RELEVANT ANATOMY AND PATHOPHYSIOLOGY
Lens Overview (FIG. 4.1.2)
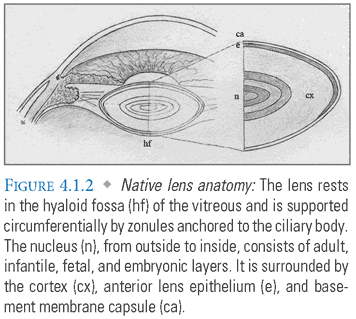
Origin: The lens is formed by embryologic invagination of the surface ectoderm.
Attachments: The equator of the lens capsule is suspended by fibrous zonules that radiate from the ciliary body.
Components:
- Capsule: An acellular, elastic basement membrane that contains the cortex and nucleus of the lens. Capsular integrity is essential for maintaining the lens contents in a relative state of dehydration. Traumatic penetration of the capsule will result in lens swelling and opacity. The anterior capsule is significantly thicker than the posterior capsule.
- Epithelium: A single anterior layer of cells that regulate lens homeostasis and differentiate into new lens fibers.
- Fibers: Thin, densely packed cells that account for most of the lens volume. The newest fibers are found in the cortex, whereas the oldest are contained within the nucleus. When mature, lens fibers do not contain nuclei or organelles.
Function:
- The lens provides between 10 and 20 diopters (D) of refraction, depending on the accommodative state, and helps focus light onto the retina. The curvature of the cornea and its interface with air supplies the majority of refractive power.
- It also protects the retina from UV radiation.
Growth:
- The lens is the only internal organ that grows throughout life. Increasing age predisposes one to angle closure glaucoma due to crowding of the anterior segment structures.
- It is nourished by aqueous diffusion through the lens capsule.
Age-related changes:
- Water content, metabolic activity, antioxidant synthesis, and elasticity decrease with age.
- The proportion of insoluble to soluble lens proteins increases with age.
- These changes predispose to cataract formation and underlie the development of presbyopia.
Key Terms
- Aphakic: The adjective used to describe an eye after extraction of the native lens without IOL placement (Noun: aphakia).
- Pseudophakic: The adjective used to describe an eye after extraction of the native lens, with IOL placement (Noun: pseudophakia).
Cataract Classification
Acquired
- Age-related (“senile”): The most common form of cataract, seen in patients 50 years of age and older with no apparent precipitating cause.
- Systemic disease-associated: Potential causes include diabetes, galactosemia, and renal insufficiency, which can cause osmotic swelling of the lens.
- Secondary: Due to primary intraocular pathology, such as uveitis.
- Postoperative: Common after vitrectomy with silicone oil or gas tamponade.
- Traumatic: Due to ocular contusion, capsule penetration, electrical injuries, or ionizing radiation.
- Toxic: Caused by exposure to drugs or toxic agents, such as corticosteroids, anticholinergics, phenothiazines, and substances in tobacco smoke.
Congenital
- May be idiopathic, hereditary, or due to transplacental infections, such as rubella, mumps, and hepatitis (remember TORCHES which refers to congenital infections such as TOxoplasmosis, Rubella, Cytomegalovirus, HErpes symplex, and Syphilis).
Cataract Morphology
- Nuclear: Accumulation of altered lens proteins causes gradual development of a central, yellow-to-brown cataract (nuclear sclerosis). The nucleus becomes denser, resulting in an increased index of refraction. In presbyopic patients, this produces lenticular myopia or “second sight.” Individuals previously dependent on reading glasses may become more comfortable without them.
- Cortical: Degeneration of the peripheral lens fibers, coupled with the development of fluid-filled vacuoles, causes early loss of VA and problems with glare. Cortical cataracts may progress to form white “intumescent” mature cataracts.
- Anterior subcapsular: A plaque of opaque connective tissue is synthesized by the lens epithelial cells. This may occur in response to anterior segment inflammation.
- Posterior subcapsular: Lens epithelial cells, normally found anterior to the equator, migrate posteriorly under the influence of various stimuli. Here they form misshapen, globular fibers called “bladder cells.” These cells accumulate, causing glare and rapid onset of visual loss despite occupying only a small volume of the lens. This form of cataract frequently develops with exposure to corticosteroids or radiation.
- Mature: This is the common end-stage of all forms of cataract; VA is generally reduced to Hand Motion or Light Perception.
- Hypermature: Liquefaction of the cortex permits leakage of lens proteins, resulting in a flaccid capsular bag. Macrophages ingest these proteins and may obstruct the trabecular meshwork, elevating intraocular pressure (phacolytic glaucoma). Emergent cataract extraction is indicated to preserve the retinal nerve fiber layer.
PREOPERATIVE SCREENING
- Exclude other potential causes of decreased vision with a careful exam of the anterior chamber and fundus. Test for the presence of a relative afferent pupillary defect (APD), which could indicate damage to the retina or optic nerve. Be sure to rule out refractive error, corneal disease, glaucoma, macular degeneration, optic nerve atrophy, and so forth.
- Perform biometry to calculate the ideal refractive power of the intraocular lens (see Appendix: Intraocular Lens Calculation for details).
- Use A-scan ultrasound to measure the axial length, anterior chamber depth, and lens thickness.
- Use keratometry to evaluate the corneal curvature and refractive power.
- Use B-scan ultrasound to assess the posterior segment if a mature cataract prevents adequate visualization of the fundus by routine ophthalmoscopy.
ANESTHESIA
Retrobulbar, peribulbar, or topical anesthesia is sufficient for most patients (see Appendix: Local Anesthesia in Ophthalmology for details).
- Retrobulbar block: Injection of local anesthetic inside the muscle cone produces reliable, rapid-onset anesthesia and akinesia, but has the highest rate of complications (e.g., globe perforation, optic nerve damage, and retrobulbar hemorrhage).
- Peribulbar block: A higher volume of anesthesia is injected outside of the muscle cone, resulting in a slower onset of anesthesia but minimizing the risk of optic nerve damage.
- Topical anesthesia: Has a rapid onset of action with few complications but does not provide akinesia, resulting in a more technically challenging case.
General anesthesia is required for children and may be necessary in rare circumstances for anxious or uncooperative adults.
PROCEDURE
SEE TABLES 4.1.1 Rapid Review of Steps and 4.1.2 Surgical Pearls, and WEB TABLES 4.1.1 to 4.1.4 for equipment and medication lists. For information on phacoemulsification techniques and settings, please refer to Appendix: Phacoemulsification Principles and Techniques.


The exact sequence varies depending on surgeon preference and the requirements of a particular case. Outlined below are the general steps that you are likely to encounter in cataract phacoemulsification with PCIOL placement.
- Preoperative Medication: Pupillary dilation permits exposure of the anterior lens capsule, and is usually achieved by combining a topical sympathomimetic (e.g., phenylephrine) with a parasympatholytic (e.g., tropicamide). Non-steroidal anti-inflammatory drugs (NSAIDs) can be added to decrease operative inflammation and help to inhibit miosis through blockage of PGE2 production. Antibiotic prophylaxis is given to cover conjunctival and skin flora that can cause endophthalmitis.
- Paracentesis Incision/Viscoelastic Injection: A 1-mm paracentesis port is made in the peripheral cornea with a fine blade, permitting injection of viscoelastic media (FIG. 4.1.3). This incision should be oriented for easy access with the surgeon’s nondominant hand. Viscoelastics are thick, jelly-like polymers that stabilize the anterior chamber while protecting the corneal endothelium and posterior lens capsule during cataract emulsification.
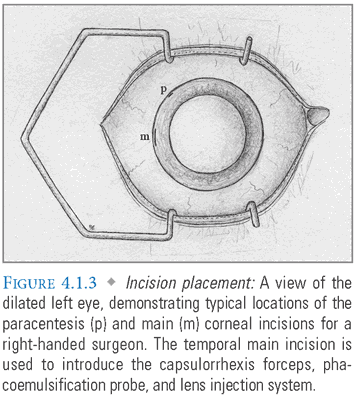
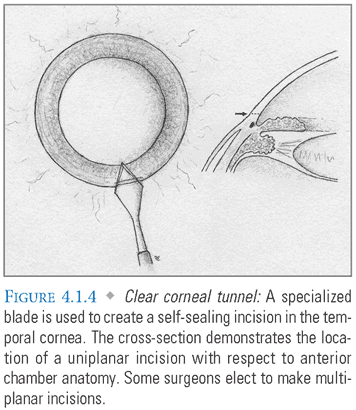
- Main Incision: A 2.2- to 2.8-mm incision in the temporal limbus or clear cornea is then made with a specialized blade that forms a self-sealing tunnel into the anterior chamber (FIG. 4.1.4). Some surgeons manipulate the blade to create a multiplanar tunnel, which may reduce the likelihood of wound leakage, but is not essential for a sutureless closure. Wider incisions should be made more peripherally to permit astigmatic neutrality and may require sutures for adequate closure. Although clear corneal incisions are popular, some surgeons prefer to create a scleral tunnel, initiating the incision approximately 1 mm posterior to the limbus. Some also place their incisions at specific clock-hour locations to achieve a predictable effect on preoperative corneal astigmatism.
- Vital Dye Injection: A good red reflex aids visualization of the capsulorrhexis. Since dense cataracts may obscure this reflex, a vital dye such as trypan blue can be injected to stain the lens capsule and help differentiate it from underlying lens material.
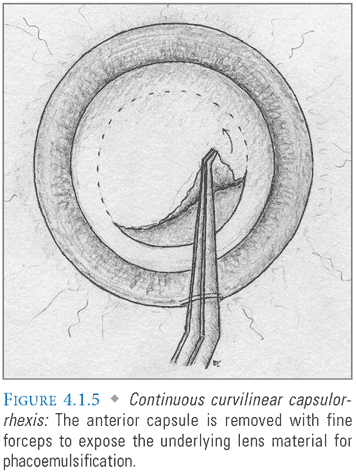
- Capsulorrhexis: A cystotome or pair of fine forceps is introduced through the main incision to make a controlled, circular tear in the anterior lens capsule, permitting access to the underlying cortex and nucleus (FIG. 4.1.5). To preserve capsular integrity and ensure IOL stability, it is essential to prevent the advancing edge of the capsulorrhexis from straying too far peripherally. This can be avoided by perpendicular application of force, and by frequent regrasping of the free capsular edge. In the event that it does occur, the capsulorrhexis can be rescued by redirecting with a series of short “can opener” incisions.
- Hydrodissection: Balanced salt solution (BSS) is injected between the posterior lens capsule and the cortex using a narrow cannula. The cortex is circumferentially adherent to the capsule at the lens equator, and depression of the nucleus will cause a wave of solution to dissect through these connections. The lens can then be rotated freely within the capsular bag, permitting easier fractioning and removal.
- Hydrodelineation: Some surgeons also inject fluid directly into the nucleus to separate the softer epinucleus from the harder endonucleus, facilitating subsequent phacoemulsification.
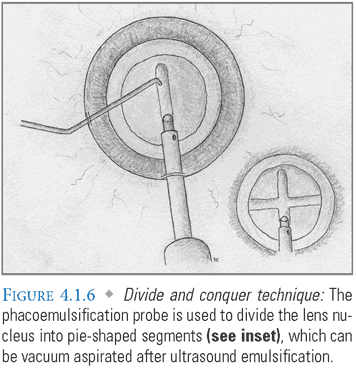
- Phacoemulsification: Numerous techniques have been developed to disassemble the nuclear component of the cataract for removal. “Chopping” is commonly employed for routine cases. The phaco tip is used to crack the nucleus into several pie-shaped segments with the assistance of a second instrument introduced through the paracentesis port. In the “divide and conquer” technique, the phaco handpiece is used to create two perpendicular grooves in a nuclear cataract. Instruments are subsequently used to crack it into quadrants (FIG. 4.1.6). After disassembly, the remaining lens fragments are emulsified and vacuum aspirated. See Appendix: Phacoemulsification Techniques for greater detail.
- Cortex Removal: The residual cortex is then removed with an irrigating and aspirating handpiece that does not require ultrasonic energy.
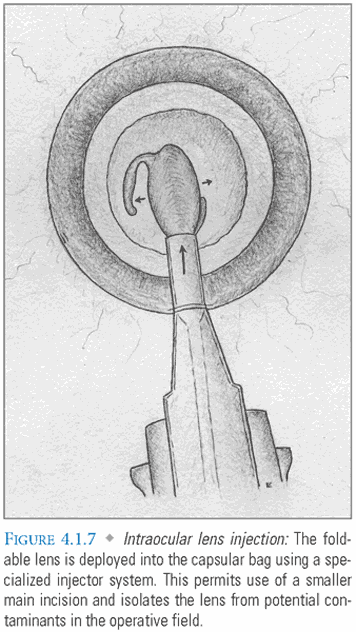
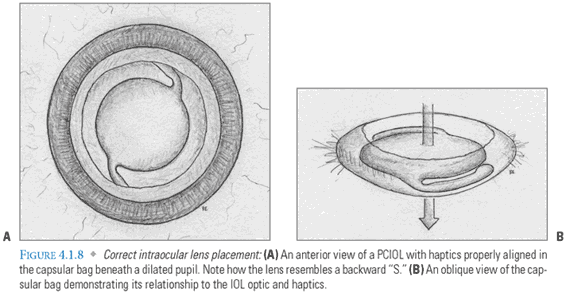
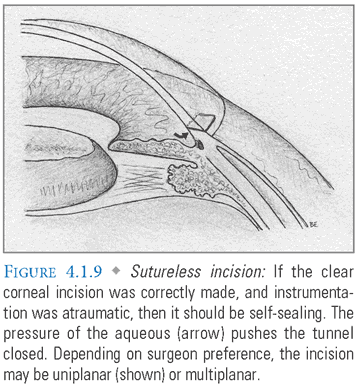
- Lens Implantation: The empty capsular bag is inflated with viscoelastic prior to insertion of a foldable IOL. This lens is implanted either with forceps or with a specialized injection system (FIG. 4.1.7). The advantage of an injection system is that it uses a smaller incision and isolates the lens from potential contaminants in the surgical field. After the lens unfolds and the haptics are properly positioned (FIG. 4.1.8A-B), viscoelastic is evacuated and replaced with BSS, which resembles aqueous in composition and pH. The main incision should be self-sealing if it was properly constructed and instrument insertion during the case was atraumatic (FIG. 4.1.9). Some surgeons “hydrate” the incision with the goal of achieving tighter closure. This entails using an instrument to induce local stromal edema within the walls of the tunnel.
- Postoperative Medication: Most surgeons instill antibiotic and steroid drops at the completion of surgery.
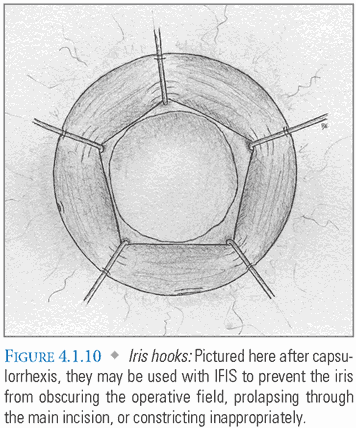
Special Considerations: Intraoperative floppy iris syndrome (IFIS) is linked to tamsulosin and other selective α-1 adrenergic receptor antagonists used to treat benign prostatic hyperplasia (BPH). These medications affect the pupillary dilator, causing the iris to billow with anterior chamber irrigation, prolapse through incisions, and progressively constrict during surgery. Routine premedication may be insufficient to produce adequate mydriasis. In such cases, IFIS can be addressed with intracameral phenylephrine, iris hook placement (FIG. 4.1.10), or a combination of these techniques. Some surgeons prefer the Malyugin ring system to iris hooks, as it does not require the creation of multiple paracentesis incisions (FIG. 4.1.11).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


