2
The Immuno-inflammatory Cascade
John J. Huang and Benjamin Erickson
To effectively manage and treat systemic and ocular autoimmune disorders, a thorough understanding of the immune-inflammatory system is essential. Mediators of immunity and inflammation are part of the intracellular and intercellular signaling that allows cells to respond based on the past and present antigen exposures. Abnormal regulation of this highly refined system leads to autoimmunity and excessive inflammatory response. There are two components to the immune-inflammatory response: the innate immune system and the adaptive immune system. Acute management of complicated autoimmune disorders often requires down-regulation of the inflammatory cells and mediators that are involved in these integrated systems. Minimizing an excessive healing response is particularly crucial to the long term management of uveitis.
IMMUNE-INFLAMMATORY RESPONSE
Innate Immune System
The innate immune system works in a nonspecific manner through direct cellular phagocytosis and destruction of invading microbes by macrophages, mast cells, and neutrophils. The mere presence of a foreign antigen is all that is required to engage the innate immune response, which is not improved by any sort of “memory” of a prior encounter with an antigen. Evolution has provided our innate immune system a repertoire of bacterial, viral and foreign antigens that are immediately recognized upon exposure. In this regard, it is different from the adaptive immune response.
Adaptive Immune System
In contrast to the innate immune response, the adaptive immune response does improve based on immunologic “memory” of a prior encounter with an antigen. This encounter is highly dependent on antigen presentation and also cell activation, cell differentiation, and cell memory. This cell memory confers long-lasting immunologic protection unlike the innate mechanism. Repeat exposure to the same antigen leads to a more rapid and effective immune response. The cells responsible for the adaptive system are antigen-presenting cells (APC) and B and T lymphocytes. Macrophages, dendritic cells, and B lymphocytes are equipped with special surface receptors and are also known as professional APC.
The adaptive system is triggered when APC engulf and internalize an invading microbe. The microbe is processed into antigen fragments, which are presented on the cell surface as part of a molecule known as the Human Leukocyte Antigen, or HLA. The antigen fragment occupies a specific groove in the HLA molecule, which makes contact with a T-cell to begin the process of T-cell activation. The HLA molecule is critical to the process and specific to each individual; APCs of one individual can not activate T-cells of another unless both individuals share the same HLA makeup, or haplotype. There are 2 classes of HLA molecules, termed class I and class II (discussed below), and each class activates different types of T-cells.
HLA II cells interact with T-cells bearing the cell surface marker CD 4, and these activated CD4+ cells are termed “helper” T lymphocytes. The role of helper T lymphocytes is to tightly regulate the adaptive immune response by controlling the activation of the antigen specific cytotoxic T and B lymphocytes. Upon activation by the APC, the helper T lymphocytes produce interleukin-2 (IL-2), a potent lymphocyte mitogen, which leads to a cascade of exponential magnification of the immune response (Fig. 2.1).
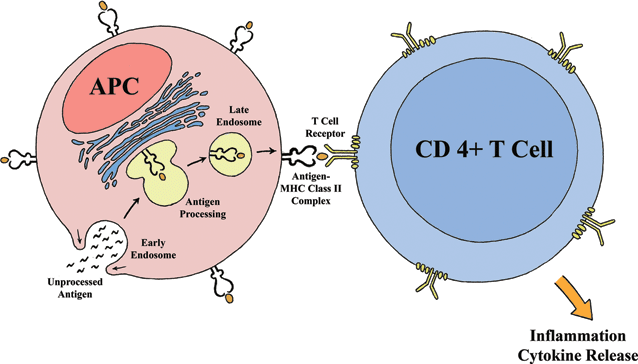
Figure 2.1 APC engulfing microbial antigens which are further processed intracellularly and presented on a MHC II cell surface epitope to a CD4+ T cell. The cellular interaction leads to maturation of the T lymphocyte and further immune activation.
The main function of CD8+ T cells is to induce cell death in viral-infected and tumor cells carrying foreign or abnormal epitopes on their cell surface. This activation is also tightly controlled by helper T cells and APC through the binding of the epitope on the HLA class I surface receptor. The interaction between these cells is managed by chemical mediators called cytokines and through direct cell-to-cell receptor interaction. Cytotoxic T lymphocyte-mediated cell death is achieved through target cell lysis or programmed cell death (apoptosis).
The maturation and differentiation of B cells into plasma cells is also tightly controlled by interactions with activated helper T cells. B cells begin as naïve lymphocytes with cell surface IgM and IgD serving as antigen receptors. After proper stimulation of the B-cell antigen receptor by the activated helper T cell, B lymphocytes are activated and ultimately differentiate into antibody producing plasma cells. Plasma cells are short lived (2–3 days) and secrete antibodies of any one type, specific to a single antigen (Table 2.1). These antibodies are released into the circulatory system, targeting their specific antigens and forming antigen-antibody complexes for the activation of the complement cascade or further processing in the liver and spleen.
Table 2.1 Characteristic of human antibodies
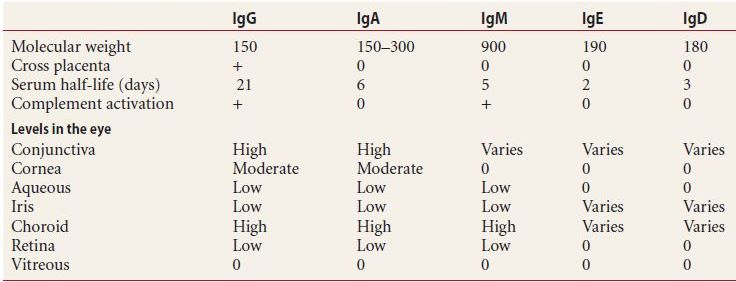
Ig, immunoglobulin.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


