Pigmentary and Other Glaucomas Associated with Disorders of the Iris and Ciliary Body
IRIS OR CILIARY BODY DISORDERS WITH ASSOCIATED GLAUCOMA
There are several conditions in which a disorder of the iris or ciliary body is believed to be involved in the initial events that eventually lead to various forms of glaucoma (Table 17.1). Most of these, such as developmental disorders, inflammatory conditions, and intraocular tumors, are considered in other chapters in Section II. In this chapter, we consider additional conditions—pigmentary glaucoma, iridoschisis, plateau iris, pseudoplateau iris, and swelling of the ciliary body—that do not fit precisely into any of these general systems of disease.
PIGMENTARY GLAUCOMA
Terminology
As a normal feature of maturation and aging, a variable amount of uveal pigment is chronically released and dispersed into the anterior ocular segment. This is best appreciated by observing the trabecular meshwork, which is nonpigmented in the infant eye but becomes progressively pigmented to various degrees with the passage of years because of the accumulation of the dispersed pigment in the aqueous outflow system. There is therefore a spectrum of ocular pigment dispersion in the general population. As can be anticipated, this spectrum of pigment dispersion is also found among individuals with various forms of glaucoma, although the pigment in most of these cases is not believed to be a major factor in the mechanism of the glaucoma. Several ocular conditions are associated with an unusually heavy dispersion of pigment, which may be significantly involved in the increased resistance to aqueous outflow. In 1940, Sugar (1) briefly described one such case with marked pigment dispersion and glaucoma. In 1949, Sugar and Barbour (2) reported the details of this entity, which differed from other forms of pigment dispersion by typical clinical and histopathologic features. They referred to the condition as pigmentary glaucoma (2). When the typical findings are encountered without associated glaucoma, the term pigment dispersion syndrome (PDS) has been advocated (3).
General Features
The typical patient is young, myopic, and male. The disorder appears most frequently in the third decade of life, and there is a tendency for it to decrease in severity or disappear in later life (4,5). Most studies agree that PDS is more common among men, with a male-to-female ratio of approximately 2:1, although studies differ on the ratio of those converting to pigmentary glaucoma (3–11). The reason for the male predilection appears to be the sex difference in anterior chamber depth, which one study showed to be 3.22 ± 0.42 mm in men and 2.88 ± 0.38 mm in women (12). (The significance of chamber depth in the mechanism of pigment dispersion is discussed later in this chapter.)
Pigmentary glaucoma is seen predominantly in whites (3–11), although it may be more common among blacks than previously recognized (13). In black patients, signs of PDS may be overlooked because dark, thick iris stroma may obscure transillumination defects and pigment granules on the stroma; corneal endothelial pigmentation may be minimal or absent; and greater degrees of trabecular meshwork pigmentation may be interpreted as normal in black patients. It has been suggested that pigment accumulation on the lens zonules and equatorial or posterior lens regions may be particularly helpful in making the diagnosis of PDS in black patients (13). In one report, 20 black patients with presumed PDS had heavy pigment deposition on the corneal endothelium and trabecular meshwork. These patients differed clinically from that described in the white population in that their mean age was 73 years, and most were hyperopic and lacked iris transillumination defects on slitlamp examination (14). One report described iridocorneal angle anomalies in a group of black probands with presumed PDS and among their first-degree relatives (15). A hereditary basis has been suggested for the classic form of PDS (see Chapter 8).
Clinical Features
Slitlamp Biomicroscopic Findings
Corneal Findings
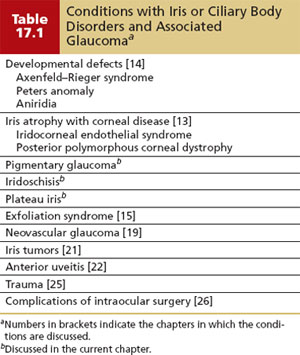
Pigment dispersion occurs throughout the anterior ocular segment but is seen by slitlamp examination primarily on the cornea and iris. Krukenberg spindle is an accumulation of pigment on the posterior surface of the central cornea in a vertical, spindle-shaped pattern (Fig. 17.1). Dispersed pigment is deposited on the cornea in this pattern because of aqueous convection currents and is then phagocytosed by adjacent endothelial cells (3). This feature is commonly seen in eyes with pigmentary glaucoma, but it is neither invariable nor pathognomonic of the disorder. In one study, only 2 of 43 patients with Krukenberg spindles developed field loss during a follow-up that averaged 5.8 years (16). Krukenberg spindle is more common in women and may have a hormonal relationship (16,17). Specular microscopy of the corneal endothelium reveals distinct pleomorphism and polymegathism (i.e., abnormality in shape and size of cells); however, normal cell counts and central corneal thickness are found (18,19).
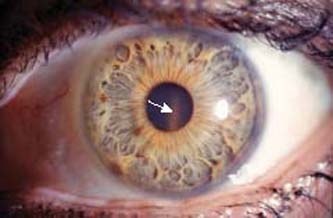
Figure 17.1 Krukenberg spindle (arrow) in a patient with pigmentary glaucoma. (Courtesy of Ralf R. Buhrmann, MD, PhD.)
Iris Findings
Iris transillumination is a valuable diagnostic clinical feature of pigmentary glaucoma because it represents areas where pigment has been dispersed. The characteristic appearance is a radial spoke-like pattern in the midperiphery of the iris (20) (Fig. 17.2). This feature can be seen during slitlamp biomicroscopy by directing the light beam through the pupil perpendicular to the plane of the iris or by using scleral transillumination and observing the retinal light reflex through the defects in the iris. In some patients, however, a dark, thick iris stroma may prevent transillumination of the defects, and the absence of this finding therefore does not rule out the diagnosis of pigmentary glaucoma. This could explain the absence of iris transillumination in black patients with pigmentary glaucoma (14). An infrared videographic technique has been developed that allows visualization of discrete iris transillumination defects not visible by slitlamp examination (21).
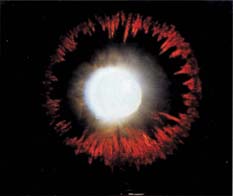
Figure 17.2 Transillumination of the iris in a patient with pigmentary glaucoma showing typical midperipheral spoke-like defects.
Pigment granules are frequently dispersed on the stroma of the iris, which may give the iris a progressively darker appearance or create heterochromia in asymmetric cases (9) (Fig. 17.3). Asymmetric pigment dispersion has also been reported in association with unilateral cataract formation or extraction (22). Patients with PDS may also have anisocoria, in which the eye with the larger pupil is on the side with the greater iris transillumination. The iris heterochromia and anisocoria of PDS may mimic Horner syndrome (23).
Figure 17.3 Pigment granules on iris stroma in a patient with pigmentary glaucoma.
Other anterior segment locations where pigment dispersion may be seen by slitlamp examination include the posterior lens capsule (Fig. 17.4), lens zonules, and the interior of a glaucoma filtering bleb (9).
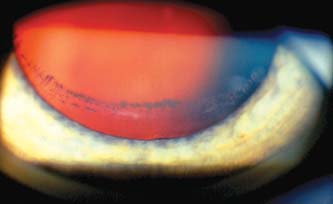
Figure 17.4 Pigmented line on posterior lens capsule at the insertion of zonules. This is termed the Zentmayer line or a Scheie stripe.
Gonioscopic Findings
The principal gonioscopic feature of pigmentary glaucoma is a dense, homogeneous band of dark brown pigment in the full circumference of the trabecular meshwork (Fig. 17.5A). The dispersed pigment may also accumulate along the Schwalbe line, especially inferiorly, creating a thin, dark band.
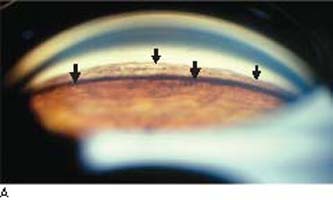
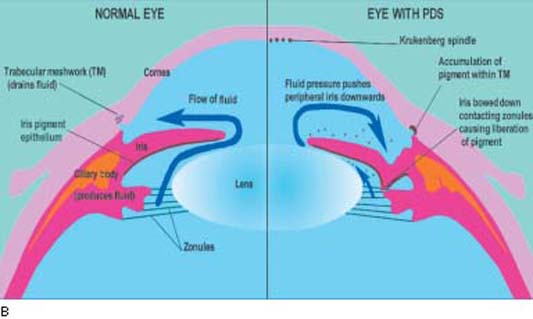
Figure 17.5 A: Gonioscopic view of patient with pigmentary glaucoma showing typical features of wide open angle with dense, homogenous pigmentation of trabecular meshwork (large arrows) and heavy pigment accumulation on the Schwalbe line (small arrows). B: Schematic diagram illustrating a cross section of a normal eye (left half) and an eye with PDS (right half). A normal anterior segment is shown on the left and illustrates normal flow of aqueous humor (curved arrow). Note the flat iris configuration and ample space between the iris and lens for fluid flow. On the right side, note that the iris is touching the lens near the pupil and only permits anterior flow of aqueous. This leads to a higher pressure anterior to the iris, causing backward bowing (concavity) of the peripheral iris. The iris pigment epithelium is thus brought into contact with the zonules and tips of the ciliary processes. This leads to cell damage and liberation of pigment that circulates with the aqueous humor into the anterior chamber, leading to the characteristic signs of PDS.
Fundus Findings
Retinal detachments are more common in patients with PDS or pigmentary glaucoma (24), occurring in 6.4% of patients in one study (11). A study of 60 patients with pigment dispersion or pigmentary glaucoma revealed lattice degeneration in 12 patients (20%) and full-thickness retinal breaks in 7 patients (12%) (25).
Clinical Course of the Glaucoma
Patients with PDS may go for years before developing pigmentary glaucoma or may never have a rise in intraocular pressure (IOP). In one study of 97 eyes with pigment dispersion throughout the anterior ocular segment, glaucoma was present in 42 (26), whereas in another study of 407 patients with the dispersion syndrome, only one fourth had glaucoma (11). In a long-term study that spanned 5 to 35 years, 13 of 37 patients with PDS (35%) converted to pigmentary glaucoma (6). The glaucoma usually develops within 15 years of the presentation of the PDS, although some may take more than 20 years (6). In another study, with follow-up averaging 27 months, progression of the iris transillumination defects and pigment dispersion could be documented in 31 of 55 patients and correlated with worsening of the glaucoma in most of these (27). A study of 111 patients with PDS or pigmentary glaucoma identified male sex, black race, high myopia, and Krukenberg spindles as risk factors for the development and severity of glaucoma in this population (10). However, another study found that sex did not influence the development or severity of glaucoma among patients with PDS (6).
In some patients, strenuous exercise, such as jogging, or spontaneous changes in the pupillary diameter may be associated with marked pigment release into the anterior chamber, although this does not appear to significantly elevate the IOP in most cases (28). In patients in whom exercise-induced pigment dispersion causes a significant IOP rise, use of pilocarpine has effectively inhibited this phenomenon. Phenylephrine-induced mydriasis also causes a significant shower of pigment into the anterior chamber in some patients with pigmentary glaucoma or PDS, although this transient liberation of pigment is not consistently associated with IOP elevations (28).
After pigmentary glaucoma becomes established, it may be somewhat more difficult to control than chronic open-angle glaucoma (COAG) is. In one long-term follow-up study of 38 patients (75 eyes), 39 eyes were controlled medically, 15 required laser trabeculoplasty, and 20 underwent a trabeculectomy (29). However, 67 eyes (89%) retained normal vision during the study (mean follow-up, 10 years). With increasing age, there is a tendency for the glaucoma to become less severe, but the condition must be treated aggressively during the active years to avoid irreversible loss of vision in later life.
Theories of Mechanism
Two fundamental questions must be considered regarding the pathogenesis of PDS and the mechanism of pigmentary glaucoma. What are the factors leading to the pigment dispersion and how do the dispersed pigment and additional features cause the glaucoma?
Mechanism of Pigment Dispersion
An inherent weakness or degeneration in the iris pigment epithelium was first proposed as a cause of PDS by Scheie and Fleischauer in 1958 (26). Subsequently, histopathologic observations of the iris in eyes with the PDS or pigmentary glaucoma have revealed changes in the iris pigment epithelium, which include focal atrophy and hypopigmentation, an apparent delay in melanogenesis, and hyperplasia of the dilator muscle (26, 30–32). In contrast, eyes with COAG and various degrees of pigment dispersion had minimal hypopigmentation of the iris epithelium with normal dilator muscle and melanogenesis (32). These observations have led some observers to think that a developmental abnormality of the iris pigment epithelium is the fundamental defect in PDS (30–32). The additional observation of retinal pigment epithelial dystrophy in two brothers with pigmentary glaucoma raises the possibility of an inherited defect of pigment epithelium in the anterior and posterior ocular segments (33). On the basis of fluorescein angiography of the iris, hypovascularity of the iris may also play a role in PDS (34,35).
Campbell (36) proposed an alternative mechanical theory for the mechanism of pigment liberation from the iris (Fig. 17.5B). He observed that the peripheral radial defects of the iris corresponded in location and number to anterior packets of lens zonules and suggested that a background bowing of the peripheral iris led to the mechanical rubbing of the lens zonules against the iris pigment epithelium with the subsequent dispersion of pigment. This hypothesis was supported by histologic studies showing a correlation between packets of zonules and deep groves in the iris pigment epithelium and posterior stroma (36,37). Sugar (7) suggested that the radial folds of iris pigment epithelium rubbing against the lens capsule itself might be an additional mechanism of pigment release.
The mechanical theory of Campbell is also supported by biometric and photogrammetric studies of anterior chamber dimensions, which revealed deeper anterior chambers and flatter lenses in the involved eyes of unilateral cases and a deeper-than-normal midperipheral chamber depth with corresponding concavity of the iris in eyes with the PDS (38,39). Further support for the mechanical theory comes from studies with ultrasonographic biomicroscopy indicating that the distance between the base of the trabecular meshwork and the point of insertion of the iris is greater in eyes with PDS than in healthy controls. Ultrasonographic biomicroscopy studies in patients with PDS have shown that the radial width of the iris compared with the size of the anterior segment is larger than normal (40). This larger size results in a floppier iris, which may predispose to iridozonular contact when combined with the posterior iris insertion. The mechanical theory is also consistent with clinical observations and helps to explain certain features of the disease. For example, the low incidence of the condition among nonwhite persons may be because of the heavy pigmentation and compactness of the iris stroma in these individuals, which prevents posterior sagging of the midperipheral iris (41). The tendency for the disease to ameliorate with increasing age may be attributed to the increasing axial length of the lens, which pulls the peripheral iris away from the zonules (5,36). A case has also been reported in which subluxation of the lens apparently caused remission of pigmentary glaucoma (42).
The mechanical theory must include an explanation of the mechanism by which the peripheral iris is bowed backward. This missing piece of the puzzle came with the observation that a laser iridotomy relieves the posterior bowing, which led to the concept of reverse pupillary block (43). This concept suggests that aqueous is moved into the anterior chamber against the normal pressure gradient, possibly by the movement of the peripheral iris in response to movement of the eye (e.g., blinking) or accommodation. Once in the anterior chamber, the aqueous is prevented from returning to the posterior chamber by a one-way valve effect between the iris and lens, resulting in a relatively greater pressure in the anterior chamber and subsequent backward bowing of the peripheral iris. The theory of reverse pupillary block has been supported by studies with ultrasound biomicroscopy and Scheimpflug photography, both of which demonstrate the posterior bowing of the iris in patients with PDS and pigmentary glaucoma (28). The posterior bowing is eliminated by a peripheral iridotomy, miotic therapy, or prevention of blinking. Exercise, however, increases the iris concavity (44). The observations regarding blinking and exercise appear to support the concept that eye movement is responsible for the pumping of aqueous into the anterior chamber. It has also been observed that accommodation in patients with PDS leads to increased posterior bowing of the iris, which the investigators explain by the forward movement of the lens, which reduces the volume, thereby increasing the pressure in the anterior chamber (45). Iridotomy abolishes the change in the iris profile that is normally seen with accommodation in patients with PDS (46). The mechanical theory, however, does not fully explain why not all eyes with myopia are subject to PDS, and it may be that additional iris defects (as previously discussed) are necessary for development of this syndrome.
Another mechanism of pigment dispersion involves elongated anterior zonules that may be pigmented (Fig. 17.6A,B), encroaching in the central visual axis (47,48). Normally, the anterior zonular insertion leaves a zonule-free zone of 6.9 mm (49). The clinical appearance and electron microscopic appearance (Fig. 17.6C) of an anterior capsule specimen obtained during cataract surgery suggest a mechanism of pigment release from the pigmented epithelium located at the papillary ruff and the central iris, which are close to the elongated zonules (48). This mechanism appears to be distinct from PDS in which iridozonular contact occurs in the region of the posterior chamber between the midperipheral iris and anterior zonule bundles (36). The zonules appear normal, as standard phacoemulsification did not indicate fragility (48,50). Although subtle and easily missed on biomicroscopy, long anterior zonules may be more common than suspected but not previously recognized as a distinct entity associated with pigment dispersion.
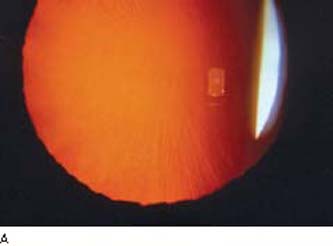
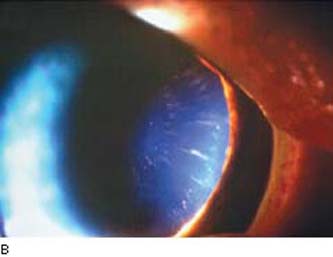

Figure 17.6 Variant of pigment dispersion syndrome in a patient with African ancestry and hyperopia illustrating a distinct mechanism of pigment dispersion. A: Long anterior zonules are visible on the anterior capsule surface following dilation with direct and retroillumination. B: Pigment is commonly noted with some of the long anterior zonules. C: Transmission electron microscopy shows central anterior lens capsule covered by an irregular zonule lamella with pigment granules and degenerative lens epithelium in pigmented long anterior zonules. (Courtesy of Ursula Schlötzer Schrehardt, PhD.)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


