20
Surgical Management of
Complications Related to Uveitis
James E. Kempton, John J. Huang, Paul A. Gaudio, and James C. Tsai
SURGICAL MANAGEMENT OF ANTERIOR SEGMENT COMPLICATIONS OF UVEITIS
Band Keratopathy (Calcium Hydroxyapatite Deposition)
Band keratopathy (BK) is the name given to a ground-glass–like opacity that forms in the interpalpebral fissure region of the cornea. The opacity is caused by calcium deposition at the level of Bowman membrane and anterior stroma of the cornea. It forms from calcium salts that are naturally present in the tear film. BK forms in the setting of chronic uveitis, chronic interstitial keratitis, silicone oil (used as a vitreous substitute in retinal detachment), hypercalcemia, and chronic mercury exposure. In addition, a primary familial form exists.
Clinical Findings
The opacity begins forming deep to the epithelium in the peripheral cornea at roughly the 4 and 8 o’clock positions, separated from the limbus by a clear space. With time, the haze will spread horizontally from these positions, eventually forming a band across the interpalpebral fissure (Fig. 20.1). In the early stages, while the band is flat and subepithelial, BK is asymptomatic. If the process continues, the band may thicken and form a discernible plaque with a rough contour that may cause epithelial breakdown and discomfort, and in this setting treatment is indicated.
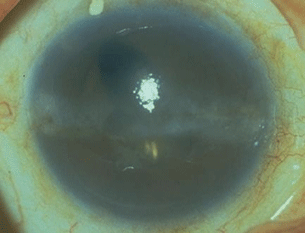
Figure 20.1 Patient with chronic anterior uveitis with corneal decompensation with extensive BK in the palpebral margin.
Treatment
The patient’s inflammation should be well controlled with minimal corticosteroid therapy before performing band chelation.
Chemical Removal
We prefer chemical removal using ethylenediaminetetraacetic acid (EDTA) chelation with superficial keratectomy. The steps are as follows:
- Obtain EDTA 0.35% (3.5 mg/mL—in fact, the concentration used varies widely between practitioners, so many other concentrations will probably work fine). Warm it by placing the EDTA container in a cup of very hot water.
- Anesthetize the eye as for a superficial keratectomy (general, local injection or topical depending on the personality and age of the patient).
- Scrape the epithelium overlying the BK with a no. 15 Bard-Parker blade.
- One needs to have a way to create a well of EDTA over the central cornea. A variety of makeshift cylindrical devices will suffice. We typically use a dull or upside down corneal trephine, while some practitioners use an amputated syringe. Microsurgical sponges may work as a way of keeping EDTA suspended over the cornea, but seem to require longer time.
- The EDTA is placed into the well and held in position for 5 minutes.
- The well is removed and the eye is vigorously irrigated with balanced salt solution.
- The knife is then used to scrape the loosened flakes of calcium. In mild BK, one or two treatments will suffice to achieve a clear stroma. In severe BK, multiple treatments may be needed.
- The procedure is repeated until all of the calcium has been completely removed.
- Cycloplegic and antibiotic medications are then instilled and the eye is patched overnight. We instruct patients to remove the patch the next day, and to start antibiotic eyedrops four times per day until the epithelium is healed.
- The epithelium will generally repopulate the corneal surface within 1 week.
- The procedure is repeated until all of the calcium has been completely removed.
Phototherapeutic Keratectomy
BK can be removed via phototherapeutic keratectomy using an excimer laser, thus avoiding the need for EDTA and scraping the cornea. This obviously requires easy access to an excimer laser, and hence is less commonly used.
CATARACTS
Background
The formation of cataracts is a common complication of uveitis, occurring as a complication of both the disease process and the corticosteroids used to treat it. The cataracts take various forms, from diffuse milky haze to limited posterior capsular opacification (Fig. 20.2). Posterior synechiae are believed to foster cataract formation by causing ischemia of the underlying lens capsule. The mechanism for corticosteroid-induced cataract formation is poorly understood.
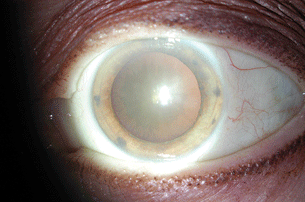
Figure 20.2 Posterior subcapsular cataract formation in a patient with chronic HLA-B27–associated anterior uveitis and topical corticosteroid use.
Goal of Therapy
Good vision is the overarching goal of ophthalmologic care, and so the treatment of cataract is an important part of the care of uveitis patients. In deciding to proceed with cataract extraction in uveitis patients, we apply more or less the same standard of vision as we to do nonuveitis patients: the patient is bothered by decreased vision that we believe is attributable to cataract, and is not treatable with spectacles.
Intraocular Lenses (IOL)
Historically, uveitis patients were considered poor candidates for IOL placement, because early design IOLs tended to incite recalcitrant inflammation, often with disastrous outcomes. Currently, due to improvements in IOL design and in the ability to control intraocular inflammation, most patients undergoing cataract surgery can (and should) undergo IOL placement at the time of surgery. Exceptions exist, and these vary considerably among practitioners. In our view
Combined Pars Plana Vitrectomy (PPV) with Cataract Surgery
We often perform PPV at the time of cataract surgery in patients with uveitis when the involvement of the posterior segment is anything but trivial—roughly 1+ vitreous cell or more, which includes many patients with iridocyclitis and almost all with intermediate, diffuse, or posterior uveitis. Our rationale along these lines is anecdotal (as is so often the case in uveitis therapy), but we do observe the following:
- Many patients’ uveitis will be easier to control postoperatively if PPV is performed primarily at the time of cataract surgery/IOL implantation. It postulated that a vitrectomized eye exposes the posterior segment to anterior chamber associated immune deviation, a phenomenon in which antigen presentation in the eye is aberrant, possibly creating an overall downregulation of the inflammatory response. There is no way to know who these patients are in advance, but we find it helpful often enough that the risk/benefit ratio is, in our view, in favor of PPV for patients with vitreous cells.
- For patients with vitreitis of any degree, it will definitely be easier to monitor their inflammation postoperatively if the vitreous space is made of liquid, since the cells that one then sees there represent active inflammation much like in the anterior chamber, rather than trapped vitreous debris from remote prior uveitic episodes.
- Patients do benefit from the improved media clarity that PPV allows, both from the standpoint of their vision and from our ability to examine their fundus.
- Being aphakic as well as vitrectomized has distinct advantages from the standpoint of inflammatory control, since it is almost always easier to control inflammation in eyes that have no lens; a liquid vitreous space; and a single, unicameral chamber. For this reason, we perform PPV and leave an open posterior capsule for all patients whom we are rendering aphakic out of concern for disease control.
- In general, 23 or 25 gauge PPV is well suited to this combined cataract surgery/PPV. Our approach is to place the 23 or 25 gauge cannulas before beginning the phacoemulsification. With the lens removed, one can perform a core PPV before or after placing the IOL. Certainly, PPV is easier to perform under aphakic optics, but the sequence of phacoemulsification-PPV-IOL placement can be cumbersome, particularly because we generally make a posterior capsular opening with the vitrector at the end of the case, and this is best done after placing the IOL. Thus, we typically place the IOL before performing the PPV unless the latter promises to be complicated.
Controlling Inflammation Before, During, and After Cataract Surgery
The success of cataract surgery in uveitis patients rests on perioperative disease control. Surgery in uveitis patients will induce far more inflammation than is seen in nonuveitis patients, and careful attention to controlling inflammation will minimize this. These are not routine cataract patients, and uveitis practitioners are commonly confronted with the sad sight of patients who were taken to cataract surgery without properly dealing with their disease, and who then developed severe inflammation as a result with devastating consequences for their vision and their eyes. We keep the following in mind.
Disease Course
It is critical that ocular inflammation be inactive for a prolonged period prior to performing cataract surgery, and that one minimizes the amount of inflammation that will result from the procedure. For us, a “prolonged period” nearly always means 3 months. Acknowledging that this time frame is somewhat arbitrary, (rigorous studies have not been performed along these lines) it is an amount of time that we feel should be adequate in the great majority of cases, leaving us the least likelihood of dealing with a marked and recalcitrant inflammatory recurrence postoperatively. (Rare exceptions to the 3-month rule, in our view, are the patient with lens-induced uveitis, and those patients in whom cataract surgery is part of a larger procedure—usually a vitrectomy—being performed to improve control of recalcitrant uveitis.)
Gauging Disease Activity
We consider uveitis inactive if there is one cell per anterior chamber high-power field or less. Flare is unavoidable in many patients with longstanding uveitis, and we do not use it to gauge active disease.
Agents Used to Control Inflammation
Topical, injectable, oral and intravenous corticosteroids all have useful roles in perioperative inflammatory control. Other immunosuppressives are often helpful in achieving long-term control in patients for whom we anticipate performing cataract surgery eventually, but we do not generally start these agents specifically for a surgical procedure. Nonsteroidal anti-inflammatory drugs (NSAIDs) are not, in our view, a useful part of the perioperative regimen.
Factors Determining Our Perioperative Regimen
The way in which we combine anti-inflammatory agents varies according to (a) the location of the patient’s uveitis; (b) how severe the inflammation has been, and; (c) how we have been controlling it in the several months prior to surgery. In the simplest scenario, a patient’s inflammation has been quiescent, (i.e., inactive with no corticosteroids and no immunosuppression). Other patients may have been achieving disease control using immunosuppressives only, corticosteroids only, or corticosteroids with immunosuppressives. In the worst-case scenario, a patient’s inflammation is active at the time of surgery. Specific examples dealing with each of these scenarios are described below.
Surgical Considerations
From the technical standpoint, uveitis-related cataract surgery differs very little from surgery for routine cataracts. The items discussed below pertain to phacoemulsification.
Choice of incisions. We generally use clear cornea incisions but find no problems with scleral tunnel incisions, or with the use of cautery.
Intraoperative materials. No special viscoelastics or intraoperative solutions are necessary or advantageous, in our view.
Posterior synechiae and pupil dilation. In general, lens-iris adhesions are easily broken with any blunt instrument (viscoelastic cannula, Sinskey hooks), and a scarred pupil can be dilated by stretching it manually using Kuglen or collar-button hooks, or with iris hooks if necessary. Pupillary sphincterotomies are reasonable to perform. Very extensive lens-iris adhesions may necessitate cutting the iris off of the lens using retinopathy of prematurity scissors, in effect creating a new pupillary margin. We occasionally cut off smaller iris adhesions that we feel are likely to stick to the lens capsule.
Capsulorrhexis. We generally try to make the capsulorrhexis decidedly larger than for routine cataract surgery, believing that this minimizes the likelihood of posterior synechiae formation. There is no contraindication to the use of trypan blue or indocyanine green (ICG) to stain the anterior lens capsule if the surgeon finds this helpful.
IOLs. We have not found any particular IOL to be preferable to others for uveitis-related cataracts, and we place our IOLs in the capsular bag. Some authors have suggested that ciliary sulcus IOL placement may be helpful in preventing posterior synechiae formation; we have found the disadvantages of this approach to outweigh this possible benefit. As noted above, we avoid IOLs altogether in certain scenarios.
Examples of Regimens to Control Perioperative Inflammation
As noted above, our choice of preoperative, intraoperative, and postoperative anti-inflammatory regimens depends on the location of the patients’ uveitis, how severe a patient’s inflammation has been, and how we have been controlling it prior to booking the surgery. For the purposes of this discussion, we will assume that surgery is booked 3 months prior to the date it is performed. In all scenarios, the inflammation should be inactive at the time of booking. We then consider ways to prevent the inflammation from recurring between the time surgery is booked and the time it is performed, (recurrence of inflammation in this 3-month period necessitates postponing the surgery), and the optimal means of minimizing postoperative inflammation.
Quiescent inflammation
Patients whose inflammation has been quiescent (inactive and requiring no treatment) or controlled with immunosuppressive drugs and no corticosteroids for several months. This is the simplest scenario.
Anterior uveitis, planning phacoemulsification only
Preoperatively: we start topical corticosteroids four times per day to prevent an inflammatory recurrence after we book the case. One week prior to the surgery, we perform an intravitreous triamcinolone 2 mg injection. Topical corticosteroids are used hourly on the day prior to the surgery. This approach greatly simplifies the intraoperative and postoperative regimens: no systemic corticosteroids are needed at any point, and topical corticosteroids four times per day are sufficient for postoperative disease control, with a tapering routine quite like a typical phacoemulsification in a nonuveitic patient. Cycloplegic eyedrops are not necessary.
Intermediate, Diffuse, or Posterior Uveitis, Planning phacoemulsification Combined with PPV
Preoperatively: any time between 12 and 6 weeks, we perform a periocular triamcinolone 20 mg injection, and 1 to 2 weeks preoperatively we repeat this. Topical corticosteroids are used hourly on the day prior to the surgery. Intraoperatively: we inject intravitreous triamcinolone 2 to 4 mg at the close of the procedure. Postoperatively: topical corticosteroids four times per day are sufficient, with a taper based on clinical response. Cycloplegics are not necessary.
Controlled inflammation
Patients whose inflammation has been controlled with corticosteroids (with or without immunosuppressive drugs in addition) for several months.
Anterior uveitis, planning phacoemulsification only
Preoperatively: we administer a periocular triamcinolone 20 mg injection when booking the case, repeating this 6 weeks preoperatively. One week preoperatively, we perform an intravitreous triamcinolone 2 mg injection. Oral corticosteroids equivalent to prednisone 1 mg/kg/day should be started 3 days before the surgery. Topical corticosteroids are used hourly the day before the procedure. Intraoperatively: methylprednisolone 1,000 mg is given intravenously during the procedure (dexamethasone 200 mg is a reasonable alternative). Postoperatively: topical corticosteroids four times per day are used, to be tapered as tolerated. Oral corticosteroids need not be taken on the day of surgery but are resumed the following day for 3 more days, and can then be discontinued without tapering. Wegenerally use atropine 1% two times per day for 1 week postoperatively.
Intermediate, Diffuse, or Posterior Uveitis, Planning phacoemulsification Combined with PPV
Preoperatively: we administer a periocular triamcinolone 20 mg injection when booking the case, repeating this 6 weeks preoperatively and again 1 week preoperatively. (We generally do not perform intravitreous corticosteroid injections in preparation for a planned vitrectomy.) Oral corticosteroids equivalent to prednisone 1 mg/kg/day should be started 3 days before the surgery. Topical corticosteroids are used hourly the day before the procedure. Intraoperatively: methylprednisolone 1,000 mg (or dexamethasone 200 mg) is given intravenously during the procedure, and triamcinolone 2 to 4 mg is injected intravitreally at the close of the case. Postoperatively: topical corticosteroids four times per day are used, to be tapered as tolerated. Oral corticosteroids need not be taken on the day of surgery but are resumed the next day for 3 more days, and can then be discontinued without tapering. We generally use atropine 1% eyedrops two times per day for 1 week postoperatively.
Special Cases
Patients Whose Inflammation is Active at the Time of Surgery
This is another scenario that one should avoid whenever possible. The typical case is a patient with lens-induced uveitis in whom the control of inflammation and IOP will be impossible without lens removal. We strongly recommend combined phacoemulsification/vitrectomy (anterior or posterior) in these cases, and plan to leave the patient with an aphakic and unicameral eye. One usually books the case at most a few days in advance, and oral corticosteroids equivalent to prednisone 1 mg/kg/day, along with topical corticosteroids hourly, are started as far as possible in advance of the case. Ideally, these patients receive methylprednisolone 1,000 mg IV starting 1 day preoperatively, and again intraoperatively and if possible 1 day postoperatively as well. Intravitreous triamcinolone 4 mg is injected at the close of the case. Postoperatively: oral corticosteroids are started following the final IV methylprednisolone dose, and are continued for an additional 2 weeks before being tapered off over an additional 2 weeks. Hourly topical corticosteroids and atropine 1% two times per day are used for at least 2 weeks postoperatively as well.
Patient with Iris Bombe and Pupillary Block with Intractably High IOP
These patients are usually actively inflamed or have only recently had their inflammation controlled, and we handle them quite like patients with lens-induced uveitis; our approach is quite similar to that described above. As with lens-induced uveitis, we find great benefit to combining phacoemulsification and PPV in this scenario, planning to leave the patient with an aphakic, unicameral eye. We prefer this approach to laser iridotomy or peripheral iridectomy (PI), since these latter options frequently result in more surgical manipulation and often more inflammation ultimately than a single, decisive initial surgical procedure. An IOL can be placed once inflammation has been controlled for 3 months.
Cataract Surgery in Patients with Scleritis
As with patients with uveitis, we require 3 months of disease control before performing cataract surgery in scleritis patients. At the time we book the case, we inject periocular triamcinolone 4 mg in each quadrant of the eye planned for surgery, repeating this 6 weeks preoperatively. Approximately 3 days prior to surgery, we start oral corticosteroids equivalent to 1 mg/kg/day, and topical corticosteroids are taken hourly the day before surgery. Methylprednisolone 1,000 mg IV is given during surgery. The surgery performed is quite like that performed for uveitis patients. Postoperatively, oral corticosteroids are taken for an additional 1 to 2 weeks before being tapered.
Postoperative Fibrin Membrane Formation
Some patients are “fibrin membrane formers” and this finding represents among the worst-case postoperative scenarios in uveitis-related cataract surgery. Typically, the reaction begins 2 to 5 days following the procedure and increases daily, as if feeding on itself. Daily examinations show strands of clear fibrin that gradually transforms into a gray membrane that extent to the wound, across the pupil, or may encase the IOL. Fibrin membranes were far more common before intravitreal corticosteroid injection became common practice. If we see a patient who we believe is developing a fibrin membrane, the options are
- “Hammer” with local corticosteroids, including subconjunctival dexamethasone 4 mg (Chapter 19 describes the use of this agent), intravitreous dexamethasone 0.4 mg in 0.1 mL, intravitreous triamcinolone 4 mg, and oral corticosteroids equivalent to prednisone 1 mg/kg/day.
- Intracameral injection of tissue plasminogen activator 12.5 μg in 0.1 mL in addition to topical and subconjunctival corticosteroids.
In either case, the fibrin membrane should show obvious improvement or at least no worsening within 1 day of therapy, and the therapy can be repeated if the membrane is not markedly improved after 2 days.
Posterior Capsule Opacification in Patients with Uveitis
Nd:YAG laser capsulotomy can be performed in patients with uveitis, but the same caveat applies: inflammation should be controlled for 3 months before performing the laser procedure. Preoperatively and postoperatively, we generally recommend hourly corticosteroid eyedrops for several days, and we may inject subconjunctival dexamethasone 1 mg just after the procedure (Chapter 19 describes the use of this agent). Patients who cannot take hourly corticosteroid eyedrops should receive a periocular triamcinolone 8 to 12 mg injection 1 week or less before the procedure, or oral corticosteroids for 2 days preoperatively and continue for 5 days afterward.
Combined Cataract/Glaucoma Surgery in Patients with Uveitis
Phacoemulsification can be combined with trabeculectomy or tube shunt placement in patients with uveitis. This approach may be disadvantageous; however, since the inflammation resulting from the cataract procedure may compromise the glaucoma procedure. If it is to be performed, perioperative corticosteroids are used as described above. In addition, we keep the capsulorrhexis small in these cases, to minimize the likelihood of the IOL prolapsing out of the capsular bag in the event of a flat anterior chamber postoperatively.
SURGICAL MANAGEMENT OF GLAUCOMA IN THE SETTING OF UVEITIS
The concept of uveitic glaucoma was first introduced by Priestly Smith in 1891. However, the first reported association between uveitis and glaucoma was in 1813 by Joseph Beer. Later, specific types of uveitic glaucoma were described in 1906 by Fuchs (Fuchs heterochromic uveitis) and in 1948 by Posner and Schlossman (glaucomatocyclitic crisis). Secondary glaucoma is seen in at least 10% of patients with uveitis. Prevalence of glaucoma varies with the type of uveitis, age of the patient, chronicity of the disease, and severity of the uveitis. Common causes of increased intraocular pressure (IOP) associated uveitis are inflammation of the trabecular meshwork, clogging of the drainage system due to inflammatory cells and pigment, peripheral anterior synechiae, posterior synechiae, formation of pupillary membranes, and corticosteroid-induced IOP steroid response.
The medical management of uveitic glaucoma is as with other forms of glaucoma; topical IOP-lowering agents are used to lower the IOP. These agents include beta-blockers, alpha 2 agonists, carbonic anhydrase inhibitors (CAIs), and prostaglandin analogs. Prostaglandin analogs (latanoprost, bimatoprost, and travoprost) were avoided for many years in the setting of uveitis, out of suspicion that these agents might facilitate inflammatory pathways and incite ocular inflammation. An increasing amount of clinical experience has not borne out this suspicion, so we do use them, although the specter of potential disease exacerbation generally makes them a second line agent for most practitioners. These agents do appear, however, to induce herpetic keratitis exacerbations.
Corticosteroid-induced IOP elevation merits special mention because it is commonly encountered in uveitis practice. A number of studies suggest that roughly one third of patients will develop this complication some time after 2 weeks of corticosteroid use. Another third will develop it at some point after months to years of corticosteroid use, and almost another third can use corticosteroids with impunity from the IOP standpoint, never developing increased IOP. A very small number (a few percent) of patients will develop severe IOP elevation after a very short exposure (days) to corticosteroids. IOP elevation is the most notable complication of periocular and intraocular corticosteroid injections, and in the former cases, we occasionally remove injected corticosteroid deposits from the subtenon or subconjunctival space. Intraocular corticosteroids will usually dissolve sufficiently soon; hence, removal is not necessary. Corticosteroid-induced IOP elevation will almost always normalize when the incinting corticosteroids dissolve, are stopped, or are removed. However, IOP elevation is a very frequent complication of the intraocular fluocinolone implant, and we counsel these patients accordingly when discussing this intervention.
General Principles
Perioperative inflammatory control is essential, and we apply the same considerations to glaucoma surgery as we do to cataract surgery: uveitis should be inactive for 3 months prior to performing glaucoma surgery. This is generally achievable for most patients, acknowledging that glaucoma surgery is not always elective, and some patients require IOP lowering much sooner. In all cases, the timing of surgery is dependent on interplay between need for acute pressure lowering, given the glaucoma status, the amount of IOP elevation, and the likelihood of surgical success given the extent of inflammation. The most obvious examples of an exception to the “3 month rule” are those with poorly controlled uveitis who develop seclusio pupillae and subsequent iris bombé with total angle closure, and often present with active uveitis and very high IOP. In this setting, there is often little alternative to achieving immediate control of the uveitis with corticosteroids and, if the IOP cannot be controlled medically, proceeding with glaucoma surgery within a matter of days to weeks. Other exceptions present themselves occasionally, but in all cases, one needs to acknowledge—and discuss with the patient—that the likelihood of a postoperative inflammatory exacerbation increases the sooner one proceeds after controlling inflammation; we plan the perioperative regimen accordingly.
Measures for perioperative inflammatory control are the same as those outlined above for phacoemulsification. We make extensive use of periocular, topical, intraocular, and systemic corticosteroids to minimize postoperative inflammation. In administering periocular corticosteroids to patients in whom we plan glaucoma surgery, we try to avoid the superior quadrants, since corticosteroid injections can cause scarring of the conjunctiva, which complicates the surgical procedure. These injections are best given transseptally along the orbital floor, or transconjunctivally in the inferior subtenon space.
We generally avoid lasers in the management of uveitic glaucoma, because anterior segment laser provokes anterior uveitis. While, for nonuveitic patients, laser iridotomy plays an important role in the treatment of narrow angle glaucoma, and laser trabeculoplasty is a useful adjunct therapy in open angle glaucoma, lasering the iris causes some anterior uveitis even in otherwise healthy eyes; in uveitic eyes, the detrimental effect often outweighs the benefit. (Lasering the iris is in fact a means of inducing uveitis for experimental purposes in animals.) The only possible exception may be selective laser trabeculoplasty (SLT), which we have seen used apparently without detrimental effect in eyes with uveitis, but we feel that more experience should be accumulated before embracing the use of SLT in this setting.
Hypotony is a common complication of glaucoma surgery in the setting of uveitis, presumably because these patients’ eyes produce aqueous humor irregularly, and often insufficiently. Thus, one is commonly faced with a patient whose eye or eyes have scarred trabecular meshwork and drainage architecture, yet produce low amounts of aqueous humor. The surgical plan and postoperative expectations need to be modified accordingly, to include the possibilities that filtering or shunt procedures may need to be revised to minimize postoperative hypotony.
The surgical technique for glaucoma surgical procedures in uveitis patients is the same as for patients without uveitis. No modifications of the surgical procedures are needed. As we have noted elsewhere, however, one needs to pay considerable attention to the perioperative regimen to minimize the likelihood of an inflammatory response postoperatively.
Surgical Therapies
For Angle Closure Glaucoma
Our decision-making in this setting depends on the extent of cataract present in the eye, and whether the eye is inflamed. (As noted above, an occasional dilemma in the management of uveitic closed angle glaucoma is the patient whose eye is or was very recently inflamed and now presents with seclusio pupillae and iris bombé, often with very high, recalcitrant IOP.)
If a fair amount of cataract is present, we generally opt for phacoemulsification, and if the uveitis is or was recently active—as in the setting of seclusio pupillae and iris bombé—we leave the eye aphakic and perform PPV at the same time. The perioperative management of this scenario is explained above in Sections 7 and 8 of the cataract surgery portion of this chapter.
If the lens is clear, with or without active inflammation, our preferred approach is usually a surgical PI, (as noted above, we avoid laser PIs because they tend to incite or worsen inflammation, and quite frequently tend to close in eyes that are actively inflamed). Our perioperative management for a surgical PI is as for phacoemulsification, as described above in paragraph 7-b-i in the cataract portion of this chapter, with the possible exception that we might forego intravitreous triamcinolone injection if the uveitis has been well controlled prior to the PI procedure.
Usually, IOP will improve if the angle can be reformed, even if iris bombé was present. Many patients with uveitis will, however, eventually require filtering or tube shunt surgery (see below) at a later date.
Open Angle Glaucoma
When medical therapy is inadequate for the treatment of uveitic open angle glaucoma, we face a choice between trabeculectomy and tube shunt placement. There are few hard and fast rules in this common scenario, although without question most specialists are leaning increasingly toward tube shunt procedures in the setting of uveitic glaucoma.
Trabeculectomy. We do nevertheless favor trabeculectomy in patients who (a) are not children or young adults and thus are more likely to sustain a bleb without extensive scarring, (b) have excellent control of their inflammation and will probably maintain good control in the future (e.g., patients on very effective immunosuppressive regimens or who have a fluocinolone intraocular implant in the affected eye), (c) have an overall level of understanding and emotional resilience to undergo postoperative revisions if necessary, and (d) require very low IOPs postoperatively (11 or less). We include mitomycin C in all cases of trabeculectomy in uveitis patients, and while this has the minor disadvantage of some limited toxic exposure, it has the advantage of avoiding the placement of hardware on the eye, thus avoiding the risk of hardware-related complications like exposure and infection. Our perioperative regimen for trabeculectomy is the same as for phacoemulsification, as described above in this chapter.
Tube shunt. We favor tube shunt placement in patients who (a) are children or young adults, (b) seem likely to experience recurrence of their inflammation requiring therapeutic modifications at some point in the not distant future, (c) seem like they would have difficulty undergoing postoperative revisions of their glaucoma surgery, and (d) are undergoing PPV at the same time as their glaucoma surgery or are likely to need it in the near future. Tube shunts have the disadvantage of being hardware on the eye, thus incurring a risk of exposure and infection, but have the advantage of being less likely than a trabeculectomy to be compromised by scarring (Fig. 20.3).
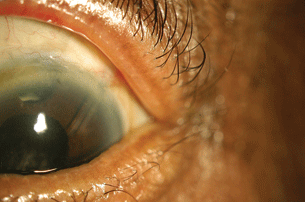
Figure 20.3 Superotemporal Ahmed valve in a patient with uveitic glaucoma.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


