Surgical Approaches for Coexisting Glaucoma and Cataract
In the management of a patient with a visually significant cataract and coexisting glaucoma, there are three basic surgical approaches: (a) cataract extraction alone, which may need to be followed by a trabeculectomy later; (b) glaucoma filtering surgery alone, followed by cataract removal later (two-stage approach); and (c) combined cataract and glaucoma surgery. Combined procedures have certain advantages and disadvantages compared with the other two options. Compared with cataract surgery alone—which itself is associated with an increased risk for posterior capsule break in glaucomatous eyes, particularly when exfoliation is present (1–4)—combined procedures are associated with a greater risk for postoperative complications, such as increased inflammation, hyphema, hypotony, shallow anterior chambers, and choroidal detachments; however, they have the advantage of reducing early intraocular pressure (IOP) rise. Compared with filtering surgery alone, with or without subsequent cataract extraction, the combined procedures may have a lower chance of long-term glaucoma control, but have the obvious advantage of a single surgery instead of two. For these reasons, the surgeon should consider each of the basic surgical options, evaluate the severity of the glaucoma, and the visual need and potential for each individual patient, and select the approach that seems most appropriate. With advances in cataract and glaucoma surgery, success rates with combined procedures have improved and relative indications have shifted. We first review the general indications for the three basic surgical approaches and then consider how advances in surgical techniques are influencing the relative indications for these procedures.
INDICATIONS
Predicting Visual Potential
In each case, it is assumed that a cataract is present for which extraction is indicated, independent of the glaucoma. However, the visual significance of the cataract is often difficult to determine in an eye with both a cataract and glaucoma, in which it is hard to know how much the glaucoma is contributing to the reduced vision. Several instruments have been developed to help predict the anticipated postoperative visual acuity. One focuses a miniaturized Snellen visual acuity chart on the retina (potential acuity meter), whereas others project stripe patterns from either a laser or white light source (Visometer). Potential acuity meter measurements do not always show good correlation to postoperative results, particularly for dense cataracts (5,6). In one study, the Visometer gave more accurate predictions than the potential acuity meter did in patients with cataract and chronic open-angle glaucoma (COAG), even with glaucomatous field loss (7). In other studies, the potential acuity meter was accurate when the glaucomatous damage was mild to moderate and the postoperative visual acuity was 20/40 to 20/50 or better, whereas the results with advanced visual field loss or a worse postoperative vision were unreliable (8,9). Automated perimetry was useful in predicting whether the vision would be better or worse than 20/ 40. Combining this with the use of the potential acuity meter further increased the predictive value (9).
When it is decided that cataract surgery is needed, the selection of the specific surgical approach is based primarily on the status of the glaucoma.
Cataract Extraction Alone
Most surgeons prefer to perform cataract extraction alone when the IOP is well controlled medically in the presence of mild to moderate glaucomatous optic neuropathy. However, cataract extraction with placement of a posterior chamber intraocular lens (IOL) can be associated with a significant IOP rise during the early postoperative course in patients with preexisting glaucoma, especially when older, extracapsular techniques are used (10–15), or when viscoelastic material is not completely removed from the eye. Although outflow facility seems to improve after phacoemulsification (16), the IOP can still be significantly elevated in the first 24 hours. Peak IOP elevation after cataract extraction often occurs 2 hours postoperatively (17,18). After extracapsular cataract extraction (ECCE) or phacoemulsification with posterior chamber IOL implantation in patients with glaucoma, more than half of patients may have an IOP greater than 25 mm Hg, or even 35 mm Hg, indicating the need for close monitoring and prophylactic medical treatment to prevent postoperative IOP spikes (12,16,19). A significant increase in IOP during the first 5 to 7 hours after surgery has been found after both ECCE and phacoemulsification, with better IOP control seen after phacoemulsification when a sutureless scleral tunnel was used (15). Use of an anterior chamber maintainer instead of viscoelastic substance for lens implantation has been associated with a lower IOP on the first postoperative day (20). Although the pressure can usually be brought under control within the first few postoperative days, patients with advanced glaucomatous damage before surgery may have additional, irreversible loss of vision during this time. Therefore, moderate to advanced glaucomatous optic atrophy and visual field loss may argue against cataract surgery alone, despite the preoperative level of IOP, although the risk may be less with phacoemulsification techniques and thorough removal of viscoelastic material from the eye. Conversely, one study found that an IOP spike greater than 30 mm Hg was almost three times as common in eyes that had a combined procedure as in eyes that had phacoemulsification alone (21).
Several studies have also looked at the IOP course in the intermediate and late postoperative periods after cataract surgery in patients with preexisting glaucoma. In general, the extracapsular techniques with posterior chamber IOLs were better tolerated than intracapsular procedures were (11), although glaucoma control postoperatively can be a problem with either technique. During the first 2 to 4 months after ECCE surgery, many glaucoma patients will have pressures above the preoperative baseline, whereas the IOP in others may be unchanged or even improved (22–24). Patients with preexisting COAG were found to have a small reduction in mean IOP and require use of fewer medications for up to 5 years after ECCE surgery (25–28). A similar trend has been seen in patients with glaucoma after phacoemulsification and IOL implantation, patients with exfoliation, and patients without glaucoma (29–35). The reasons for IOP lowering after phacoemulsification are unclear, but one proposed hypothesis involves an induction of a potential stress response in the trabecular meshwork by the ultrasound (36). Anterior chamber depth increased after cataract extraction with posterior chamber IOL implantation in patients with angle-closure glaucoma and COAG, and IOP was well controlled in most cases (37,38). However, this trend generally reverses with time (28,39). Cataract surgery alone should not be relied on as a means of treating uncontrolled glaucoma. However, as stated, when the IOP is well controlled in the presence of mild glaucomatous damage, cataract surgery alone, especially small-incision phacoemulsification with posterior chamber IOL implantation, is often a reasonable choice.
Filtering Surgery Alone
When the glaucoma is uncontrolled despite maximum tolerable medical therapy and laser trabeculoplasty, the surgical procedure of choice is the one that has the greatest chance of providing immediate and long-term IOP control. In most cases, this is a filtering operation performed alone. In some patients, eliminating the need for IOP-lowering therapy postoperatively may improve quality of life and vision enough to delay the need for cataract surgery. In other patients, the cataract can be removed 4 to 6 months later, after the filtering bleb is well established, as the second part of a two-stage approach. In one study, patients who underwent the two-stage procedure had a greater percentage of long-term IOP reduction than those who had cataract surgery alone or a combined cataract–glaucoma operation (14). Other studies have found no difference in success rates between two-stage procedures and combined phacoemulsification with trabeculectomy (40,41). In a study of 21 patients undergoing ECCE with posterior chamber IOL implantation in eyes with established filtering blebs followed up for a minimum of 2 years, the IOP increased by an average of 3.5 mm Hg, with six eyes requiring resumption of medical therapy and two requiring repeated filtering surgery (42).
Temporal clear corneal phacoemulsification did not cause a significant difference in IOP control in patients with filtering blebs after 1 year of follow-up in one study (43). In another study, phacoemulsification through a superior clear corneal incision in eyes with previous trabeculectomy increased the IOP within 1 year, but at 2 years, there was no significant difference from baseline in IOP control (44). Retrospective studies have shown that in patients with glaucoma who had trabeculectomy and subsequent cataract surgery, the IOP appeared to be better controlled by phacoemulsification than by ECCE (45,46). However, the bleb is still likely to become smaller and the IOP is likely to increase even after phacoemulsification, especially if the preoperative IOP is greater than 10 mm Hg, the iris is manipulated intraoperatively, or the patient is younger than 50 years (47,48). The IOP usually increases after phacoemulsification in eyes with preexisting hypotony (49), but resolution of the hypotony is unpredictable (48).
Combined Cataract Extraction–Glaucoma Surgery
Between the two extremes already noted—that is, the patients whose glaucoma is well controlled and those whose glaucoma is uncontrolled and poses an immediate threat to vision—there is a third group of patients with borderline glaucoma status and visually significant cataracts. For these patients, a combined procedure may be indicated. A combined approach might be preferred in the following scenarios: (a) glaucoma under borderline control despite maximum tolerable medical therapy and laser trabeculoplasty; (b) adequate IOP control, but significant drug-induced side effects; (c) adequate IOP control on well-tolerated medical therapy, but advanced glaucomatous optic atrophy; or (d) uncontrolled glaucoma, but an urgent need to restore vision or when two operations are infeasible.
The rationale for a combined procedure, as opposed to cataract surgery alone, in eyes with good IOP control but advanced damage, is the risk of a transient pressure rise in the early postoperative period. Even if laser trabeculoplasty has achieved good IOP control, it may still be necessary to combine glaucoma surgery with the cataract extraction, because a good response to laser therapy before cataract surgery does not guarantee postoperative pressure control (50). Studies have shown that the early postoperative pressure rise is significantly less after a combined procedure than after cataract extraction alone (13,14), and this was probably the primary benefit of the combined surgery during the era of ECCE surgery, when long-term glaucoma control after combined procedures was less predictable. However, with the advent of small-incision cataract surgery and the adjunctive use of antimetabolites with the filtering surgery (discussed later in this chapter), the long-term results of combined procedures have improved and the relative indications for this surgical option have expanded (51,52). Nevertheless, there is still a role for each of the three basic surgical options, the selection of which depends not only on the status of the individual patient but also on the results that each surgeon experiences with the various approaches.
Cataract extraction by phacoemulsification or ECCE, combined with IOL implantation and trabeculotomy, has been found to be a safe and effective treatment for patients with coexisting glaucoma and cataract (53,54). However, most studies find that the use of the smaller incision with phacotrabeculectomy has a higher success rate and more rapid visual recovery (Fig. 42.1). Several retrospective studies have found that the postoperative complication rate and IOP were lower when trabeculectomy was combined with phacoemulsification than with ECCE after 1 to 2 years of follow-up (55,56), and that ECCE may be a risk factor for unsatisfactory late IOP control and filtering bleb appearance. The frequency of fibrin formation and the incidence of an IOP spike of more than 25 mm Hg were lower in one study after the phacoemulsification than after the ECCE (57). More frequent IOL dislocation has been found when trabeculectomy was combined with ECCE than when it was combined with phacoemulsification (58).
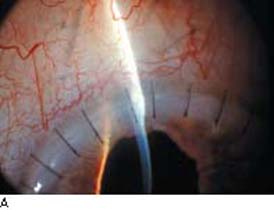
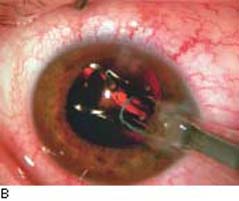
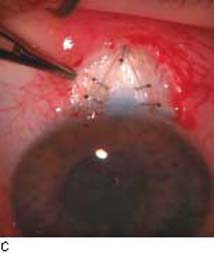
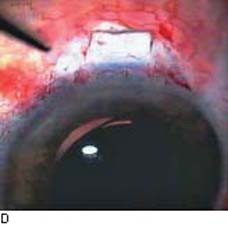
Figure 42.1 The anatomic advantage of small-incision cataract surgery for the patient with glaucoma. A: Long-term bleb function with a large cataract incision is difficult to achieve with ECCE– trabeculectomy or trabeculectomy followed later by ECCE. Inflammation, bleeding, and long-term wound healing stimulate fibroblasts, increasing the likelihood of bleb failure. B,C: Two-site phacotrabeculectomy has the advantage of modern-day small-incision cataract surgery combined with separate- site trabeculectomy. A smaller incision size results in less inflammation and cataract wound healing that is largely confined to the temporal area. Visual rehabilitation with phacoemulsification and foldable IOL is also faster. The likelihood of long-term filtration is greater with phacotrabeculectomy. D: Singlesite phacotrabeculectomy is another option. The lens extraction and the trabeculectomy are performed through the same small limbal incision. (From Fellman RL, Starita RJ, Godfrey DG, et al. Cataract extraction in patients with glaucoma. In: Tasman W, Jaeger EA, eds. Duane’s Clinical Ophthalmology. Vol 6. Philadelphia:Lippincott Williams & Wilkins; 2008:chap 16.)
Small-incision cataract surgery can be readily combined with trabeculectomy in patients with COAG (59–62). Phacoemulsification and posterior chamber IOL implantation, combined with trabeculectomy, is usually associated with a significant improvement in visual acuity, and with lowering of the IOP and the number of glaucoma medications (63). A retrospective analysis of phacoemulsification with posterior chamber IOL implant, combined with mitomycin C–augmented trabeculectomy with fornix-based conjunctival flaps, has shown that the filtering blebs were large, diffuse, and noncystic, achieving good control of IOP and improvement of visual acuity (64). A meta-analysis of techniques found that two-site surgery had better outcomes compared with single-site surgery. However, combined cataract and trabeculectomy did not perform as well as trabeculectomy alone did (65).
TECHNIQUES
Cataract Surgery in Eyes with Glaucoma
Miotic Pupil
In some cases, the cataract operation can be performed in the surgeon’s usual manner, with no special measures for the coexisting glaucoma. A previously common problem with cataract surgery in the glaucomatous eye, although one that is less common today, is the irreversible miosis from chronic miotic therapy. This became more important with the advent of phacoemulsification, in which adequate pupillary dilatation is needed to perform the surgery safely and effectively. A wide variety of techniques have been described to surgically enlarge the pupil. One approach is to make a sector iridotomy above, often with two inferior sphincterotomies (66), or multiple sphincterotomies and a peripheral iridectomy (67). If a sector iridotomy is made, some surgeons will elect to close it with sutures after implanting the lens (68,69), although it can be left open if the lens haptics are rotated horizontally away from the iridotomy. One study compared patients with sutured and unsutured sector iridotomies and found no difference in glare sensitivity (70). Sector iridectomies and sphincterotomies are less commonly used since the advent of more modern techniques to manage the small pupil (described later).
Several iris retractors have been developed to mechanically enlarge a miotic pupil (71–74). One of these instruments is the three- or four-point Beehler pupil dilator, which has two or three extendable “microfingers” through 2.5- to 3.0-mm incisions and can dilate a 2- to 3-mm pupil to approximately 6 to 7 mm. Flexible nylon hooks and the Malyugin ring are also useful to dilate and control a small pupil during cataract surgery (Fig. 42.2) (72,75–77). Other techniques include mechanical stretching of the pupil, various iris suture techniques, a maneuver of tucking the iris pillars of a sector iridectomy, and a pupil-expanding ring (74,78–82). It has also been suggested that phacoemulsification can be performed through a pupil of 4 mm or more if the capsulorrhexis is intact and the nucleus is fractured into small segments in the capsule (83), although success depends on the skill of the surgeon. In addition to stretching the pupil, the surgeon can insert the iris hooks into the capsular bag under the anterior capsule, after performing capsulorrhexis to stabilize the lens capsule in eyes with weak or damaged zonules (e.g., exfoliation syndrome) (84–87). Pupil stretch during phacoemulsification appears to have no negative effect on best-corrected visual acuity, IOP, inflammation, or other potential complications (88).
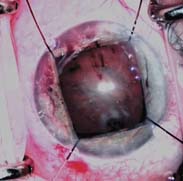
Figure 42.2 Four flexible iris retractors inserted through clear cornea stab incisions are used to dilate a chronically miotic pupil during combined cataract and glaucoma surgery.
Viscoelastic Substances
Viscoelastic substances, such as hyaluronic acid, should be used with caution in eyes with glaucoma. They are especially useful during the anterior capsulotomy, not only for maintaining a deep anterior chamber and protecting corneal endothelium but also for providing additional pupillary dilatation. However, viscoelastic substances increase the risk for early postoperative IOP rise and should be carefully removed at the end of the procedure. There were no significant differences in postoperative IOP spikes in one study when Healon 5, Healon, and Healon GV were used, although viscoelastic substances with lower viscosity appear to cause less elevation in IOP (89).
Capsulorrhexis Size
Making a capsulorrhexis diameter smaller than 5 to 6 mm prevents dislocation of the IOL from the capsular bag into anterior chamber and often eliminates the need for postoperative pupillary constriction. However, if pupillary constriction is needed after lens implantation, intracameral carbachol may be preferable to acetylcholine, because the former has been associated with better early postoperative pressure control (89). In another study, use of a combination of intraoperative acetylcholine and postoperative acetazolamide prevented an acute IOP rise more effectively than use of either agent alone did (90). It has been reported that a flap of anterior lens capsule can be included in the trabeculectomy site to facilitate filtration in combined trabeculectomy with ECCE and posterior chamber IOL implantation (91).
Intraocular Lens Selection
Selection of the proper IOL is also important in eyes with glaucoma. Posterior chamber silicone, polymethylmethacrylate, and acrylic lenses appear to be well tolerated (92), although one study found higher postoperative IOP with the acrylic IOLs than with the silicone lenses (93). Anterior chamber IOLs should, in most cases, be avoided in glaucomatous eyes. However, when loss of capsular support precludes the standard implantation of a posterior chamber IOL, the surgeon usually must decide between a sutured posterior chamber IOL and an anterior chamber IOL. Several techniques have been described for the former option (94–99), most of which use the basic principle of passing two 10-0 Prolene sutures attached to the lens haptics through the ciliary sulcus and sclera, and securing them beneath conjunctival and partial-thickness scleral flaps. These can all be difficult techniques, however, especially if they are not performed frequently, and it has been reported that the much easier procedure of implanting a semiflexible, one-piece, open-loop anterior chamber IOL is associated with reasonable long-term IOP control in most glaucomatous eyes (100); however, the tendency toward increased IOP in eyes with an anterior chamber IOL has also been observed (101,102).
Placing of releasable sutures on the scleral flap has been advocated for the combined procedure (103).
Cataract Extraction after Filtering Surgery
When extraction of the cataract becomes necessary in an eye with a functioning filtering bleb, the cataract incision should be positioned to maximize bleb survival. Phacoemulsification typically has less effect on the postoperative IOP elevation than ECCE does, although both approaches can be associated with an increase in IOP (45,104,105). Intraoperative complications during cataract surgery, especially vitreous loss, have been associated with bleb failure (104). Phacoemulsification with a foldable posterior chamber IOL through a clear corneal incision with or without a corneal suture has become a popular approach for cataract surgery in eyes with an established filtering bleb (Fig. 42.3). Most surgeons prefer to use a temporal corneal incision for phacoemulsification (106), although a clear corneal incision elsewhere, depending on the location of the filtering bleb, may be used. These basic methods generally preserve function of the filtering bleb comparably, although most eyes will have a slightly higher IOP postoperatively and many will require more glaucoma medication (44,107,108). As would be expected, eyes with a well-controlled IOP after trabeculectomy appear to have a better prognosis after cataract surgery (109).
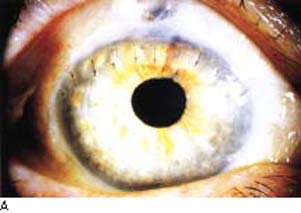
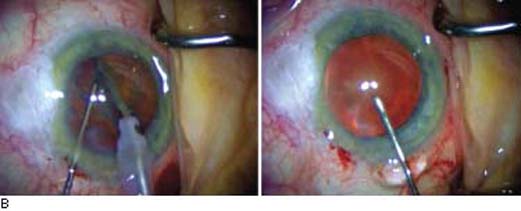
Figure 42.3 A: Slitlamp view of an eye with functioning glaucomafiltering bleb in which extracapsular cataract extraction and posterior chamber lens implantation were performed through a clear corneal incision to preserve the preexisting bleb. B: Intraoperative view of cataract surgery performed by using a temporal clear corneal incision in a patient with a preexisting bleb. Main wound and paracentesis wounds avoid the area of the bleb.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


