Glaucomas Associated with Intraocular Tumors
A variety of intraocular tumors and tumor-like ocular disorders can give rise to glaucoma (Table 21.1) (1). In one survey of 2597 patients with intraocular tumors, 5% of the tumor-containing eyes had tumor-induced elevated intraocular pressure (IOP) at the time of the tumor diagnosis (2). In some cases, the mass lesions represent life-threatening malignancies, whereas other tumors are benign, creating critical problems with diagnosis and management.
Glaucoma secondary to intraocular tumors should be considered in all cases of unilateral or heavily asymmetric glaucoma, particularly when certain features such as iris heterochromia, lack of response to IOP-lowering treatment, or lack of response to steroids, which may indicate pseudouveitis, are present (1). B-scan ultrasonography and ultrasonographic biomicroscopy (UBM), as well as other diagnostic techniques (Table 21.2) (1), may be essential to demonstrate the presence of an intraocular tumor, particularly when it may be masked by a cataract, vitreous hemorrhage, retinal detachment, or other opacity in the ocular media. For patients with an intraocular malignant tumor, the emphasis shifts from the prevention of blindness to the preservation of life, but care must be taken in eyes with benign lesions to avoid loss of vision from unnecessary treatment. In this chapter, we consider the differential diagnosis and management of glaucomas associated with intraocular tumors.
PRIMARY UVEAL MELANOMAS
Melanomas of the uveal tract, the most common primary intraocular malignancy, are frequently associated with glaucoma by several mechanisms and with a variety of clinical presentations. In one large histopathologic study of eyes with malignant melanomas involving one or more portions of the uveal tract, the overall prevalence of glaucoma was 20% (3). Anterior uveal melanomas lead to IOP elevation more frequently than posterior melanomas, with reports of 41% and 45% of patients in two series, and choroidal melanomas were found to have associated glaucoma in 14% in one study (3,4). These figures are undoubtedly skewed, because one series represents histopathologic material and the other involves primarily patients referred to a glaucoma service (3,4). A clinical series from an oncology service may provide more meaningful statistics, in which 3% of 2111 eyes with uveal melanomas had associated IOP elevation, including 7% with iris melanomas, 17% with ciliary body melanomas, and 2% with choroidal melanomas (2). (Metastatic melanomas are rarely found in the eye but can cause glaucoma and are discussed later, under “Systemic Malignancies.”)
Anterior Uveal Melanomas
Clinical Presentations and Mechanisms of Glaucoma
Melanomas of the anterior uveal tract most often arise from the ciliary body. These may be difficult to visualize directly because they often manifest as a smooth-domed elevation of the overlying iris. Wide dilatation may allow gonioscopic visualization of the lesion, which is typically seen as a dark brown mass between the iris and lens. In other cases, a primary melanoma of the ciliary body may extend through the peripheral iris and become visible as a nodular mass on the iris stroma and in the anterior chamber angle. Primary melanomas of the iris are usually easily seen by slitlamp biomicroscopy and gonioscopy, typically as slightly elevated, brown masses on the stroma (Fig. 21.1) with attendant hyperchromic heterochromia. Some melanomas of the iris, however, may be amelanotic and often have an associated, secondary vasculature and hypochromic heterochromia.
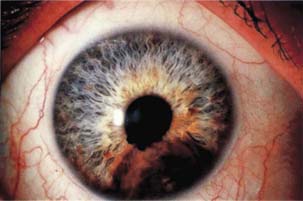
Figure 21.1 Slitlamp view of a malignant melanoma of the iris.
Melanomas of the anterior uvea may manifest as unilateral glaucoma. They may lead to glaucoma by open-angle or angle-closure mechanisms; the former mechanism is more common (Fig. 21.2). Aqueous humor outflow in the open anterior chamber angle can be obstructed by direct extension of the tumor or by trabecular meshwork seeding by tumor cells or melanin granules (1) (Fig. 21.3). In some eyes, the melanoma may arise from the iris, ciliary body, or iridociliary junction and spread circumferentially, creating a ring melanoma. A review of 14 patients with ring melanoma of the anterior chamber angle found that all patients presented with elevated IOP in the affected eye (5). In a rare presentation, a ring melanoma also masqueraded as pigmentary glaucoma (6). An anterior uveal melanoma may also extend posteriorly, causing a retinal detachment and giving the impression of a choroidal tumor. Others may extend into the anterior chamber, causing elevated IOP due to infiltration of the angle with occasional iris nodularity and heterochromia (7). Patients with ciliary body melanomas can present with chronic uveitis and refractory glaucoma (8). In melanomalytic glaucoma, macrophages containing melanin from a necrotic melanoma obstruct the trabecular meshwork (3,9). Ultrastructural studies reveal infiltration of the angle with melanin-laden macrophages, phagocytosis of melanin by trabecular endothelial cells, and tumor cells on the iris and in the meshwork (1).
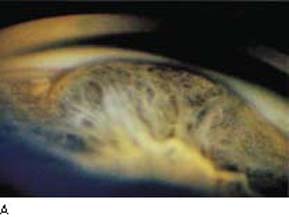
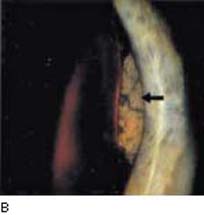
Figure 21.2 A: Gonioscopic view shows elevation of the peripheral iris in a patient with ciliary body melanoma. B: After wide dilation of the same eye, one can see a solid mass with irregular pigmentation between the iris and lens.
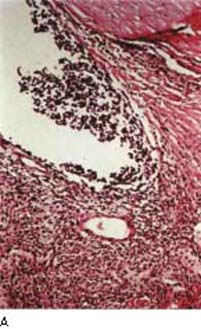
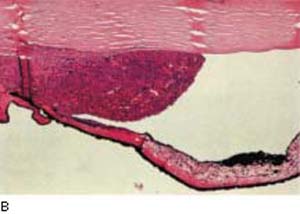
Figure 21.3 Light microscopic view of malignant melanoma cells seeding the anterior chamber angle causing glaucoma (A) and solid tumor involving the angle (B).
Another variation of anterior uveal melanoma and glaucoma occurs with a tapioca iris melanoma. This rare melanoma of the iris creates a nodular appearance resembling tapioca pudding and typically consists of low-grade spindle-type cells, although a case with epithelioid-type cells and metastases has been reported (10). Glaucoma is reported to occur in one third of the cases with tapioca melanoma (11).
An alternate mechanism of IOP elevation with an iris melanoma is neovascular glaucoma, which may resolve after excision of the tumor (12). Ciliary body melanomas may also cause an angle-closure form of glaucoma due to compression of the root of the iris into the anterior chamber angle or forward displacement of the lens–iris diaphragm (13) (Fig. 21.4).
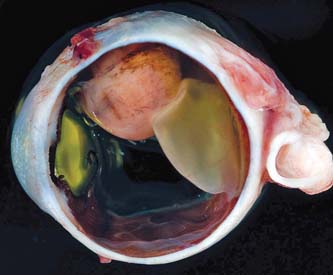
Figure 21.4 Gross appearance of a sectioned eye, showing a malignant melanoma of the ciliary body and anterior uvea adjacent to the lens, creating closure of the anterior chamber angle. (Courtesy of Prithvi Mruthyunjaya, MD.)
Some eyes with a melanoma confined to the ciliary body may have a slightly lower IOP than that of the fellow eye (14). Any alteration in tension can be an indication of an anterior uveal melanoma.
Differential Diagnosis
Several conditions can be confused with glaucoma and an anterior uveal melanoma. Associated changes may mask an underlying melanoma, whereas other mass lesions may simulate an anterior uveal melanoma. For example, iritis may appear to be present in some cases of glaucoma and melanoma, which usually represents tumor cells in the anterior chamber (4), whereas other eyes may have primary iritis with inflammatory nodules that may be confused with a malignancy (15). In one large series, a primary cyst of the iris was the most common lesion to be confused with a melanoma of the iris (16). However, anterior uveal melanomas can masquerade as cysts of the iris or ciliary body because of separation of the two epithelial layers by an eosinophilic exudate (4,13). Iris nevi may be especially difficult to distinguish from iris melanomas clinically and histologically. (Other benign tumors and metastatic malignancies must be included in the differential diagnosis of uveal melanomas and are discussed later in this chapter.)
Choroidal Melanomas
Occasionally, a patient with a melanoma of the choroid may present with acute angle-closure glaucoma. This is usually caused by the forward displacement of the lens–iris diaphragm by a large posterior tumor, which is commonly associated with a total retinal detachment. The finding of a retinal detachment, especially if serous, and glaucoma in the same eye should alert the clinician to the possibility of an underlying malignant melanoma. Other reported mechanisms of IOP elevation in association with choroidal melanomas include neovascular glaucoma and pigment dispersion in the vitreous with melanomalytic glaucoma (3,17). In addition to retinal detachment, other conditions that may mask the presence of a choroidal melanoma include intraocular inflammation and hemorrhage, and a dislocated lens nucleus may mimic a choroidal melanoma, especially when inflammatory glaucoma is also present (18,19).
Diagnostic Adjuncts
The difficulty in detecting a uveal melanoma and distinguishing it from other intraocular tumors occasionally necessitates the use of special diagnostic measures.
Ultrasonography
UBM is a valuable tool to help determine the location, size, and extent of uveal melanomas (20–22). It can also help in differentiating retroiridal cysts from tumors (23), but it cannot distinguish between benign and malignant tumors. B-scan ultrasonography may be useful in demonstrating the presence of a ciliary body melanoma (Fig. 21.5) or a choroidal melanoma when the latter is masked by a retinal detachment, vitreous hemorrhage, or other opacity in the ocular media. However, this technique does not always distinguish a neoplasm from other masses of the posterior ocular segment (24).


Figure 21.5 UBM features of malignant melanoma of the iris. A: Iridociliary melanoma replacing the peripheral iris and ciliary body and filling the anterior chamber angle. Mass is slightly sonolucent compared with normal iris stroma.B: Larger iridociliary melanoma. Iris appears to arise from the side of the mass. (From Corrêa ZM, Augsburger JJ. Ultrasound biomicroscopy of the anterior ocular segment. In: Tasman W, Jaeger EA, eds. Duane’s Foundations of Clinical Ophthalmology. Vol 2. Philadelphia, PA: Lippincott Williams & Wilkins; 2008:chap 106.)
Fluorescein Angiography of the Iris
Fluorescein angiography of the iris has been useful in distinguishing melanomas from benign lesions of the iris, such as leiomyomas and benign melanocytic tumors (25–27). The angiographic characteristic of a melanoma of the iris is diffuse and eventually confluent fluorescence emanating from ill-defined vascular foci (27).
Cytopathologic Studies
An aqueous or vitreous aspirate may provide sufficient material for a histopathologic diagnosis of a primary or metastatic malignancy (28,29). This may be especially useful for suspected melanomas of the anterior uvea when cells can be seen in the aqueous by slitlamp biomicroscopy (Fig. 21.6). The technique involves aspiration of aqueous with a small-gauge needle through the limbus or peripheral cornea. For neoplastic cells, cytologic preservation is best with a Millipore filter (29).
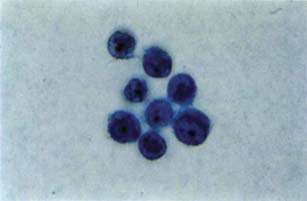
Figure 21.6 Light microscopic view of melanoma cells aspirated from the aqueous of an eye with anterior uveal melanoma (Papanicolaou, ×400). (From Ritch R, Shields MB, eds. The Secondary Glaucomas. St. Louis, MO: CV Mosby; 1982, with permission.)
A fine-needle aspiration biopsy may also be used to obtain material for cytopathologic study from a suspected anterior uveal or choroidal melanoma (30).
Frozen Section Diagnoses
A frozen section may be helpful in identifying a tumor of the iris and in determining the surgical resection margins of the lesion (31). In sending the iridectomy specimen to the pathologist, care must be taken to identify the orientation of the tissue and to avoid curling of the edges (31).
Prognosis
When a uveal melanoma is associated with glaucoma, the prognosis appears to be worse for metastasis and death, compared with that for a melanoma without glaucoma. In one study, three of four patients with a primary melanoma of the ciliary body and glaucoma died of metastatic disease within 2.5 years after enucleation (4), and another investigation of uveal melanomas in children and adolescents identified glaucoma as a predominant factor relating to a fatal outcome (32). Histopathologic studies of eyes with a ciliary body melanoma and glaucoma often reveal tumor cells in the aqueous outflow system, which is a potential route of extraocular metastasis (4). Patients with choroidal melanomas and glaucoma also have a more guarded prognosis, because the tumor is usually large by the time the glaucoma has developed.
Melanomas of the iris generally have a better prognosis than other uveal melanomas (33,34). The relatively benign nature may reflect the earlier detection and small size at the time of detection, allowed by the more obvious anterior location. However, metastases have been reported to occur. In one series of 1043 reported iris melanomas, 31 (3%) eventually metastasized (35). In another series of 169 microscopically proven iris melanomas, 5% developed distant metastasis over 10 years (36). In the latter series, metastases were significantly more likely to develop in older patients who demonstrated tumors of iris root or angle location with elevated IOP and extraocular extension. The cell type influences the rate of metastasis, with none among spindle A tumors in one series, 2.6% with spindle B tumors, 6.9% with epithelioid tumors, and 10.5% with mixed-cell tumors (35). The presence of glaucoma with an iris melanoma may also increase the risk for metastasis. Ring melanoma of the trabecular meshwork and angle structures, despite the relatively small tumor volume, has resulted in liver metastases in 25% of patients over a mean follow-up of 6 years (5).
Observed growth of iris lesions may be the best indicator of malignant potential. However, one study of 175 patients with melanocytic tumors of the iris, followed for 1 to 12 years, reported only a 4.6% incidence of tumor growth, and the growth was not always indicative of the presence of malignant cells (37). This low incidence may in part reflect the slow growth of iris melanomas in some patients. In one case report, an iris melanoma was followed over 41 years before producing glaucoma and resulting in enucleation (38). Continued close observation is essential in managing these patients.
Management
Considering the poor prognosis in cases of ciliary body or choroidal melanoma associated with glaucoma, the recommended management most often is enucleation, although fine-needle aspiration biopsy can be considered first to confirm the presence of malignancy if deemed appropriate. By the time glaucoma has developed, the melanoma is usually too large or diffuse for local treatment. However, there are exceptions, especially when the eye with the melanoma is the patient’s only eye with useful vision. In such cases, local excision of an anterior uveal melanoma or radiotherapy of anterior or posterior tumors is occasionally attempted. In one series of 52 patients undergoing iridocyclectomy for lesions of the iris or ciliary body, one half of the excised lesions were benign and almost one third of the group eventually required enucleation (39). Radiotherapy, especially with anterior tumors, may be complicated by hemorrhage and further pressure elevation, as well as failure to eradicate the melanoma. Palladium-103 radiation therapy has been associated with fewer sight-limiting complications than iodine-125 and with excellent local tumor control (40).
Care must be taken to avoid artificial elevation of the IOP during diagnostic and surgical maneuvers for fear that this may accelerate extraocular dissemination of tumor cells (41–43). Enucleation techniques have been developed to minimize intraoperative pressure elevation, which include manometric pressure-regulating systems and the use of a wire snare to cut the optic nerve (42–44).
Iris melanoma and glaucoma are usually managed more conservatively because the tumors are typically small when first detected and can be observed for evidence of growth. However, iris melanomas may metastasize, and the degree to which associated glaucoma increases this risk is uncertain. In general, iris melanomas should be documented by photography (and UBM, when practical) and followed for evidence of growth. If slight growth is detected, continued close observation is still reasonable, whereas pronounced and progressive growth requires surgical intervention (45). This usually consists of complete excision by a sector iridectomy, although photocoagulation may be used to eradicate some iris melanomas (46). However, if the tumor has disseminated or diffusely extended in the anterior segment, which is often the case when associated glaucoma is present, enucleation is usually indicated.
The management of the associated glaucoma in eyes with a uveal melanoma is best limited to medical therapy. Filtering surgery should be avoided, because seeding of iris melanoma cells through a trabeculectomy site into the filtering bleb with extraocular dissemination and fatal metastases have been documented in such cases (37,47). When additional intervention for the glaucoma is required, especially in eyes with iris melanomas, an ab externo cyclodestructive procedure may be the procedure of choice.
SYSTEMIC MALIGNANCIES
Metastatic Carcinomas
Two large studies have been reported in which at least one eye from autopsy cases with known malignancies was examined for ocular metastases (48,49). The most common primary sites for metastasis to the eye were the lung and breast. The two studies reported similar results regarding the incidence of ocular metastases from the lung (6% and 6.7%), although the results differed for metastases from the breast (37% and 9.7%) and the overall incidence of metastasis from all primary carcinomas (12% and 4%) (39,48–50). Metastatic carcinoma of the eye is reported to be the most common form of intraocular malignancy (51).
The most common site of ocular metastasis is the posterior uvea, although glaucoma is more often associated with metastases to the anterior segment (1). In one study of 227 cases of carcinoma metastatic to the eye and orbit, glaucoma was detected in 7.5% of the total group and in 56% of the 26 cases with anterior ocular metastasis (52,53). In another series of 256 eyes with uveal metastases, associated IOP elevation was present in 5% of the total group but in 64% and 67% of eyes with iris and ciliary body metastases, respectively (2). The clinical appearance of metastatic carcinomas of the anterior uvea is a gelatinous or translucent mass, which may be a single lesion or multiple nodules on the iris, often associated with rubeosis iridis, iridocyclitis, or hyphema.
Mechanisms of glaucoma in eyes with anterior uveal metastatic carcinoma include obstructions of the trabecular meshwork by sheets of tumor cells or by infiltration with neoplastic tissue. Other mechanisms include angle closure due to compression of the iris from the tumor or by peripheral anterior synechiae. In some cases, ocular signs or symptoms may be the first manifestation of metastatic malignancies.
In the management of metastatic carcinoma to the anterior uvea, paracentesis with aqueous aspiration for cytopathologic examination is often helpful in establishing the diagnosis (Table 21.2). When the cytology is nondiagnostic, a serologic tumor marker (e.g., in the aqueous) has been used to make the diagnosis (54). Treatment of the metastatic carcinoma usually includes radiation therapy and occasional chemotherapy (1). Enucleation is usually reserved for blind, painful eyes. If the associated glaucoma persists, it should be controlled medically whenever possible.
Metastatic Melanomas
Although ocular melanomas usually are primary malignancies, metastatic melanomas of the eye occur and can occasionally cause glaucoma (28). A unique form has been called black hypopyon, in which a disseminated cutaneous malignant melanoma metastasized to the eye, where it became necrotic, possibly in response to immunotherapy or irradiation, resulting in a hypopyon of tumor cells and pigment-laden macrophages with associated glaucoma (55).
Leukemias
In an autopsy survey of 117 eyes of individuals who died of acute or chronic leukemia, the incidence of leukemic infiltrates in the ocular tissues was 28% (49). In a slitlamp study of 39 children with acute leukemia, the same percentage of cases was found to have flare or cells in the anterior chamber (56). In another series of childhood leukemias, the 5-year survival rate for patients with ocular manifestations was 21.4%, compared with 45.7% for those without ophthalmic involvement (57). Although this series began in 1972, when treatment was less effective than it is today (58), these findings emphasize that any child with leukemia and apparent iritis should be regarded as having a relapse, and anterior chamber aspiration and iris biopsy are essential procedures in establishing this diagnosis (58,59). In a series of 135 patients who had fatal leukemia, ocular leukemic infiltration was found in one third of the cases, most often in the choroid (60).
A leukemic infiltration of the anterior ocular segment leads to glaucoma in some cases, which may present in association with hyphema and hypopyon (Table 21.2). These patients more often have acute lymphocytic leukemia. In adults, acute and chronic leukemias may also have ocular involvement, with reported cases manifesting variously as bilateral hypopyon or a massive subretinal hemorrhage with acute angle-closure glaucoma. Aqueous aspirates may need to be studied to confirm that the patient is in relapse. In most cases, after the diagnosis has been established by cytologic examination of the aqueous aspirate, treatment usually includes irradiation and chemotherapy. In one reported case, the glaucoma cleared after washing necrotic tumor cells from the anterior chamber (61).
Other Neoplasias
Lymphomas
An autopsy review of 60 eyes from patients with lymphomas revealed ocular involvement in 4 (6.7%) (49). The anterior ocular segment may be involved, presenting as iridocyclitis, with occasional IOP elevation (62).
Histiocytosis X
This uncommon multisystem disorder, characterized by accumulation of histiocytes in various tissues, includes three clinical subsets: eosinophilic granuloma (i.e., lesions confined to bone), Hand–Schüller–Christian disease (i.e., bone and soft-tissue involvement), and Letterer–Siwe disease (i.e., predominantly soft-tissue involvement in infants). Histiocytosis X can involve the anterior chamber with associated glaucoma (63). This situation is apparently rare, because a study of 76 children with histiocytosis X revealed 18 with orbital involvement but none with intraocular involvement (64).
Multiple Myeloma
Apparent nongranulomatous anterior uveitis with associated glaucoma in a patient with multiple myeloma was found by cytologic examination to represent an infiltrate of neoplastic plasma cells (65). Mechanisms of glaucoma are summarized in Table 21.2.
Myelodysplastic Syndrome
In patients with the myelodysplastic syndrome, hematopoietic precursors of all three cell lines (i.e., erythroid, myeloid, and megakaryocytic lines) are abundant but morphologically abnormal, and 10% to 30% of cases are fatal because of acute blastic transformation of nonlymphocytic leukemia. Cases have been reported in which patients presented with acute angle-closure glaucoma associated with uveal effusion and nonrhegmatogenous retinal detachment and hemorrhage (66,67).
OCULAR TUMORS OF CHILDHOOD
In addition to leukemia and other systemic neoplasia in which the eye may be involved secondarily, there are certain childhood tumors in which the ocular involvement is a primary part of the disorder. These include retinoblastoma, medulloepithelioma, and rhabdomyosarcoma. Juvenile xanthogranuloma, a histiocytic lesion, is included in this category because it can mimic malignancy and also give rise to glaucoma.
Retinoblastoma
Incidence of Glaucoma
Although glaucoma is not commonly recognized clinically in children with retinoblastoma, histopathologic studies suggest that glaucoma is a frequent complication of this disease. One study of 149 eyes found histologic evidence of a glaucoma-inducing mechanism in 50% of the cases, although an elevated IOP had been clinically recorded in only 23% (68). In another series of 303 eyes with retinoblastoma, 17% had documented pressure elevation (2). Glaucoma was found in one study to be the presenting sign in 7% of eyes with retinoblastoma, with leukocoria (i.e., white pupillary reflex) and strabismus being the most common presentations, at 60% and 20%, respectively (69).
Mechanisms of Glaucoma
Neovascularization of the iris is a frequent histopathologic finding in eyes with retinoblastoma and is the most common cause of the associated glaucoma (1). Angiogenesis may be mediated by vascular endothelial growth factor (70). Rubeosis iridis and neovascular glaucoma are frequently overlooked clinically and should be considered in all cases of retinoblastoma. Two additional causes of glaucoma are angle closure due to massive exudative retinal detachment and obstruction of the anterior chamber angle by inflammatory cells or necrotic tumor tissue (2,68). In a review of 1500 patients with retinoblastoma, anterior chamber tumor involvement was seen in 30 cases and was found to indicate a poor prognosis (71). Specular microscopy has demonstrated clusters of retinoblastoma cells on the corneal endothelium as a bright, lacy network of reflections within a dark area (72).
Differential Diagnosis
Conditions that simulate retinoblastoma have been called pseudogliomas and include retinopathy of prematurity, persistent hyperplastic primary vitreous, retinal dysplasia, Coats disease, toxocariasis, and infantile retinal detachment (73,74). Each of these conditions also has a high incidence of rubeosis iridis so that this factor is not helpful in distinguishing retinoblastoma from the pseudogliomas (73,75).
Management
The presence of rubeosis iridis, with or without glaucoma, indicates a worse prognosis for patients with retinoblastoma (75). This is also true when glaucoma is present without iris neovascularization, because it usually indicates a large tumor with angle closure or dissemination of tumor cells into the anterior chamber (71). Although many treatments are available for retinoblastoma, including chemoreduction, laser photocoagulation, thermotherapy, cryotherapy, and plaque radiotherapy (76), enucleation is often indicated when glaucoma mechanisms are present.
Medulloepithelioma
Medulloepithelioma, or diktyoma, is a primary tumor of childhood that arises most often from nonpigmented ciliary epithelium. The clinical appearance is a whitish-gray mass or cyst of the iris or ciliary body. In a study of 56 cases, glaucoma was observed clinically in 26 eyes (77). Histopathologic evidence of glaucoma was seen in 18 cases, 11 of which had rubeosis iridis. Peripheral anterior synechiae and shallow anterior chambers were also commonly observed. In one report, glaucoma was associated with two white flocculi floating in the anterior chamber, delicate iris neovascularization, and a globular ciliary body mass (78). Some medulloepitheliomas are malignant, although the mortality rate is low. Although enucleation may be required for malignant cases, success has been reported with iridocyclectomy, and local excision has been recommended when the tumor is small and well circumscribed (77).
Rhabdomyosarcoma
Rhabdomyosarcoma is the most common malignant orbital tumor of childhood. It is rarely intraocular but has been reported to arise from the iris or ciliary body (79).
Juvenile Xanthogranuloma
Juvenile xanthogranuloma is a benign, self-limiting histiocytic disease of infants and young children, with rare cases occurring in young adults (80). It is characterized by discrete, yellow, papular cutaneous lesions primarily of the head and neck and by salmon-colored to lightly pigmented lesions of the iris. It is usually diagnosed by a biopsy of a skin lesion or an aqueous tap and iris biopsy, the histology of which reveals foamy histiocytes and Touton giant cells. The iris lesions are typically unilateral and may cause spontaneous hyphema. Glaucoma may occur from invasion of the anterior chamber angle with histiocytes or from the hyphema or a secondary uveitis. Treatment for eyes with juvenile xanthogranuloma and associated glaucoma includes topical and subconjunctival corticosteroids and occasional external-beam irradiation. Invasive surgery should be avoided if possible because prognosis is poor (81).
BENIGN TUMORS OF THE ANTERIOR UVEA
In the differential diagnosis of anterior uveal tumors, several benign lesions must be considered. These include nevi, cysts, melanocytoses, melanocytomas, adenomas, and leiomyomas. Glaucoma may be associated with several of these conditions, further complicating diagnosis and treatment. UBM may be a useful adjunct in distinguishing between some malignant and benign lesions.
Nevi of the Iris
One or more nevi on the stromal surface of the iris are a common clinical finding. They are usually recognized as small, discrete, flat, or slightly elevated lesions of variable pigmentation. Some, however, may be confused with melanomas, which has led to unnecessary surgical intervention. In a retrospective clinicopathologic study of 189 lesions of the anterior uvea that were originally diagnosed as melanomas, 80% were reclassified as nevi of several cell types (82). These investigators found no clinical features to distinguish benign from malignant tumors, including diffuse spread or the presence of glaucoma. In another study of melanocytic iris tumors, however, five clinical variables were associated with a higher risk for malignancy: diameter greater than 3 mm, pigment dispersion, prominent tumor vascularity, elevated IOP, and tumor-related ocular symptoms (83). Fluorescein angiography of the iris, aqueous aspiration for cytologic examination, or biopsy may aid in the important differentiation between melanomas and benign lesions of the iris. Diffuse, pigmented, and nonpigmented nevi of the iris can cause glaucoma by direct extension across the trabecular meshwork (82,84). This is rare in children, but the case of a 16-year-old girl has been reported in which an aggressive iris nevus caused glaucoma by invading the trabecular meshwork (85).
A specific form of iris nevus with associated glaucoma is the iris nevus syndrome. In these cases, diffuse nevi of the iris are associated with progressive synechial closure of the angle and subsequent IOP elevation (86). A subset of the iridocorneal endothelial syndrome, the Cogan–Reese syndrome, has a similar clinical appearance, but the pedunculated nodules on the surface of the iris are composed of tissue resembling iris stroma (87). The benign lesions of the iris in both of these conditions have been mistaken for malignant melanomas, which has led to enucleation in some patients. (These conditions are discussed further in Chapter 16.)
Cysts
Cysts of the iris are classified as primary and secondary, with the former arising from the epithelial layers of the iris and ciliary body or, less often, from the iris stroma (88). Most primary cysts are stationary lesions, rarely progressing or causing visual complications. However, families have been described in which multiple cysts of the iris and ciliary body, presumably of autosomal dominant inheritance, caused angle-closure glaucoma (89). Other reported mechanisms of IOP elevation associated with iris cysts include pigment dispersion (90) and a mucus-producing epithelial cyst of the iris stroma of unknown origin (91). Iris cysts have been successfully treated by laser cystotomy, although others may require surgical excision if uncontrolled IOP or other symptoms persist.
Secondary cysts of the iris may result from surgery, trauma, or neoplasia and are more likely than primary cysts to lead to inflammation and glaucoma (88,92). Ultrasonography has been a useful diagnostic adjunct for primary and secondary iris cysts.
Melanocytomas
These tumors are classified as benign nevi and are seen clinically as darkly pigmented lesions, usually on the optic nerve head and, less often, in the choroid, ciliary body, or iris. Tumors in the latter location can cause glaucoma by direct spread into the anterior chamber angle or by dispersion of pigment into the angle from a necrotic melanocytoma (93,94).
Melanoses
Melanosis iridis is characterized by verrucous-like elevations on the surface of a darkly pigmented, velvety iris. It is usually unilateral and sometimes sectorial, although bilateral involvement has been reported (95). Melanosis oculi has additional hyperpigmentation of the episclera, choroid, or both and has been reported in association with open-angle glaucoma in which the mechanism appears to be heavy pigmentation of the trabecular meshwork (96). A similar condition has been observed in oculodermal melanocytosis (nevus of Ota, which is considered later in this chapter).
Adenomas
Benign adenomas may arise from the epithelium of the anterior uvea, especially the ciliary body (Fuchs adenoma) (97). They occur predominantly in adults, although they have been observed in a child with associated hyperplastic primary vitreous (98). Although common among the elderly, adenomas are rarely observed clinically (99). Some can involve the iris primarily or secondarily and have caused glaucoma from pigment dispersion (100,101). Adenomas must be distinguished from anterior uveal cysts and melanomas (100). Adenocarcinomas may also arise from the ciliary body epithelium and may cause glaucoma (102).
Leiomyomas
These rare tumors may appear as a slow-growing grayish-white, vascularized nodule on the surface of the iris. Glaucoma is not a typical complication.
PHAKOMATOSES
In 1932, Van der Hoeve (103) coined the term phakomatosis, meaning “mother spot” or “birthmark,” to denote a group of disorders that are characterized by hamartomas, which are congenital tumors arising from tissue that is normally found in the involved area. The hamartomas primarily involve the eye, skin, and nervous system, although other systems may be involved to a lesser degree, including pulmonary, cardiovascular, gastrointestinal, renal, and skeletal. In some cases, the anomalies are present at birth, whereas others become manifest later in life. The four conditions that traditionally comprise the phakomatoses are the von Hippel–Lindau syndrome, von Recklinghausen neurofibromatosis, tuberous sclerosis or Bourneville disease, and the Sturge–Weber syndrome. Several other disorders subsequently have been included by various investigators. The following discussion is limited to phakomatoses that frequently or occasionally are associated with glaucoma.
Sturge–Weber Syndrome
General Features
The hamartoma occurring in the Sturge–Weber syndrome (i.e., encephalotrigeminal angiomatosis) arises from vascular tissue and produces a characteristic port-wine hemangioma of the skin along the trigeminal distribution (Fig. 21.7) and an ipsilateral leptomeningeal angioma. The angiomata are present at birth and are usually unilateral, although bilateral cases also occur. In one review of 51 patients, the condition was recognized before 24 months of age in more than half the cases (104). The nervous system involvement frequently causes seizure disorders, hemispheric motor or sensory defects, and intellectual deficiency. A characteristic radiographic finding is cortical calcifications that develop after several years and appear as double densities or “railroad tracks.” There is no race or sex predilection, and no hereditary pattern has been established.
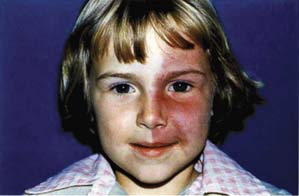
Figure 21.7 Child with the Sturge–Weber syndrome and unilateral glaucoma. Notice the typical port-wine hemangioma of the skin along the distribution of the left trigeminal nerve.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


