Surgery of the Anterior Chamber Angle and Iris
In this chapter, we consider the laser and incisional operations that are designed to reduce the intraocular pressure (IOP) through increased aqueous outflow by treating specific structures of the anterior chamber angle and the iris. (Filtration procedures and glaucoma drainage-device surgery, which involve not only the anterior chamber angle but also limbal and external ocular tissues, are considered in Chapters 38 and 39, respectively; procedures for children are discussed in Chapter 40.)
LASER TRABECULOPLASTY
Historical Background
In 1961, Zweng and Flocks (1) introduced the concept of applying light energy to the anterior chamber angle for the treatment of glaucoma. Using the xenon-arc photocoagulator of Meyer-Schwickerath (discussed later in this chapter), they selectively coagulated the filtration angles of cats, dogs, and monkeys and reported subsequent lowering of the IOP. Histopathologic examination of the treated tissue revealed fragmentation of the trabecular lamellae, atrophy of ciliary muscle, and destruction of ciliary processes. Little more was said about this technique, however, until more than a decade later, when several investigators revived the concept by using the light energy of the laser. Yet another decade of investigative work would elapse before the operation would achieve widespread clinical popularity.
In the early 1970s, reports began to appear from several parts of the world, most notably from Krasnov (2) in Russia, Hager (3) in Germany, Demailly and associates (4) in France, and Worthen and Wickham (5) in the United States, regarding attempts to improve aqueous outflow by creating holes in the trabecular meshwork with laser energy. Although trabecular perforations were achieved, they eventually closed in most cases due to fibrosis, and IOP reduction was usually temporary. The value of laser treatment to the trabecular meshwork came under further question when, in 1975, Gaasterland and Kupfer (6) reported that experimental glaucoma could be produced by applying argon laser energy to the meshwork of rhesus monkeys. The following year, however, Ticho and Zauberman (7) noted that long-term reduction in IOP occurred in some patients despite the lack of permanent trabecular openings. This led to a new concept in laser trabecular therapy in which lower energy levels were used to photocoagulate, rather than to penetrate, portions of the meshwork. In 1979, Wise and Witter (8) described the first successful protocol of what has become known as laser trabeculoplasty. Their preliminary work was corroborated in 1981 (9–11).
In the subsequent years, many different energy sources producing different wavelengths of laser light, such as krypton (red [647.1 nm] or yellow [568.2 nm] wavelengths), Nd:YAG (neodymium:yttrium–aluminum–garnet) (continuous-wave [1064 nm] and frequency-double Q-switched [532 nm]), and diode (840 nm), have been studied for laser trabeculoplasty (12–20). At the time of publication, the only other laser that has attained popularity is the frequency-doubled Nd:YAG laser, otherwise known as selective laser trabeculoplasty (SLT).
Theories of Mechanism
Argon Laser Trabeculoplasty
Tonographic studies indicate that argon laser trabeculoplasty (ALT) reduces IOP by improving the facility of outflow (12, 21–24), while showing no significant influence on aqueous production on fluorophotometric investigations (23,25,26). Although fluorescein leakage into the anterior chamber is seen during the first week after trabeculoplasty, suggesting a breakdown in the blood–aqueous barrier, it is gone within 1 month and does not seem to be a factor in the long-term effect of this procedure (27).
The mechanism of improved aqueous outflow facility by ALT is uncertain. Wise and Witter (8) originally postulated that the thermal energy produced by pigment absorption of laser light caused shrinkage of collagen in the trabecular lamellae. They believed that the subsequent shortening of the treated meshwork might enlarge existing spaces between two treatment sites or expand the Schlemm canal by pulling the meshwork centrally. Laboratory studies have provided partial support for this theory but have also suggested alternative or additional mechanisms of action.
Light and electron microscopic and immunohistochemical evaluations of trabecular meshwork from normal and glaucomatous human eyes, obtained hours to weeks after ALT, revealed disruption of trabecular beams, fibrinous material, and necrosis of occasional cells, followed by shrinkage of the collagenous components of the meshwork and accumulation of fibronectin in the aqueous drainage channels (28–33). Surviving endothelial cells near the laser lesions showed phagocytic and migratory activity (29,30). Specimens obtained several months after therapy had partial or total occlusion of intertrabecular spaces by a monocellular layer (28,30,31). These observations were thought to support the theories of heat-induced shrinkage of collagen in the trabecular lamellae with possible stretching of the meshwork between two treatment sites and fibronectin-mediated attachment of trabecular beams supporting an adhesive tightening of the trabecular components (31).
Studies with monkeys have provided similar observations to those noted in humans, with some additional insight into the mechanism of ALT. Within the first few hours, there is disruption of the trabecular beams and coagulative necrosis with accumulation of debris in the juxtacanalicular region (34). As with human eyes, surviving trabecular endothelial cells are noted to have increased phagocytic activity with removal of tissue debris and increased cell division (34,35). By 1 month, the treated regions are flat with collapsed beams and are covered with an endothelial layer (36). The latter is more likely to occur when the laser energy is applied to the anterior portion of the trabecular meshwork (37). Perfusion with ferritin shows lack of flow through the treated meshwork, with diversion of flow through the adjacent nonlasered meshwork, which becomes structurally altered to compensate for the overload of flow (38). It has also been suggested that concomitant collagen degeneration and loss of trabecular cells may widen the intertrabecular spaces with improved outflow (39). However, light and electron microscopic studies of the trabecular meshwork and the inner wall of the Schlemm canal 3 to 17 months after 360-degree trabeculoplasty in monkeys revealed no significant difference from untreated eyes (40). Whether the human eye has similar reparative capacity is unclear, but this and other studies suggest that alternative or additional mechanisms to the mechanical theory must account for the long-term benefit of laser trabeculoplasty.
Studies of human autopsy eyes treated with ALT revealed a significant reduction in the trabecular cell density and an increase in radioactive sulfate incorporation into the extracellular matrix of laser-treated eyes (41). The latter findings have also been reported with human trabecular tissue treated with ALT before trabeculectomy and subsequently studied with radioactive leucine (31), and in cat eyes that were studied in vivo with radioactive thymidine following trabeculoplasty (42,43). Studies with a human corneoscleral organ culture system indicate that ALT causes an early trabecular endothelial cell division in the anterior meshwork, with migration of the new cells to repopulate the burn sites over the next few weeks (44,45).
It has been postulated that ALT eliminates some trabecular cells, which may stimulate the remaining cells to produce a different composition of extracellular matrix with improved outflow properties (41–43). This hypothesis is further supported by demonstration of induction of matrix metalloproteinases in response to laser trabeculoplasty (46–48). The matrix metalloproteinases are the enzymes that normally break down the extracellular matrix to maintain normal turnover of the trabecular meshwork (49). Manipulation of activity of these enzymes has been demonstrated in perfused human anterior segment organ culture to increase outflow facility with increasing matrix metalloproteinases (50). Evaluation of two members of the matrix metalloproteinases family, stromelysin and gelatinase B, after ALT of anterior segment organ cultures also supports the hypothesis that extracellular matrix turnover is important in the regulation of aqueous humor outflow. An increase of stromelysin expression has been demonstrated in the juxtacanalicular region of the meshwork in response to laser trabeculoplasty (47). This would be expected to degrade trabecular proteoglycans, a presumed source of outflow resistance in the juxtacanalicular meshwork. If reduced juxtacanalicular extracellular matrix turnover is responsible for the reduction in aqueous humor outflow, an increase in stromelysin in this specific area of the meshwork should increase the outflow (47).
Additional studies have been designed to identify factors that mediate the matrix metalloproteinases response to ALT. Matrix metalloproteinases expression was increased by adding recombinant interleukin-1α in human anterior segment organ cultures and tumor necrosis factor-α in porcine trabecular meshwork (50,51). Expression of stromelysin was partially blocked by either interleukin-1 receptor antagonist or tumor necrosis factor-α–blocking antibodies (48).
Although the precise mechanism of ALT still remains only partially understood, an initial mechanical injury appears to trigger activation of unique signaling pathways resulting in cellular response and tissue remodeling, leading to an improved outflow (52).
Selective Laser Trabeculoplasty
In 1995, Latina and Park reported that the energy of a Q-switched, frequency-doubled Nd:YAG laser would preferentially be absorbed by pigmented trabecular meshwork cells, in culture (53), called an SLT (54,55). The laser selectively targets pigmented trabecular meshwork cells without causing structural damage to nonpigmented cells. Experimental study on the trabecular meshwork from human autopsy eyes after SLT revealed no coagulative damage or disruption of the corneoscleral or uveal trabecular beams (32). The only evidence of laser tissue interaction with SLT was cracking of intracytoplasmic pigment granules and disruption of trabecular endothelial cells, suggesting that it may potentially be a repeatable procedure (32). Evaluation of the trabecular meshwork after ALT revealed crater formation in the uveal meshwork at the junction of the pigmented and nonpigmented trabecular meshwork, with coagulative damage at the base and along the edge of craters, disruption of the collagen beams, fibrinous exudate, lysis of endothelial cells, and nuclear and cytoplasmic debris (32). However, in another study, the mechanical damage observed after low-power ALT and SLT was similar, with both lasers producing disruption of trabecular beams, cellular debris, and fragmentation of endothelium (33). The similarity of changes in the trabecular meshwork produced by both lasers may explain their similar IOP-lowering responses (33).
The impact of 360-degree SLT on free oxygen radicals and antioxidant enzymes of the aqueous humor has been evaluated in rabbits. Concentrations of lipid peroxide in the aqueous humor of the treated eyes were significantly higher than those in the untreated eyes until the 7th day (56). Glutathione S-transferase levels were significantly decreased between 12 hours and 7 days after the trabeculoplasty, suggesting that free oxygen radicals are formed in the pigmented trabecular meshwork during SLT and may be responsible for the inflammatory complications of this procedure (56).
Basic Techniques
Instruments
The original laser unit for trabeculoplasty is the continuous-wave argon laser. It has traditionally been operated in the blue-green, biochromatic wavelength spectrum (454.5 to 528.7 nm). No differences were noted in the postoperative IOP course or incidence of complications when compared with the use of green, monochromatic laser light (514.5 nm) (57). As noted in the previous chapter, however, green-only argon light may be safer for the surgeon with regard to an influence on color vision. The Q-switched Nd:YAG laser has only one wavelength setting, at 532 nm.
A contact lens with a mirror for visualization of the anterior chamber angle (gonioprism) is used in trabeculoplasty. As with all contact lenses for laser application, it should have an antireflection coating on the front surface. A standard Goldmann-type three-mirror lens, in which one mirror is inclined at 59 degrees for gonioscopy, or a single-mirror gonioscopy lens can be used (Fig. 36.1). Both, however, have the slight disadvantage of requiring rotation of the lens to view all quadrants of the anterior chamber angle. This disadvantage can be eliminated by using the Thorpe four-mirror gonioscopy lens, in which all mirrors are inclined at 62 degrees, or the Ritch trabeculoplasty laser lens, in which two mirrors are inclined at 59 degrees for viewing the inferior quadrants and two at 64 degrees for viewing the superior angle (58,59). In the latter lens, a 17-diopter (D) planoconvex button lens over two mirrors provides 1.4× magnification, reducing a 50-µm laser spot to 35 µm, which may be particularly useful, because a 50-µm spot size with most argon lasers produces a burn in excess of 70 µm (60). A double-mirror gonioscopic lens has also been developed to facilitate the visualization of the anterior chamber angle (61). The Latina lens was specifically designed for SLT and has a single mirror at a 63-degree angle; it has a 1.0× magnification to maintain the 400-µm spot size.

Figure 36.1 The Goldmann-type three-mirror lens, modified with antireflection coating, is a commonly used gonioprism for visualizing the anterior chamber angle and for use in performing laser trabeculoplasty.
Gonioscopic Considerations
Successful laser trabeculoplasty requires accurate identification and treatment of the trabecular meshwork. The surgeon must, therefore, have a detailed knowledge of the anterior chamber angle anatomy and its many variations. The basic aspects of this subject are discussed in Chapters 2 and 34; additional features that are pertinent to laser trabeculoplasty are considered here.
Two variations of the anterior chamber angle that may interfere with accurate laser application to the trabecular meshwork are (a) the degree of pigmentation and (b) the width of the chamber angle. With regard to pigmentation, some angles are so diffusely pigmented from the ciliary body band to the Schwalbe line that the exact location of the meshwork is obscured. This is usually most marked in the inferior quadrants, and a careful inspection of all quadrants before starting treatment usually discloses the functional position of the meshwork in some areas, which can then be used as a guide in locating the meshwork in the remainder of the angle. At the opposite extreme, the trabecular meshwork in some angles is so lightly pigmented that it is hard to see. In some cases, iris processes, which normally extend to the meshwork, may be a useful indicator. Identification of the ciliary body band or Schwalbe line may also help determine the relative position of the meshwork.
A narrow anterior chamber angle can lead to improper placement of the laser burns or may prohibit performing trabeculoplasty. If the peripheral iris obscures the visualization of the meshwork, a heavily pigmented Schwalbe line may be mistaken for the meshwork. Rotating the contact lens in relation to the eye, by asking the patient to look in the direction of the mirror being used, often provides a deeper view into the angle, enhancing visualization of the meshwork. Care must be taken with this maneuver, however, not to distort the size and shape of the aiming beam. If positioning of the contact lens is not sufficient to expose the meshwork, the chamber angle may be deepened by applying low-energy laser burns to the peripheral iris, a technique called iridoplasty or gonioplasty (discussed later in this chapter). If the angle is still too narrow, a laser iridotomy (also discussed later in this chapter) should be performed, and the trabeculoplasty should be done at a later date.
Original Protocol
The original protocol of Wise and Witter (8) has remained the standard approach to ALT against which variations in technique have been evaluated. A 25× magnification in the slitlamp delivery system usually provides an optimum balance between detail and field of view. Argon laser settings of 0.1-second duration exposure and 50-µm beam diameter have remained constant through most variations in protocol. One study compared durations of 0.2 to 0.1 second and found no advantage to the former (62). The most commonly used power levels range between 700 and 1500 mW, with an average of 1000 mW. A survey by the American Society of Cataract and Refractive Surgery in 1999 indicated that most general ophthalmologists use a duration of 0.1 second and a spot size of 50 µm, and that 39% of the respondents use initial power between 501 and 799 mW and 41% use 800 to 1000 mW (63). One study evaluated powers ranging from 100 to 1000 mW and found that power of more than 500 mW gave the maximum success rates (64). The power should be adjusted to produce a depigmentation spot or a small gas bubble at the treatment site (Fig. 36.2). This response is influenced by the amount of pigment in the trabecular meshwork. With a heavily pigmented meshwork, a lower power level may be sufficient, whereas lightly pigmented meshworks require higher levels. In a retrospective study, the decrease of IOP was greater in the eyes in which ALT was a primary therapy and was not influenced by the power level (65). The initial IOP response to ALT in patients with glaucoma associated with exfoliation syndrome was greater than in patients with chronic open-angle glaucoma (COAG) (66,67), although the long-term outcome was similar (66). A preoperative IOP higher than 31 mm Hg and visual field defect and light pigmentation of the trabecular meshwork were found to be predictive of ALT failure (67).
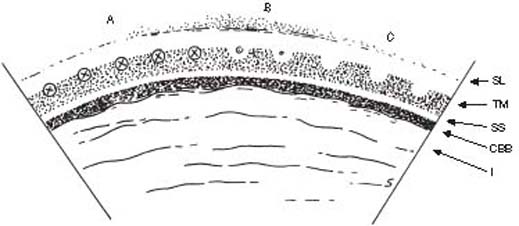
Figure 36.2 Placement of laser burns (A) along anterior portion of trabecular meshwork (TM). Desired visual result is depigmentation of the treatment site (B,C) or a small gas bubble (B). SL, Schwalbe line; SS, scleral spur; CBB, ciliary body band; I, iris.
Originally, ALT laser burns were applied onto or immediately posterior to the pigmented band of the trabecular meshwork, with approximately 100 applications evenly spaced around the full 360 degrees of the meshwork (8). Complications associated with this basic protocol, however, led to variations in technique. We consider, first, the complications and how they are managed and, then, the variations in technique that have been used to minimize the complications.
With SLT, a total of approximately 50 to 70 adjacent, nonoverlapping spots are placed over 180 degrees of the trabecular meshwork, with energy ranging from 0.5 to 1.2 mJ per pulse, set to prevent bubble formation. Typically, the power is titrated until the appearance of tiny air bubbles are released from the site of the laser burn, termed “champagne bubbles.” After the bubbles are seen, the power is slightly reduced to eliminate their appearance.
Alternative Protocols with Argon Laser
The parameter evaluated most extensively has been the total number of laser applications and the amount of trabecular meshwork treated. Applying 25 burns to 90% of the meshwork is less effective than protocols with larger amounts of treatment (68,69). However, the application of 50 burns to 180 degrees or 360 degrees has a similar effect on IOP reduction as treatment of 100 burns to 360 degrees of the meshwork (69–71). In one such study, the eyes receiving 50 applications over 180 or 360 degrees had a lower probability of requiring subsequent filtering surgery than those receiving 100 applications over 360 degrees (72). A two-stage protocol, in which treatment of the full 360-degree circumference is divided into two sessions, 1 month apart, had the same IOP reduction as the full treatment in one session (73). With the latter technique, most of the pressure reduction is achieved with the first stage of therapy, although some patients may have minimal benefit from the first stage and yet a substantial pressure reduction after the second stage (74). The main advantage of the lower number of laser applications during a single session is a reduction in the transient IOP rise in the immediate postoperative period (69–71,73–77). In one study, however, the frequency and magnitude of postlaser IOP increase were the same in groups receiving 360-degree treatment in one or two stages (78). The long-term outcome does not appear to be influenced by which quadrants are treated first. One study randomly assigned patients into initial inferior versus superior halves and found no significant difference between the two groups (72).
Another variation from the basic protocol that appears to minimize the complication of early posttreatment IOP rise is the placement of the laser applications along the anterior portion of the pigmented meshwork (Fig. 36.3) (69,71,77). An anterior placement of the laser burns also reduces the complication of peripheral anterior synechiae (79,80). It may, however, increase the potential complication of cellular proliferation from the corneal endothelium over the trabecular meshwork (37).

Figure 36.3 Gonioscopic view of patient following argon laser trabeculoplasty. Note the typical blanched lesions of the pigmented trabecular meshwork, which may persist for several days.
Complications and Postoperative Management
Transient IOP elevation in the immediate postoperative period is the most serious early complication of ALT (75,76,81–84). In most cases, the pressure rise is mild and lasts less than 24 hours, causing no long-term problems. In some patients, however, the elevation is marked and sustained and can lead to further loss of vision, especially in eyes with advanced visual field loss before trabeculoplasty. The IOP rise occurs within 2 hours after treatment in most cases, although some eyes may not develop an increase until 4 to 7 hours after therapy (82,83). Postoperative management, therefore, should include a pressure check within the first few hours after the procedure. Patients who have a significant early postoperative pressure rise or who have advanced glaucomatous damage may require an IOP check the following day. However, a pressure rise on the first postoperative day is uncommon, with only 4.2% having a rise greater than 3 mm Hg in one study, and seeing most patients in 1 to 3 weeks postoperatively is considered reasonable (85). With SLT, approximately 25% of patients had a transient IOP elevation of 5 to 6 mm Hg (54,86,87), and in one study, some patients had an IOP elevation of more than 10 mm Hg (88).
Histopathologic studies suggest that the mechanism of posttrabeculoplasty pressure rise after ALT is an inflammatory reaction, with fibrinous material and tissue debris in the meshwork (28,34,89,90). Laboratory studies in bovine eyes indicate that the trabecular meshwork can contract in response to endothelin-1, which may be a mechanism in the immediate posttrabeculoplasty IOP elevation (91,92). This hypothesis is supported by the finding of an increased concentration of endothelin-1 in the aqueous humor of rabbit eyes after ALT (93–95).
The main patient characteristic associated with the transient pressure rise is meshwork pigmentation (84). Two patients with exfoliation syndrome had a delayed IOP rise during the first postlaser month, associated with inflammatory precipitates on the trabecular meshwork (96). It should also be noted that eyes with active inflammation are at a high risk for marked IOP rise after ALT, and the operation is contraindicated in these eyes.
Iritis is a common early posttrabeculoplasty complication. In one study, by using a laser flare-cell meter, 49% of 71 eyes showed significant inflammation, which peaked 2 days after treatment (97). The inflammation was significantly more frequent in eyes with exfoliation syndrome or pigmentary glaucoma than in those with COAG. Postoperative iritis is usually mild and transient and is easily controlled with a brief postoperative course of topical corticosteroids. A typical protocol for postoperative management of ALT includes prednisolone, 1%, fluorometholone, 0.1%, or the equivalent four times daily for 5 days. Pretreatment with topical steroids or nonsteroidal anti-inflammatory agents (98–101) has been shown to reduce posttrabeculoplasty inflammation but had no effect on the postoperative IOP elevation (102,103). There is no definite consensus on the posttrabeculoplasty anti-inflammatory regimen following SLT. Various protocols, ranging from use of topical prednisolone acetate, 1%, to use of a topical nonsteroidal anti-inflammatory agent, to use of no anti-inflammatory agents, have been used. However, a greater anterior chamber reaction was seen after the SLT than after ALT in one study (104).
The formation of peripheral anterior synechiae is also a common complication of trabeculoplasty (79). These are typically small and tented, corresponding to the location of the laser applications. Alterations of corneal endothelium after ALT may include a significant increase in cell size (105), although another study showed no statistically significant changes (106). The formation of peripheral anterior synechiae after SLT is rare.
The most serious late-posttrabeculoplasty complication is, at the present time, more theoretical than real. Histopathologic studies, as previously described, show changes in the trabecular meshwork, including an endothelial layer over the inner surface (Fig. 36.4), which could eventually lead to an increase in resistance to aqueous outflow (28–30,36,37). One retrospective study evaluated ALT specimens treated with one or more ALT procedures before trabeculectomy and found that eyes treated with argon laser had an increased incidence of membrane formation in the chamber angle. Half of the specimens had a cellular and collagenous membrane covering the entire trabecular meshwork, which was more common in eyes in which more ALT procedures were performed (107). Whether these structural changes eventually make the glaucoma more difficult to control has never been proven, despite more than 30 years of experience. There is, however, a limit to the amount of laser treatment that an eye can tolerate, and the success is time limited in nearly all patients, as discussed later in this chapter. There has also been concern that laser trabeculoplasty might interfere with the success rate of subsequent filtering surgery, causing a higher rate of encapsulation in eyes with previous ALT (108), although this did not appear to be the case in another study (109).

Figure 36.4 Scanning electron microscopic view of trabeculectomy specimen from eye with failed argon laser trabeculoplasty showing endothelial growth over portions of the intertrabecular spaces (arrows).
Pharmacologic Control of Increased Pressure
Topical application of the α2-adrenergic agonist apraclonidine, 1%, at 1 hour before and immediately after laser trabeculoplasty was shown to have a marked effect on minimizing the postoperative pressure rise (110). When compared with eyes treated with pilocarpine, 4%, timolol, 0.5%, dipivefrin, 0.1%, or acetazolamide, 250 mg, each given 1 hour before and immediately after trabeculoplasty, only 3% of apraclonidine-treated eyes had IOP increases greater than 5 mm Hg, in contrast to 33%, 32%, 38%, and 39%, respectively, with the other treatments (111). A single drop of apraclonidine 15 minutes before or immediately after the laser treatment is as effective as the two doses, and apraclonidine, 0.5%, is as effective as 1% (112–115). This has now become a standard part of laser trabeculoplasty for most surgeons. So profound is the benefit of apraclonidine that treatment in two sessions of 180 degrees each may no longer be necessary to avoid the transient IOP rise. In one study, 360-degree trabeculoplasty with perioperative apraclonidine had the same early postoperative IOP course as the 180% treatment without apraclonidine (116). However, caution is advised for patients on long-term α2-adrenergic agonist therapy, in which case the apraclonidine may be less effective.
The selective α2-adrenergic agonist brimonidine, 0.5%, has been shown to effectively control the postlaser pressure rise when given either before or after the laser surgery (117,118). Brimonidine, 0.2%, has also been found to be as effective as apraclonidine, 1.0%, in preventing IOP spikes after ALT (119).
Pilocarpine, 4%, alone immediately after ALT was also shown to be effective in minimizing the IOP elevation (120). In a randomized trial, apraclonidine, 1%, was not effective in preventing the IOP spikes in patients on long-term apraclonidine (121). Pilocarpine, 4%, was only slightly less effective in patients on long-term pilocarpine therapy and was at least as effective as apraclonidine, 1%, in post-ALT IOP spike prophylaxis. Another study found that adding pilocarpine to apraclonidine therapy further reduced the incidence of postoperative pressure rise (122). Pilocarpine, therefore, can be considered as a first choice for prevention of posttrabeculoplasty IOP spike, especially in patients treated with apraclonidine (121) or possibly with other α2-adrenergic agonists.
Acetazolamide was also shown to reduce the IOP rise following ALT in one study (123), although, as previously noted, it is less effective than apraclonidine (111). As discussed previously, neither corticosteroids (98), nor the prostaglandin synthetase inhibitors indomethacin or flurbiprofen, significantly influenced the postoperative IOP (99,100,124,125). One study showed that patients receiving topical indomethacin had higher pressures after 1 month than those receiving a placebo (124). Prostaglandin synthetase inhibitors also appear to have no influence on the postoperative iritis (100,126).
Results
Short-Term Intraocular Pressure Control
Most reports show that useful IOP reduction is achieved in approximately 85% of eyes treated with ALT (8–11,22,75,127). Some eyes may have a pressure drop within the first few hours after treatment, although days or weeks are usually required to achieve the full response to ALT and SLT, with further IOP reduction rarely occurring beyond 1 month. The magnitude of the final pressure reduction averages 6 to 9 mm Hg, which is usually insufficient to allow discontinuation of all medical therapy, although the medication can occasionally be reduced or eliminated (128). One study suggested that pilocarpine may lose its effectiveness after ALT (129), and it may be advisable to re-evaluate the efficacy of any miotic treatment approximately 1 month after the laser treatment. However, a later study showed no difference between the IOP-lowering effect of pilocarpine, 1%, before and after ALT (130).
The IOP reduction after the SLT ranged from 3 to 18 mm Hg (86). Six months after 180-degree SLT, the mean IOP reduction was 4.4 mm Hg, with a success rate of 64.6%. An elevated preoperative IOP was the significant determinant for success, whereas age, sex, history of ALT, and trabecular meshwork pigmentation were not significantly related to success (87). When the trabecular meshwork was treated 360 degrees with the SLT, the IOP was reduced in all eyes by approximately 40% at 6 weeks after the treatment (88). In a prospective study, 50 eyes were treated with SLT, with the mean IOP reduction of approximately 5 mm Hg at 1, 3, 6, and 12 months (131). In another clinical trial of 10 eyes treated with SLT, the IOP was reduced only slightly less in the exfoliative glaucoma than in the COAG (132). In a randomized trial, patients with previously failed ALT had a better IOP reduction with the selective laser than with a repeated argon laser (104). IOP lowering during the first 6 months after the SLT was similar to that of ALT and appears to diminish over the first year of follow-up (133).
Factors Affecting IOP Response
Many factors influence the IOP response to ALT. Eyes with a higher pretreatment IOP tend to have a greater decrease in IOP (134), but a pretreatment IOP greater than 30 mm Hg has been associated with a higher frequency of failure (135,136), whereas eyes with pressures closer to the target IOP may obtain useful pressure reduction after trabeculoplasty (137–139).
Another significant factor influencing IOP response to ALT is the type of glaucoma. A particularly favorable response is obtained with COAG, exfoliation syndrome, and pigmentary glaucoma (21,127,134,135,140–143). Success in the latter two conditions is most likely related to the favorable influence of increased trabecular meshwork pigmentation (144). In pigmentary glaucoma, younger patients appear to have a more sustained pressure reduction than older patients with the same condition do (142,143). Some clinicians have noted that eyes with darkly pigmented trabecular meshwork are at greater risk for an immediate IOP spike following SLT (145). In these eyes, decreasing the power of the SLT is generally recommended.
Other forms of glaucoma that respond to ALT, although less well than those noted earlier, include open-angle glaucoma in aphakia or pseudophakia and angle-closure glaucoma after an iridotomy (140,146). Although eyes that have had multiple operations generally do not do well with ALT (141), those with a single failed trabeculectomy may obtain useful pressure reduction after the laser surgery (147). Other forms of glaucoma that do not respond well to ALT include glaucoma associated with uveitis, angle-recession glaucoma, and congenital or juvenile glaucoma (140,141).
Some investigators believe that young age has an unfavorable effect on the results of laser trabeculoplasty (127,136,148), although one study showed no effect of age (135). As previously noted, young patients with pigmentary glaucoma appear to do better than older patients with the same condition (142,143).
Race may influence the results of laser trabeculoplasty (149). In the Advanced Glaucoma Intervention Study (AGIS), eyes were randomly assigned to an ALT–trabeculectomy–trabeculectomy sequence or a trabeculectomy–ALT–trabeculectomy sequence. The initial report from this randomized clinical trial recommended the initial use of the ALT for all black patients (150). However, a later report from the AGIS provided only a weak suggestion that an initial trabeculoplasty delays the progression of glaucoma more effectively in black patients than in white patients (151).
Long-Term Intraocular Pressure Control
A major question regarding the results of laser trabeculoplasty is how long the IOP reduction will last. Although a high percentage of patients show an initial favorable reduction in IOP, most patients gradually lose this effect (152–159). Failure is most common in the first year, with reported rates of 19% to 23%, and thereafter failure occurs at a rate of 5% to 9% per year (157,159). As a result, approximately half of the patients will have lost the benefit of the initial trabeculoplasty by 5 years, and two thirds within 10 years, after the procedure (159).
Repeated Trabeculoplasty
If a successful IOP reduction is never achieved after 360-degree ALT, further argon laser treatment is generally not thought to be indicated. When an initial good response to treatment, lasting for approximately 1 year or more, was followed by a return to higher pressures, repeated trabeculoplasty was once common practice. However, most studies have shown a much lower success rate with repeated ALT than with the initial treatment, in the range of one third to one half (158,160–166). In one long-term study, success rates with repeated ALT were 35% at 6 months, 21% at 12 months, 11% at 24 months, and 5% at 48 months (166). Although one study suggested that the ALT can be repeated with good results (167), most surgeons no longer recommend repeated ALT. Some studies have noted a higher incidence of transient IOP rise after repeated ALT (160,161,166), and it is probably advisable to perform these in two stages of 180 degrees each, if a repeated procedure is attempted.
SLT caused more significant IOP lowering in patients with previously failed ALT compared with repeated ALT in a randomized trial (104). Repeated SLT may be almost as effective as initial SLT based on a single retrospective study (168). Repeated SLT, after either initial SLT or initial ALT, may become accepted practice, although further long-term experience is needed. Because of the greater preservation of trabecular meshwork with SLT, it has been suggested that SLT may be less likely to interfere with future incisional surgery (169).
Indications
Laser trabeculoplasty may be indicated in the treatment of those forms of open-angle glaucoma in which favorable responses have been reported, including COAG, exfoliation syndrome, pigmentary glaucoma, and open-angle glaucoma in aphakia or pseudophakia. The IOP-lowering effect was more pronounced in pseudophakic than aphakic eyes, and in eyes that had extracapsular surgery rather than intracapsular surgery (170). ALT was also preferred to cyclocryotherapy for the initial treatment of patients with uncontrolled glaucoma after a penetrating keratoplasty (171).
During the first decade of experience with ALT, the procedure was used as a supplement to maximum tolerable medical therapy, and studies have shown it to be effective in this regard (75,172). The rationale for this approach was based not only on the risk for early postoperative complications, especially the transient pressure rise, but also on the concern that eyes treated with laser trabeculoplasty might eventually become more difficult to control than if they had been left on medical therapy. The histopathologic studies showing proliferation of a cellular layer over the trabecular meshwork have given reason to seriously consider this theoretical complication (28,30,36,37). Nevertheless, short-term and long-term studies of ALT for open-angle glaucoma suggest that the procedure may be a safe and effective initial treatment of glaucoma (172–179).
In a multicenter clinical trial (the Glaucoma Laser Trial), 271 patients with newly diagnosed open-angle glaucoma were randomly assigned to initial ALT in one eye and timolol, 0.5%, in the other eye, with the same stepped regimen of additional medical therapy in either eye as required (178). During the first 2 years of follow-up, the laser-treated eyes had a slightly lower mean IOP of 1 to 2 mm Hg, although more than half of these eyes eventually required the addition of one or more medications. In a follow-up study of 203 of these patients, with a mean duration of 7 years, eyes initially treated with laser trabeculoplasty had 1.2–mm Hg greater reduction in IOP, 0.6-dB greater improvement in visual field, and slightly less optic nerve head deterioration (179). Although these findings suggest that initial treatment with ALT is at least as efficacious as initial treatment with topical medications that were available at the time of the study, medical therapy is still more commonly used in North America, particularly with the newer, more efficient IOP-lowering topical medications.
A shorter-term study comparing SLT and a topical prostaglandin, latanoprost, found that the two therapies were equally effective over 1 year (180,181). Randomized, controlled trials comparing treatment of 180 degrees of trabecular meshwork with ALT versus SLT showed no difference regarding effectiveness of IOP lowering up to 5 years (182,183). However, most clinicians treat 360 degrees of trabecular meshwork with SLT at one time. Some support in the literature indicates that 360 degrees is more effective than 180 degrees (181,184). Furthermore, a second treatment of the trabecular meshwork using SLT after SLT or ALT as the first laser trabeculoplasty is effective (168,185).
LASER IRIDOTOMY
Historical Background
In 1956, Meyer-Schwickerath (186) first reported the use of light energy to create a hole in the iris. Using the xenon-arc photocoagulator, he and others found that a peripheral iridotomy could be produced, but that the amount of heat required damaged the cornea and the lens (186,187). With the introduction of lasers in the 1960s, investigation of this treatment modality continued, primarily with ruby lasers (188–191). However, as with laser trabeculoplasty, laser iridotomy became clinically practical after the advent of argon laser technology in the 1970s. By the mid-1970s, several reports of successful argon laser iridotomy appeared in the literature (192–195), and by the end of that decade, laser iridotomy had replaced incisional iridectomy as the surgical procedure of choice for angle-closure glaucomas. During the 1980s, continued study of laser iridotomy techniques led to the popular use of the Nd:YAG laser for this operation.
Techniques
The basic principle of laser iridotomy is the creation of a hole in the peripheral iris with an argon or Nd:YAG laser, which allows equalization of the pressure between the posterior and anterior chambers, deepening of the anterior chamber, and opening of the anterior chamber angle.
Instruments
Several different types of lasers and surgical techniques can be used to create an iridotomy. The unit most commonly used in the early days of laser surgery was the continuous-wave argon laser (192–201). Other lasers were also shown to be effective for creating iridotomies, including the pulsed argon laser and the krypton laser (197,202,203). However, the pulsed Nd:YAG laser subsequently gained popularity and is the most commonly used unit for creating laser iridotomies today (204–211). A portable Nd:YAG laser is effective for use in remote geographic areas (212). Other lasers have also been evaluated for performing iridotomies. Those units and the relative merits of the argon versus Nd:YAG laser iridotomies are considered later in this chapter.
A contact lens is helpful in performing a laser iridotomy, because it (a) keeps the lids separated, (b) minimizes corneal epithelial burns by acting as a heat sink, and (c) provides some control of eye movement. In addition, convex-surfaced contact lenses have been designed to increase the power density on the iris (213–215). The most commonly used is the Abraham iridotomy lens, which has a 66-D planoconvex button bonded to the front surface of the contact lens (Fig. 36.5) (213). This lens doubles the laser-beam diameter at the level of the cornea, while reducing it to approximately one half of the original size on the iris, which reduces the power density at the cornea to one fourth of the original level and increases it on the iris by a factor of four. Another contact lens, the Wise iridotomy–sphincterotomy lens, has a 103-D optical button decentered at 2.5 mm, which further reduces the iris focal spot and increases the energy density (215). These principles have their greatest application with the argon laser, although the same contact lenses are also useful with the Nd:YAG laser.

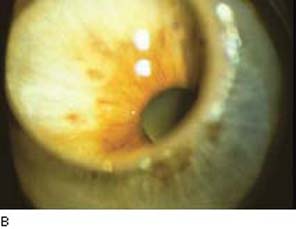
Figure 36.5 A: Abraham contact lens with planoconvex button bonded to front surface for laser iridotomy. B: Slitlamp view of an iris magnified with the Abraham iridotomy lens.
With all lasers and contact lenses, a high magnification (e.g., 40×) should be used in the slitlamp delivery system.
Preoperative Medication
Topical pilocarpine may be instilled before the procedure, which helps to maximally thin and stretch the peripheral iris. If the patient presents with an acute attack of angle-closure glaucoma, it is best to break the attack medically, if possible, and maintain the patient on medication to allow clearing of any corneal edema and to facilitate constriction of the pupil. If significant iritis persists after breaking the attack, it may be advisable to use topical steroids for 24 to 48 hours before proceeding with the laser surgery. However, if the attack does not respond to medical therapy, laser iridotomy (or iridoplasty or pupilloplasty, as discussed later in this chapter) may be effective in breaking the attack (216).
In nearly all cases, only topical anesthesia, such as proparacaine, 0.5%, is required. Only rarely is a retrobulbar injection needed for a patient who has nystagmus or is uncooperative. It has become a standard practice among most surgeons to also use topical apraclonidine to reduce the risk for a postoperative IOP rise (217). In the original studies, apraclonidine, 1%, was instilled 45 to 60 minutes before and immediately after the procedure (218), although a single postoperative drop of apraclonidine, 0.5%, has been shown to be as effective as the 1% concentration in preventing IOP elevation (114).
Selection of Treatment Site
Any quadrant of the iris can be used to create the laser iridotomy, although our preference is between 11 and 1 o’clock if the opening will be entirely covered by the lid, and otherwise temporally. The reason for this is to avoid the iridotomy in a location where the lid margin bisects the iridotomy, as this can result in monocular optical symptoms such as transient ghosting of images, blurring, shadows, halos, glare, crescents, or a horizontal line (219,220). When argon laser iridotomy is performed, the 12-o’clock position is usually avoided, because gas bubbles may collect in that area and interfere with completion of the procedure. One exception to the selection of a superior iris quadrant is the patient with silicone oil in an aphakic eye, in which case the iridotomy should be placed inferiorly to avoid blockage by the oil, which rises to the top of the eye.
Whichever quadrant is used, the slitlamp should always be positioned so that the laser beam is directed away from the macula. The iridotomy is usually placed between the middle and peripheral thirds of the iris. However, if this is not feasible, because of peripheral corneal haze or close proximity between peripheral iris and cornea, a more central location can be used, as long as it is peripheral to the sphincter muscle.
Several features of the iris may facilitate creation of the iridotomy. An area of thin iris or a large crypt is usually easier to penetrate. In lightly pigmented eyes, a local area of increased pigmentation, such as a freckle, may improve absorption of argon laser energy. In addition, the radially arranged white collagen strands in the stroma can be very difficult to penetrate, especially with argon laser, and selecting a treatment site where two strands are more widely separated is helpful (221). The collagen strands may also contain radial vessels, which should be avoided with Nd:YAG laser iridotomy.
Techniques with Continuous-Wave Argon Laser
Several basic techniques have been advocated for producing iridotomies with the continuous-wave argon laser. The “hump” technique involves first creating a localized elevation of the iris with a large-diameter, low-energy burn, and then penetrating the hump with small, intense burns (222). In the “drumhead” technique, large-diameter, low-energy burns are placed around the intended treatment site to put the iris on stretch, and that area is then penetrated with small, high-energy burns (198,223). A third, and probably the most commonly used, approach goes directly to penetrating burns (197,200,201). This last technique may be modified by using multiple short-duration burns (224,225). None of these approaches, however, is ideal for all situations, and it is best to tailor the iridotomy technique primarily according to the color of the iris. For irides of any color, the argon laser settings are first selected for the iris stroma and then adjusted for the pigment epithelium.
Medium Brown Iris
This is the easiest iris to penetrate with continuous-wave argon laser, and the following method represents one technique to use in these patients. Protocols for irides of other colors are modifications of this basic technique.
Argon laser settings of 0.1- to 0.2-second duration, 50-µm spot size, and 700 to 1500 mW (average, 1000 mW) are initially used to create a crater in the iris stroma. The first few applications may produce gas bubbles, which usually float up away from the treatment site (Fig. 36.6). If the bubble does not move, it can be dislodged by going through it with the next laser application or by placing the beam adjacent to the bubble. A cluster of several contiguous burns is used to produce a stromal crater of approximately 500 µm in diameter. Additional laser applications are then placed in the bed of the crater until the pigment epithelial layer is reached, as evidenced by a cloud of pigment.
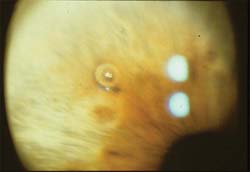
Figure 36.6 Argon laser was used to create a crater in the stroma of this medium brown iris. Gas bubbles (shown) can form with the first few laser applications; they usually float away from the treatment site but can be dislodged by subsequent laser applications, if necessary.
When most of the stroma in the crater has been eliminated and only pigment epithelium remains, the laser intensity should be reduced to clean away the remaining tissue. Typical settings for this stage of the procedure are 100 µm and 500 to 700 mW, or 50 µm and 200 to 600 mW, with a duration of 0.1 to 0.2 second. Higher-intensity burns at this stage of the treatment may dislodge adjacent pigment epithelium, creating a “cascade phenomenon,” which causes further obstruction of the iridotomy. These same settings can be used for irides of other colors because the pigment epithelial layer is similar in all eyes.
This two-stage technique for argon laser iridotomy in the medium brown iris normally takes 30 to 60 laser applications to create a patent iridotomy.
Dark Brown Iris
A laser iridotomy is more difficult to achieve in these eyes, partially because of the thick, dense stroma. Standard initial settings (as described previously) often produce a black char in the stromal crater, making the site resistant to further penetration. One way to minimize this complication and achieve a patent iridotomy in the dark brown iris is to use multiple short-duration burns, called the “chipping” technique (222,224,226). The important feature of this modification is the short exposure time of 0.02 to 0.05 second, with standard settings of 50 µm and 700 to 1500 mW. With this approach, minute fragments of stroma are “chipped away,” often requiring 200 to 300 applications to penetrate the stroma. Once the pigment epithelial layer is reached, the settings should be changed to the lower intensity level, as described for the medium brown iris, to complete the procedure.
Blue Iris
These eyes can also be difficult with argon laser iridotomy because the lightly pigmented stroma does not absorb laser light sufficiently to produce a burn through this portion of the iris. The pigment epithelium near the treatment site may be dislodged, leaving intact stroma that is impermeable to aqueous flow. Some surgeons prefer a two-stage approach, in which settings of 500 µm and 200 to 300 mW are first used to create a local tan-colored area of increased stromal density, followed by penetration burns of 50 µm, 500 to 700 mW, and 0.1 second to create a full-thickness hole in the stroma (227). Others have suggested a direct approach, using settings of 50 µm, 1000 to 1500 mW, and a prolonged duration of 0.5 second, which usually burns a hole through the stroma in two to three applications (221,226). With either technique, the settings should then be changed to those described for the medium brown iris to penetrate or remove the remaining pigment epithelium from the iridotomy site.
Techniques with Nd:YAG Laser
As previously noted, Nd:YAG is now the most commonly used technique for laser iridotomy. The extremely high energy levels and short exposure times of these lasers electromechanically disrupt tissue, independent of pigment absorption and the thermal effect. As a result, they are particularly useful in creating iridotomies in light blue irides but are effective in all eyes. The technique usually involves simultaneous perforation of the iris stroma and pigment epithelium with energy levels in the range of 5 to 15 mJ (204–211). The pulse duration is fixed for each instrument, in the range of 12 nanoseconds, but the number of pulses per burst can be adjusted in most units, with surgeons generally preferring 1 to 3 pulses per burst. The spot size is also fixed, although some units provide a choice between a single focal point and multiple focal points, with the latter creating a larger lesion. Because the wavelength of the Nd:YAG laser is beyond the visible spectrum, a helium–neon or diode laser beam is typically used for focusing on the iris. With instruments that allow a selected separation between the focal points of the two laser beams, the setting should be such that they are coincident when performing a laser iridotomy.
The standard technique uses the same criteria as for argon laser iridotomy in selecting the iris site, although it is often possible to place the iridotomy more peripherally with the Nd:YAG laser. The latter is desirable, among other reasons, to avoid injuring the lens. When selecting the treatment site, attention should be given to avoid any apparent iris vessels, because these are more likely to bleed with Nd:YAG than with argon laser surgery. A patent iridotomy can often be created with a single laser application, and rarely are more than two or three required, especially in blue or light brown eyes. However, the iridotomy may be smaller than those produced with an argon laser (Fig. 36.7A,B). Cases have been reported of acute angle-closure glaucoma in eyes with patent, but small, Nd:YAG laser iridotomies, and it has been suggested that an iridotomy should be at least 150 to 200 µm in diameter (228). The iridotomy may change in shape and position and occasionally in area after dilatation (229), and it is good practice to make it large enough initially but also to check it after dilatation. If there is doubt regarding the size of the iridotomy during the procedure and it is difficult to enlarge it, creating more than one iridotomy is advisable.
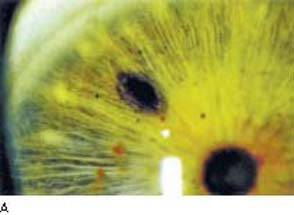
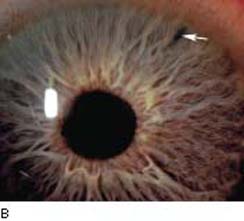
Figure 36.7 A: Argon laser iridotomy with patency demonstrated by visualization of anterior lens capsule. B: Typical appearance of peripheral iridotomy created with an Nd:YAG laser.
Several variations in technique have been described. One uses both the argon and Nd:YAG lasers by first creating a stromal crater with short-duration argon laser burns and then penetrating the iris with low-energy, single-pulse Nd:YAG applications (230–232). This has an advantage of minimizing bleeding by first coagulating iris vessels. It is especially useful in eyes with thick, dark iris stroma, in which the Nd:YAG laser energy may cause considerable disruption and dispersion of stromal tissue before penetrating the iris. Another technique involves multiple low-energy (1.0 to 1.7 mJ) applications in a line across the radial iris fibers to create an iridotomy of larger, more controllable size, which was thought to be safer than a similar approach with the argon laser or the standard, higher-energy Nd:YAG technique (233,234). Iridotomies have also been created experimentally with the transscleral application of longer-duration thermal Nd:YAG laser burns via a fiber-optic system (235).
Techniques with Other Lasers
Pulsed Argon Laser
This instrument emits laser energy in a chain of very short pulses, rather than in a continuous wave, which vaporizes the absorbing tissue with minimal heat loss and destruction to the surrounding area. These features provide some advantage over continuous-wave argon lasers for producing iridotomies in that more energy is used in penetrating the iris, with less distortion and disruption of the surrounding tissue (202).
The basic technique is similar to that for continuous-wave argon laser iridotomy. The settings, however, differ considerably for the pulsed argon laser unit. The perforating mode is used, and the power setting is 20 to 25 W. The usual parameters are 50 µm, 0.2 second, and 300 pulses/sec, adjusted according to tissue response (the individual pulse is fixed at 128 microseconds). With these settings, the number of exposures to achieve an iridotomy varies from 2 to 250, depending on the type of iris (202).
Neodymium: Yttrium Lithium Fluoride (Nd:YLF) Laser
This 1053-nm laser can create iridotomies of precise size and shape with minimal thermal damage to the surrounding tissue because of low energy per pulse levels, with a short pulse duration in picoseconds, and a high repetition rate. Optimal settings, established in a series of cadaver eyes, included a rectangular cutting pattern of 0.3 × 0.3 mm, 500-µm cutting depth, 50-µm spot separation, and 200 to 400 pulses/sec (236,237).
Semiconductor Diode Laser
As noted in Chapter 35, the semiconductor diode laser has several distinct advantages over other lasers, including the small, portable size, the solid-state construction, which provides durability and relatively low maintenance requirements, and the need for only a standard electric outlet and no water cooling. With a wavelength of approximately 805 nm and operation in the continuous-wave mode, the mechanism of iridotomy is like that of the argon laser—that is, absorption by melanin, resulting in photocoagulation, rather than the electromechanical disruption of the Nd:YAG and Nd:YLF lasers. In rabbit studies and preliminary clinical trials, the settings and the clinical and histopathologic results were all similar to those noted in this chapter for the argon laser (238–240).
Other Lasers
As noted previously, the krypton laser was found to be effective in creating iridotomies (203). A Q-switched ruby laser was also found in monkey studies to be suitable for producing iridotomies (241), and dye lasers have been used clinically to create iridotomies with a single pulse (242). The continuous-wave, frequency-doubled Nd:YAG laser with a wavelength of 532 nm, pumped by a diode laser, successfully created patent iridotomies in rabbit eyes with the thermal damage zones comparable to the argon laser (243).
Results of Argon and Nd:Yag Laser Iridotomies
When laser iridotomy is performed for an acute angle closure, the IOP decreases and remains stable without requiring additional surgery in approximately two thirds of white patients and half of Asian patients (201,244–246). The difference may be explained by mechanisms other than pupillary block, such as changes in the angle morphologic characteristics, a longer duration, and severity of the attack (245,247).
In chronic angle-closure glaucoma, despite widening of the angle in 73% to 97% of eyes (246,248), eyes that have optic disc and visual field damage require filtering surgery in approximately half of the patients, despite the presence of a patent iridotomy (249,250). The outcomes appear to be similar among white and Asian patients (250). The effect of iridotomy on one eye is predictive of the effect on the fellow eye (248).
Prophylactic laser iridotomy prevented IOP elevation in 88.8% of fellow eyes in patients with acute angle closure within 4 years of follow-up and is recommended for the treatment of fellow eyes of patients with acute angle closure (250,251). However, because some of the fellow eyes may experience IOP elevation within 6 to 12 months, despite the presence of a patent iridotomy, close follow-up is recommended (245,252).
Comparison of Argon and Nd:Yag Laser Iridotomies
Histologic studies have shown that iridotomies created with an argon laser have more extensive early edema and tissue destruction at the margins of the treatment site than iridotomies created with the Nd:YAG laser, in which the lesions are more circumscribed with limited tissue alterations at the margins (202,209,211,253). However, freeze-frame analysis of high-speed cinematography in ox eyes showed particles traveling over 8 mm from the Nd:YAG treatment site at speeds in excess of 20 km/h (210), and the shock waves affected the trabecular meshwork and corneal endothelium of monkey eyes when the Nd:YAG application was within 0.8 mm of the limbus (254).
Argon and Nd:YAG laser iridotomies were compared in human autopsy eyes using a high-magnification video recording system that allowed real-time observation of the posterior iris during the laser procedures (255). With argon laser iridotomy, gradual mounding up of iris pigment epithelium occurred with each successive energy application before final penetration. In contrast, Nd:YAG laser iridotomy caused a complete disruption and dispersal of the pigment epithelium with a single pulse of energy. These observations may explain the tendency for argon laser iridotomies to become obstructed with pigment epithelium, which is rarely seen with Nd:YAG laser iridotomies.
In clinical comparisons of the two surgical approaches, the Nd:YAG laser iridotomies had the disadvantage of frequent bleeding, although this usually stops spontaneously or by applying pressure to the eye with the contact lens and rarely leads to significant complications (206,207,256,257). Disadvantages of argon laser iridotomy, on the other hand, include more iritis, pupillary distortion, and late closure of the iridotomy. When an iridotomy could not be created with the argon laser, a patent iridotomy was be achieved in all eyes with the Nd:YAG laser in single sessions (258). Nd:YAG laser iridotomies, in general, require considerably fewer total applications, with a marked reduction in total energy delivery, compared with argon laser iridotomies.
Prevention and Management of Complications
As with laser trabeculoplasty, a transient IOP rise and a mild anterior uveitis are common early postoperative complications. Other potential complications include closure of the iridotomy, corneal damage, hyphema, cataract formation, retinal burns, malignant glaucoma, and monocular blurring.
Transient Intraocular Pressure Rise
This is one of the most common serious complications in the early period after argon or Nd:YAG laser iridotomy (259,260). It was reported in 24% of the eyes undergoing Nd:YAG iridotomy (246). The IOP rise is caused by reduced outflow facility, with an actual decrease in aqueous production (261). A biphasic IOP response has been seen in rabbits, in which the initial IOP rise of 0.5 to 2 hours’ duration is followed by a prolonged IOP reduction lasting 6 to 24 hours (262). Studies in rabbits also suggest that this pressure response is related to a release of prostaglandin and prostaglandin-like substances into the aqueous with a breakdown in the blood–aqueous barrier and an accumulation of blood plasma and fibrin in the anterior chamber angle (262–268). A histopathologic study in monkey revealed a rapid accumulation of particulate debris in the angle (269), which may also contribute to the transient IOP elevation.
Clinically, the risk for a transient IOP rise was found in one study to be related to the total energy delivered, but not to the presence of chronic angle-closure glaucoma (260), whereas another study found no correlation with total laser energy but did find that the preoperative outflow facility was directly related to the maximum postoperative IOP elevation (261). As previously noted, 1 drop of apraclonidine, 0.5% to 1%, 1 hour before or immediately after the laser surgery has a profound effect on minimizing this complication (114,218).
Pretreatment with latanoprost was associated with an increase in IOP within the first 2 hours following iridotomy (270). This was likely due to the short time interval between drug instillation and laser treatment, which prevents the medication from achieving its peak effect and limits the effectiveness of latanoprost as a prophylactic medication in anterior segment laser surgery (270).
Anterior Uveitis
Some degree of transient iritis occurs after laser iridotomy in all eyes, which is associated with the blood–aqueous barrier breakdown noted in animal studies (264,266–268). Topical steroids for the first 3 to 5 postoperative days are sufficient to control this mild complication in most cases. In rare cases, however, an eye may have a marked inflammation, sometimes occurring days or weeks after the procedure, with associated hypopyon (271,272). Granulomatous endophthalmitis was reported following laser iridotomy, associated with several large tears in the anterior lens capsule of a blind eye with a mature cataract (272). A case of prolonged iritis with transient cystoid macular edema has also been described (273), and two cases have been reported in which postoperative inflammation and long-term miotic therapy were thought to be responsible for occlusion of the pupil with a pigmented pseudomembrane (274).
Closure of Iridotomy
The iridotomy may close during the first few weeks, especially with argon laser iridotomy, due to accumulation of pigment granules and debris. It may be advisable, therefore, to continue use of pilocarpine for the first 4 to 6 postoperative weeks. If the iridotomy remains patent, stopping use of the miotic after this time is usually safe, unless it is needed to control a chronic pressure elevation. Some authors suggested that a mydriatic provocative test should be used after stopping use of the miotic to confirm the functional reliability of the iridotomy (201). Late closure is rare with Nd:YAG laser iridotomies. In one series of 200 cases, the two late closures were in eyes with preexisting chronic uveitis (275).
As discussed earlier, the minimum diameter of a laser iridotomy that is needed to prevent further attacks of angle-closure glaucoma is yet to be determined and probably differs from one patient to the next. Cases have been reported in which angle closure recurred despite patent but small iridotomies, and as previously noted, a minimum diameter of 150 to 200 µm has been recommended (228,276). In some eyes following argon laser surgery, the laser iridotomy spontaneously enlarges over months or years (277), although this should not be relied on in borderline situations, in which case the opening should be further enlarged.
Patency of the iridotomy is best confirmed by visualizing anterior lens capsule or vitreous face through the opening (Fig. 36.7A). Transillumination can also be used, although this is sometimes misleading, especially with a blue iris, in which dislodged pigment epithelium can produce a transillumination defect despite an intact overlying stroma, which is impermeable to aqueous flow.
Corneal Damage
Focal epithelial and endothelial burns of the cornea are not uncommon when larger amounts of laser energy are used, although these usually heal quickly with no apparent sequelae. In monkey eyes, laser iridotomy was not associated with significant endothelial cell damage (278). In several clinical trials, pachymetry has revealed no significant difference in corneal thickness before and after laser iridotomy (279–281). Specular microscopic studies have been less conclusive, however, with some showing no significant difference in endothelial cell count (279,280), whereas others have revealed a loss of endothelial cells or an increase in cell size (105,281–283). Generalized corneal decompensation has been reported in several series, nearly all of which involved argon laser iridotomy (283–287). This often begins with focal corneal edema overlying the iridotomy site, followed by generalized corneal decompensation, which may not appear until months to years after the laser surgery. These cases frequently require penetrating keratoplasty, histology of which typically reveals abnormalities characteristic of Fuchs endothelial corneal dystrophy (285,287). Factors that may predispose to this complication include episodes of angle-closure glaucoma with pressure elevations and inflammation, cornea guttata, diabetes, and high total laser energy (284–287). Descemet membrane detachment after laser iridotomy was also reported (288).
Hyphema
As previously noted, a small amount of bleeding from the iridotomy site is common following Nd:YAG laser iridotomy but is rarely serious (204–208). Persistent bleeding from the treatment site can usually be stopped by applying pressure to the eye with the contact lens for a few seconds to a minute. Hyphemas are uncommon after argon laser iridotomies but may occur (289,290), especially in eyes with rubeosis iridis or uveitis.
Cataract Formation
Focal anterior lens opacities are common beneath an iridotomy produced with argon laser energy (206,208,291). Most of these are nonprogressive, although reduced visual acuity due to cataract progression has been documented (291). The rate of progression is similar to that following incisional surgical iridectomy (201), and a clear cause-and-effect relationship between either surgical approach and cataracts has not been established. Lens changes are much less common with Nd:YAG laser iridotomies (206–208), although capsular damage with rare cataract formation has been reported (292–294). In two rabbit studies, no lens damage was seen with either argon or Nd:YAG laser iridotomy, even when additional laser applications were placed through patent iridotomies (295,296). A study in monkeys, however, suggested a threshold for lens damage with Nd:YAG laser iridotomy, with no damage at 6 mJ or less and one to two pulses per burst, but local damage with higher energies or three pulses per burst (297).
Retinal Injuries
Most visual function studies have shown no adverse effect from argon laser iridotomy (298), and the same is presumably true for Nd:YAG laser iridotomy. One study, however, did reveal static perimetric and fluorescein angiographic evidence of focal retinal damage in the quadrant of treatment 6 months after argon laser iridotomy (299). Retinal damage is best minimized by always aiming the laser beam toward peripheral retina. Failure to do so may result in serious retinal burns, and acute permanent loss of vision due to inadvertent foveal photocoagulation during argon laser iridotomy has been reported (300). Macular injuries from an Nd:YAG iridotomy have also been reported (301). The final visual acuity in these cases depends on the distance between the injury and the fovea (301). The risk is also reduced but not eliminated by using an Abraham lens (302). One case of temporary bilateral serous choroidal and nonrhegmatogenous retinal detachment following Nd:YAG laser iridotomy has also been reported (303).
Malignant Glaucoma
Cases of possible malignant glaucoma have also been reported following laser iridotomy for acute or chronic angle-closure glaucoma (304–307), one of which was a simultaneous bilateral case 4 weeks after bilateral laser iridotomy (308).
Monocular Blurring
If the iridotomy is not fully covered by the upper lid, the patient may report monocular blurring, diplopia, or “ghost images.” Diplopia, or “ghost images,” may occur when the upper lid and associated tear film bisect the light path through the patent iridotomy. In some patients, diplopia, often alleviated when the lid is lifted away from the eye, may result despite an iridotomy that is well covered by the upper eyelid. This may result from a prism-like effect of the tear meniscus along the margin of the upper eyelid. If tinted glasses or sunglasses fail to relieve symptoms, a cosmetic contact lens can be helpful in unusually symptomatic cases. Some investigators report that diplopia occurs less frequently when the iridotomy is placed in the horizontal axis (3- or 9-o’clock positions) (309).
LASER PERIPHERAL IRIDOPLASTY (GONIOPLASTY)
There are times in which a patent iridotomy may fail to relieve angle closure, as with a microphthalmic or nanophthalmic eye, the swelling (e.g., sulfonamide-induced uveal effusion) or forward rotation of the ciliary body (i.e., plateau iris syndrome), or the presence of peripheral anterior synechiae. In each of these situations, the anterior chamber angle may be opened by applying low-energy argon laser contraction burns to the peripheral iris. The procedure has been referred to as laser peripheral iridoplasty, gonioplasty, or peripheral iris retraction.
Mechanisms of Action
The mechanism of action is a tightening of the peripheral iris, which pulls it posteriorly from the trabecular meshwork. The histopathology of eyes treated with peripheral iridoplasty revealed contraction furrow formation, proliferation of fibroblast-like cells, collagen deposition on the iris surface, denaturation of stromal collagen, and coagulative necrosis of blood vessels in the anterior two thirds of the iris stroma (310). These findings are believed to suggest that the immediate, short-term mechanism of peripheral iridoplasty is heat shrinkage of collagen, whereas the long-term effects may be related to contraction of a fibroblastic membrane. The observation of coagulative necrosis of iris blood vessels also provides a note of caution that overtreatment may lead to iris necrosis.
Techniques
Suggested argon laser settings for peripheral iridoplasty vary considerably, with ranges of 50- to 500-µm spot size, 0.5-second duration, and 150 to 1000 mW of power (217,311–313). In general, however, a laser application of relatively large area, long duration, and low power is preferable, and reasonable initial settings are 200 µm, 0.2 second, and 400 mW. The power or duration should be increased if no contraction is produced but reduced if pigment liberation is produced by the laser application. The recommended number of applications also varies. Approximately 10 to 15 burns are usually applied to peripheral iris in each quadrant, and additional applications can be placed in a row adjacent to the first burns if necessary (Fig. 36.8). It is usually advisable to treat no more than 180 degrees of an angle in a single session. Gonioplasty with a diode laser was also reported for the treatment of chronic angle-closure glaucoma and acute angle closure (314,315).
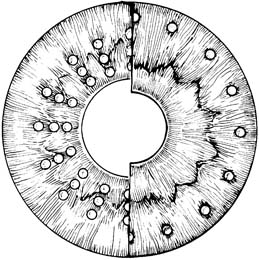
Figure 36.8 Laser peripheral iridoplasty (right) deepens the anterior chamber angle with low-energy contraction burns to the peripheral iris, while laser pupilloplasty (left) dilates the pupil with low-energy contraction burns to the more central iris.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


