremitting, and painless. Usually 20/200 to hand motions but 20/20 to NLP range. 1. Telangiectatic microangiopathy of optic neuropathy (Figure 6.15). 2. Apparent swelling of nerve fiber layer around disk. 3. Fluorescein angiogram often shows pseudoedema, but usually no leakage of dye at edge of disk. Attenuated arterioles. Nerve fiber layer loss, especially, papillomacular bundle. Optic nerve pallor (temporally). May develop nonglaucomatous cupping. Associations Usually systemically normal but may have some associated findings. Occasional cardiac conduction defects. Dystonia and other minor neurologic deficits. Evaluation Exclude other causes for an optic neuropathy. Primary mitochondrial DNA mutation (e.g., mtDNA 11778, 3460, 14484). Treatment Reducing metabolic stress on the optic nerve (e.g., smoking, known optic nerve toxins, and trauma). Medical therapy remains unproven. Multivitamins. Folate. Vitamin B12. Thiamine. Idebenone and other coenzyme Q10 analogues. FIGURE 6.15. Hyperemic optic disk with telangiectatic vessels in a young male patient with Leber hereditary optic neuropathy. Prognosis Some patients experience spontaneous improvement. Most remain unchanged. References Chalmers RM, Harding AE. A case-control study of Leber’s hereditary optic neuropathy. Brain 1996;119:1481–1486. Riordan-Eva P, Sanders MD, Govan GG, et al. The clinical features of Leber’s hereditary optic neuropathy defined by the presence of a pathogenic mitochondrial DNA mutation. Brain 1995;118:319–337. Infiltrative or Inflammatory Optic Neuropathy Summary Infiltration or inflammation of the optic nerve may mimic demyelinating or idiopathic optic neuritis. Signs and Symptoms Visual loss. Decreased color vision. Ophthalmic Findings Unilateral or bilateral. Afferent pupillary defect. Optic disk pallor or swelling. Optic disk may appear normal (retrobulbar). Atypical features for optic neuritis: Bilateral simultaneous onset. Lack of pain. Lack of improvement. Anterior or posterior segment inflammation. Progressive or recurrent episodes. Steroid responsive or steroid dependent. Etiology Plasmacytoma. Carcinomatous meningitis. Leukemia. Lymphoma. Cryptococcal meningitis. Aspergillus. Tuberculosis (TB). Syphilis. Contiguous sinus disease. Sarcoidosis. Systemic lupus erythematosus. Inflammatory bowel disease. Evaluation Optic Pathway Tumors Two common tumors of the optic pathway include optic nerve gliomas and meningiomas. Optic Gliomas Summary Optic gliomas are glial tumors that may affect the optic nerve, chiasm, or tract and are found primarily in children and young adults. Epidemiology Age: Can occur at any age. Seventy percent of patients with gliomas are diagnosed by age 10. Ninety percent of gliomas are detected by age 20. Reported in adults. No gender predilection. Association Neurofibromatosis (NF) type 1. Incidence (Uncommon) One to three percent of all orbital tumors. One percent of intracranial neoplasms. Three to six percent of intracranial tumors in childhood. Location One or both optic nerves. In general, the more anterior, the better the prognosis. Disease Course Most gliomas are benign juvenile pilocytic astrocytomas. Mortality: depends more on location than histologic appearance. Optic nerve alone, 14%. Chiasm, 47% (variable). With hydrocephalus, 77%. Hypothalamic involvement, 91%. Malignant optic nerve gliomas occur rarely in adults, usually men. Signs and Symptoms Painless progressive visual loss (optic neuropathy). Overall vision loss 87%. Vision of 20/20 to 20/40, 26%. Vision of 20/50 to 20/200, 19%. Vision of 20/300 or worse, 55%. Hypothalamic symptoms or endocrinologic. Diabetes insipidus. Diencephalic wasting. Precocious puberty. Somnolence. Growth failure. Visual field defects (central or bitemporal). Ophthalmic Findings Proptosis (orbital, 94%; chiasmal, 18%). Disk swelling (35%) or atrophy (60%). Rare optociliary shunt vessels. Fifty percent of intraorbital tumors have disk edema. Only 20% of chiasmal tumors have disk edema. Strabismus (27%). Nystagmus (spasmus nutans–like or see-saw nystagmus). Evaluation Consider glioma in any child with the following: Unexplained decreased visual acuity. Monocular or asymmetric nystagmus. Optic atrophy. Hypothalamic symptoms. Diencephalic syndrome. Spasmus nutans. See-saw nystagmus. Hydrocephalus. Neuroimaging: MRI scan head and orbit with fat suppression with gadolinium superior to CT scan (Figure 6.16). Can appear as a fusiform enlargement of optic nerve and/or kinking of optic nerve. Search for stigmata of NF. Assessment of growth and monitoring for precocious puberty. Examine family members. No biopsy usually indicated: If clearly intrinsic to nerve or chiasm. Intra-axial tumor. History of NF type 1. Possible biopsy indications. Atypical features. Infiltration or compression of chiasm. FIGURE 6.16. Magnetic resonance image with gadolinium enhancement of a fusiform, hyperintense lesion corresponding to an optic nerve glioma.
 Head posture, head tilt, face position.
Head posture, head tilt, face position.
 Fixation preference (near and distance).
Fixation preference (near and distance).
 Fixing with paretic eye or nonparetic eye.
Fixing with paretic eye or nonparetic eye.
 Dominance or better vision in fixing eye.
Dominance or better vision in fixing eye.
 Ductions (graded-4 [no movement] to 0 [normal]).
Ductions (graded-4 [no movement] to 0 [normal]).
 Monocular movements.
Monocular movements.
 Cover fellow eye during testing.
Cover fellow eye during testing.
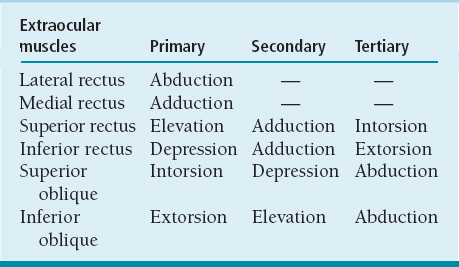
 Movements (Table 6.1 and Figure 6.1):
Movements (Table 6.1 and Figure 6.1):
 Most elevation is due to superior rectus muscle (SRM) and most depression is due to inferior rectus muscle (IRM).
Most elevation is due to superior rectus muscle (SRM) and most depression is due to inferior rectus muscle (IRM).
 Versions.
Versions.
 Binocular movements.
Binocular movements.
 Ductions may be better than versions.
Ductions may be better than versions.
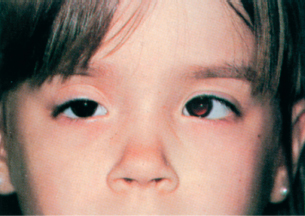
 Check cover-uncover test first; result must be negative (no tropia).
Check cover-uncover test first; result must be negative (no tropia).
 Have the patient fixate on target.
Have the patient fixate on target.
 Cover one eye.
Cover one eye.
 Rapidly move cover to other eye and look for shift in eye as it is uncovered.
Rapidly move cover to other eye and look for shift in eye as it is uncovered.
 Latent deviation kept in check by fusion.
Latent deviation kept in check by fusion.
 Cover-uncover test breaks down fusional control.
Cover-uncover test breaks down fusional control.
 Latent deviation revealed by eye movement when cover moved to other eye.
Latent deviation revealed by eye movement when cover moved to other eye.
 RE higher = right hypertropia (RHT).
RE higher = right hypertropia (RHT).
 Four possible paretic muscles.
Four possible paretic muscles.
 If deviation is worse in left gaze:
If deviation is worse in left gaze:
 If deviation is worse in RHT:
If deviation is worse in RHT:
 Diagnosis: R-SO palsy.
Diagnosis: R-SO palsy.
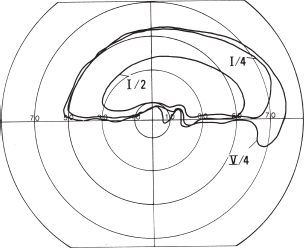
 Test with tangent screen.
Test with tangent screen.
 Monocular hemianopias or binasal or bitemporal hemianopia.
Monocular hemianopias or binasal or bitemporal hemianopia.
 Overlapping field from other hemifield.
Overlapping field from other hemifield.
 Usually, a mass lesion–compressing junction of optic nerve at chiasm (e.g., pituitary adenoma, craniopharyngioma, aneurysm).
Usually, a mass lesion–compressing junction of optic nerve at chiasm (e.g., pituitary adenoma, craniopharyngioma, aneurysm).
 Ipsilateral central or cecocentral visual field defect and superotemporal field defect in contralateral eye (junctional scotoma) (Figure 6.3).
Ipsilateral central or cecocentral visual field defect and superotemporal field defect in contralateral eye (junctional scotoma) (Figure 6.3).
 Monocular ipsilateral hemianopic (nasal or temporal) visual field defect (junctional scotoma of Traquair).
Monocular ipsilateral hemianopic (nasal or temporal) visual field defect (junctional scotoma of Traquair).
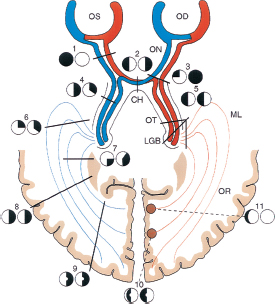
 Retrochiasmal disease.
Retrochiasmal disease.
 Complete homonymous hemianopsia is not further localizing (i.e., localizes only to contralateral retrochiasmal pathway).
Complete homonymous hemianopsia is not further localizing (i.e., localizes only to contralateral retrochiasmal pathway).
 More congruous (equal in appearance, size, shape of field) lesions are more posterior.
More congruous (equal in appearance, size, shape of field) lesions are more posterior.
 Congruity does not apply to complete hemianopsia.
Congruity does not apply to complete hemianopsia.
 Abnormal appearance of optic nerve.
Abnormal appearance of optic nerve.
 Glistening white bowl-shaped excavation occupies an enlarged optic disk (Figure 6.4).
Glistening white bowl-shaped excavation occupies an enlarged optic disk (Figure 6.4).
 Bare sclera visible because of absence of retina, retinal pigment epithelium (RPE), and choroid adjacent to and continuous with optic nerve.
Bare sclera visible because of absence of retina, retinal pigment epithelium (RPE), and choroid adjacent to and continuous with optic nerve.
 Usually inferonasal.
Usually inferonasal.
 Unilateral or bilateral.
Unilateral or bilateral.
 Absolute scotoma corresponding to coloboma.
Absolute scotoma corresponding to coloboma.
 Associated findings:
Associated findings:
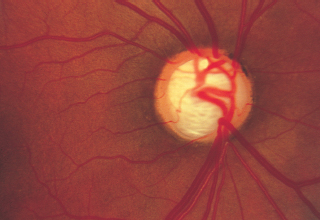
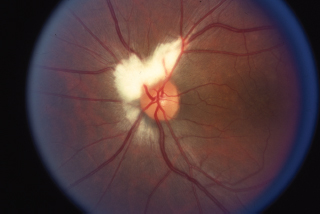
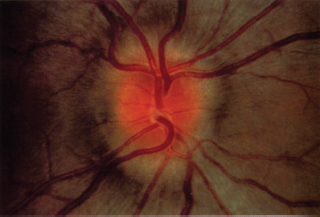
 Optic disk edema (Figure 6.8)
Optic disk edema (Figure 6.8)
 Visual field defects (enlarged blind spot, generalized constriction).
Visual field defects (enlarged blind spot, generalized constriction).
 No afferent pupillary defect unless severe and asymmetric.
No afferent pupillary defect unless severe and asymmetric.
 Absent spontaneous venous pulsations.
Absent spontaneous venous pulsations.
 Venous outflow obstruction.
Venous outflow obstruction.
 Endocrine, metabolic, or systemic diseases.
Endocrine, metabolic, or systemic diseases.
 Exogenous agents.
Exogenous agents.
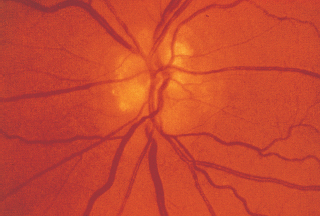
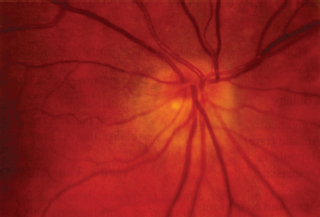
 Nonarteritic:
Nonarteritic:
 Arteritic:
Arteritic:
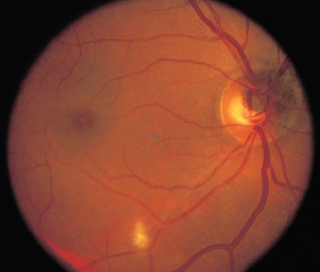
 Nonarteritic anterior ION:
Nonarteritic anterior ION:
 Arteritic:
Arteritic:
 Nonarteritic:
Nonarteritic:
 Arteritic:
Arteritic:
 Nonarteritic:
Nonarteritic:
 Arteritic:
Arteritic:
 Typical optic neuritis is characterized by the following clinical features:
Typical optic neuritis is characterized by the following clinical features:
 Atypical features:
Atypical features:
 If normal MRI imaging and typical optic neuritis:
If normal MRI imaging and typical optic neuritis:
 If abnormal MRI consistent with MS:
If abnormal MRI consistent with MS:
 Nutritional deficiencies:
Nutritional deficiencies:
 Toxins:
Toxins:
 Visual acuity loss:
Visual acuity loss:
 Visual field loss (central or cecocentral scotomas).
Visual field loss (central or cecocentral scotomas).
 Bilateral involvement (second eye affected days to weeks to months later but 40% to 50% have simultaneous onset).
Bilateral involvement (second eye affected days to weeks to months later but 40% to 50% have simultaneous onset).
 Fundus findings at the time of visual loss.
Fundus findings at the time of visual loss.
 Triad of the “suspect fundus”:
Triad of the “suspect fundus”:
 Fundus appearance after visual loss:
Fundus appearance after visual loss:
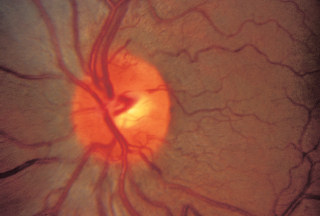
 Neoplastic:
Neoplastic:
 Paraneoplastic disease.
Paraneoplastic disease.
 Infectious:
Infectious:
 Systemic inflammatory diseases:
Systemic inflammatory diseases:
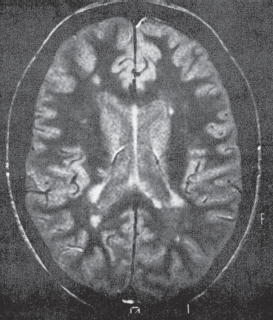
 MRI of the head (and orbit if orbital signs present).
MRI of the head (and orbit if orbital signs present).
 CBC.
CBC.
 Syphilis serologic testing (e.g., fluorescent treponemal antibody, absorbed [FTA-Abs], rapid plasmin reagin [RPR]).
Syphilis serologic testing (e.g., fluorescent treponemal antibody, absorbed [FTA-Abs], rapid plasmin reagin [RPR]).
 Antinuclear antibody test.
Antinuclear antibody test.
 Chest radiograph.
Chest radiograph.
 Angiotensin-converting enzyme, lysozyme.
Angiotensin-converting enzyme, lysozyme.
 Lumbar puncture.
Lumbar puncture.
 Consider gallium or PET scan if suspect sarcoidosis.
Consider gallium or PET scan if suspect sarcoidosis.
 Purified protein derivative skin testing if TB suspected.
Purified protein derivative skin testing if TB suspected.
 Anti–double-stranded DNA, complement levels if systemic lupus erythematosus suspected.
Anti–double-stranded DNA, complement levels if systemic lupus erythematosus suspected.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree