Purpose
To evaluate the correlation between changes in tear osmolarity, symptoms, and corneal fluorescein staining in patients with dry eye disease (DED).
Design
Retrospective, clinic-based cohort study.
Methods
In this single-institution study, we reviewed the charts of 186 patients with DED from whom we had data on tear osmolarity, symptoms, and corneal fluorescein staining from 2 separate visits. Main outcomes included the correlation of the changes between the 2 visits for tear osmolarity (TearLab system), symptoms (Ocular Surface Disease Index), and corneal fluorescein staining (modified Oxford scheme). For tear osmolarity and corneal fluorescein staining the scores from the eye with highest readings were analyzed. The correlations were repeated on subgroups based on proposed cutoffs for DED severity and on patients’ treatment.
Results
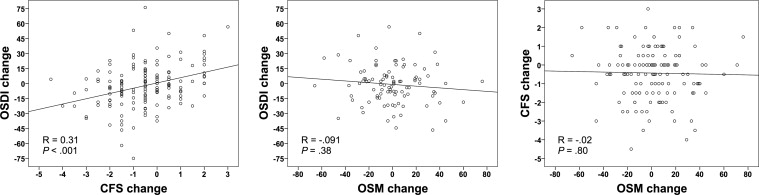
We found a modest, though statistically significant, correlation between changes in corneal fluorescein staining and symptoms of DED ( R = 0.31; P < .001). However, there was no correlation between the recorded change in tear osmolarity and symptoms ( R = −0.091; P = .38) or between changes in tear osmolarity and corneal fluorescein staining ( R = −0.02; P = .80). This lack of correlation was consistent in all the subgroups studied. A multivariate analysis revealed that changes in corneal fluorescein staining had predictive value on symptom changes, whereas tear osmolarity changes did not.
Conclusions
Changes in tear osmolarity do not correlate significantly with changes in patient symptoms or corneal fluorescein staining in dry eye disease.
Dry eye disease (DED) is one of the most prevalent causes of ophthalmic consultation in the developed world. It is reported that 5%-35% of the general population suffers from moderate to severe DED, and the prevalence increases with age. DED presents in patients of all ages and both sexes, although it is more prevalent among older women. Diagnosis and follow-up of DED are complex and are based on a combination of patient-reported symptoms and a series of clinical tests. However, most of these tests have shown a lack of consistency in assessing the disease and are always subject to clinical interpretation. Indeed, none of the available tests used to date for the diagnosis and follow-up of DED have demonstrated very high reliability.
Tear hyperosmolarity is recognized as a significant pathophysiologic factor in the ocular surface disease suffered by patients with DED. Subjects with clinically diagnosed DED present with higher tear osmolarity levels than normal controls, and these levels reportedly increase with the severity of the disease. Until recently, tear osmolarity quantification was restricted to research endeavors given the technical difficulties it involved, such as rapid evaporation of the samples and the need for equipment limited to laboratory facilities. With the advent of the TearLab Osmolarity System (TearLab Corp, San Diego, California, USA), approved by the Food and Drug Administration for assessment of DED in 2009, in-clinic evaluation of tear osmolarity has become readily available. Several clinical studies using the TearLab osmometer have shown that this method of quantifying tear osmolarity is a reliable diagnostic tool for DED. However, in spite of the increasing availability of the osmometer in the clinical setting, ocular surface staining and patient-reported symptoms remain the main criteria used by clinicians and clinical trial experts to assess DED severity and progression. One reason may be that very few data are available to assess the correlation between change in tear osmolarity and other indicators of disease severity, such as patient symptoms or degree of corneal fluorescein staining. Such relationships could be important for assessment of the natural history of DED as well as response to treatment.
In this study, we evaluated changes in corneal fluorescein staining and symptoms among patients with a diagnosis of DED between 2 separate visits, and assessed their correlation with changes in tear osmolarity.
Methods
We conducted a single-center, retrospective study that involved a systematic chart review of patients with DED. In this study, we evaluated the correlation between changes in tear osmolarity and changes in signs and symptoms of DED. This study was conducted at the Cornea Service of the Massachusetts Eye and Ear Infirmary, Boston, Massachusetts, USA, with the approval of the Human Studies Committee, and with adherence to the tenets of the Declaration of Helsinki.
We included patients 18 years and older with a previous diagnosis of DED, who had continued presence of dry eye symptoms and corneal fluorescein staining, and for whom we had data for at least 2 of the following: symptoms, represented by Ocular Surface Disease Index (OSDI; Allergan, Inc, Irvine, California, USA), corneal fluorescein staining, and tear osmolarity (TearLab Osmolarity System; TearLab Corp) from 2 visits. We excluded those with a history of microbial keratitis, atopic eye disease, corneal transplantation, or contact lens use in either eye.
Patients’ symptoms were measured by self-response to the OSDI questionnaire. Tear osmolarity from both eyes was obtained and the higher reading of the 2 eyes was used for the analysis, as recommended by the manufacturer and the labeling of the device. Tear osmolarity acquisition was the first clinical assessment performed during the clinical visit. For this, a trained technician placed the TearLab osmometer probe on the inferior lateral tear meniscus until a signal was heard, and immediately proceeded to analyze the sample. We included cases in which we were able to obtain readings from both eyes, or if the reading from 1 eye was “below range,” to ensure that we obtained the higher reading of both eyes (valid tear osmolarity readings could not be obtained, from 1 or both eyes, in 23 patients). Following the measurement of osmolarity, fluorescein was added to the ocular surface and evaluated after 3 minutes using the modified Oxford scheme. Corneal fluorescein staining from the most severely affected eye was used for analyses (regardless of whether it was the same eye with higher tear osmolarity) to reveal the greatest degree of disease involvement at the time measured. We also correlated the changes among the 3 parameters (from the study’s overall population) using tear osmolarity and corneal fluorescein staining from the same eye: (1) the eye with higher tear osmolarity, and (2) the eye with the highest corneal fluorescein staining.
Statistical Analysis
We used the Pearson or Spearman coefficients to analyze the correlation between the changes in tear osmolarity, corneal fluorescein staining, and symptoms at 2 separate visits. Additionally, we investigated this correlation in the cases that met the criteria (cutoff) for dry eye proposed by Miller and associates for symptoms (OSDI >12), and by Lemp and associates for tear osmolarity (>308 mOsm). We then performed the paired correlations in a subgroup of patients who experienced the “minimal clinically important difference” between the two visits, as reported by Miller and associates, for mild to moderate DED (improvement or worsening of symptoms of ≥4.5 OSDI units). Next, we assessed the change for corneal fluorescein staining and tear osmolarity, in patients who experienced the “minimal clinically important difference” reported for severe DED (change of ≥9.9 OSDI units), by comparing the mean scores of the 2 visits using the Wilcoxon matched pairs test. We further subgrouped patients based on the therapy they received, and investigated the correlation between changes in the 3 variables measured.
Finally, we performed a multiple linear regression analysis to examine predictors of the change in OSDI (dependent variable), using the baseline OSDI score and either the changes in corneal fluorescein staining or tear osmolarity as independent variables. A value of P < .05 was considered statistically significant.
Results
We analyzed data on 186 subjects who met our inclusion criteria, with a mean time between the 2 visits of 137 ± 95 days. The mean age of patients was 54 ± 15 years, and 122 of 186 (66%) of the subjects were female. Across the entire study population, the mean OSDI, corneal fluorescein staining, and tear osmolarity changes were −2.3 ± 21.9, −0.5 ± 1.4, and 1.1 ± 23.6, respectively. Table 1 depicts a summary of the baseline values and changes for each variable, including a subdivision by patients who improved or worsened.
| Variable | All Subjects (Mean ± SD) | Improved (Mean ± SD) | Worsened (Mean ± SD) |
|---|---|---|---|
| OSDI score at baseline | 44.7 ± 23.1 | 51.4 ± 23.2 | 35.8 ± 18.6 |
| OSDI change | −2.3 ± 21.9 | −17.3 ± 14.2 | 16.1 ± 15.3 |
| CFS score at baseline a | 1.7 ± 1.2 | 2.4 ± 1.1 | 0.9 ± 0.8 |
| CFS change a | −0.5 ± 1.4 | −1.4 ± 0.9 | 1.2 ± 0.6 |
| Osmolarity score at baseline b | 312.8 ± 20.3 | 323.7 ± 17.7 | 302.3 ± 14.7 |
| Osmolarity change b | 1.1 ± 23.6 | −17.7 ± 14.4 | 18.5 ± 16.9 |
There was a statistically significant, though modest, correlation between the changes in corneal fluorescein staining and OSDI ( R = 0.31; P < .001). However, there was no statistically significant correlation between the recorded change in tear osmolarity and OSDI ( R = −0.091; P = .38), or between changes in tear osmolarity and corneal fluorescein staining ( R = −0.02; P = .80) ( Table 2 ; Figure 1 ).
| Number of Cases | Mean Age | Sex, % Female | Mean TOsm Value at Baseline (mOsm/L) | Mean CFS Value at Baseline (Oxford units) | Mean OSDI Value at Baseline | Coefficient of Correlation | P Value | |
|---|---|---|---|---|---|---|---|---|
| All cases | ||||||||
| CFS-OSDI | 146 | 53 ± 15 | F (64%) | – | 1.8 ± 1.2 | 45.3 ± 23.0 | 0.31 | <.001 |
| TOsm-CFS | 135 | 54 ± 15 | F (64%) | 312.1 ± 19.3 | 1.6 ± 1.2 | – | −0.02 | .80 |
| TOsm-OSDI | 97 | 53 ± 14 | F (60%) | 312.8 ± 19.5 | – | 43.0 ± 21.8 | −0.091 | .38 |
| OSDI >12 | ||||||||
| CFS-OSDI | 138 | 53 ± 15 | F (64%) | – | 1.8 ± 1.2 | 47.5 ± 21.7 | 0.30 | <.001 |
| TOsm-CFS | 94 | 53 ± 15 | F (62%) | 312.2 ± 19.4 | 1.6 ± 1.2 | – | 0.02 | .84 |
| TOsm-OSDI | 90 | 52 ± 14 | F (61%) | 312.6 ± 19.3 | – | 45.8 ± 20.1 | −0.10 | .36 |
| TOsm >308 | ||||||||
| CFS-OSDI | 66 | 53 ± 13 | F (64%) | – | 1.8 ± 1.1 | 45.3 ± 21.6 | 0.35 | .005 |
| TOsm-CFS | 71 | 53 ± 14 | F (63%) | 326.5 ± 13.7 | 1.8 ± 1.2 | – | −0.07 | .59 |
| TOsm-OSDI | 53 | 52 ± 13 | F (57%) | 326.8 ± 13.3 | – | 42.8 ± 19.9 | 0.003 | .98 |
| MCID OSDI (>4.5) | ||||||||
| CFS-OSDI | 66 | 56 ± 15 | F (61%) | – | 1.7 ± 1.2 | 36.7 ± 17.8 | 0.46 | <.001 |
| TOsm-CFS | 35 | 52 ± 14 | F (57%) | 315.5 ± 19.0 | 1.6 ± 1.0 | – | −0.09 | .58 |
| TOsm-OSDI | 37 | 54 ± 15 | F (59%) | 315.8 ± 19.4 | – | 37.6 ± 18.7 | −0.12 | .47 |
When we assessed the level of correlation between changes in corneal fluorescein staining and tear osmolarity from the same eye, we found no significant correlation when selecting either the eyes with higher tear osmolarity ( R = 0.02; P = .78), or the eyes with higher corneal fluorescein staining ( R = −0.11; P = .21) from each patient at baseline.
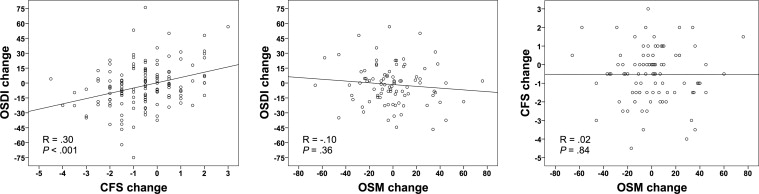
OSDI >12 is believed to signify patients with “ocular surface disease.” By limiting our analysis to this group of patients (OSDI >12 at baseline), we noted a statistically significant, though modest, correlation between changes in corneal fluorescein staining and OSDI ( R = 0.30; P < .001). However, in the same group of patients, we did not find a significant correlation between change in tear osmolarity and changes in OSDI ( R = −0.10; P = .36) or corneal fluorescein staining ( R = 0.02; P = .84) ( Table 2 ; Figure 2 ).
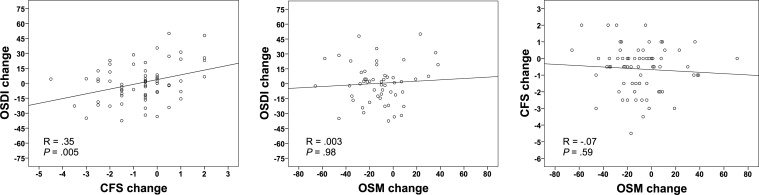
A tear osmolarity value >308 is recommended to be used as a threshold for the most sensitive detection of DED. In this subset of patients, there was a modest, though statistically significant, correlation between changes in corneal fluorescein staining and OSDI ( R = 0.35; P = .005). However, there was no statistically significant correlation between tear osmolarity and OSDI changes ( R = 0.003; P = .98), or between tear osmolarity and corneal fluorescein staining changes ( R = −0.07; P = .59) ( Table 2 ; Figure 3 ). When we analyzed only patients with tear osmolarity >314 mOsm/L at baseline (meant to represent the most specific threshold to detect DED) the findings were similar to those with >308 mOsm/L; there was a statistically significant correlation between changes in corneal fluorescein staining and OSDI ( R = 0.29; P = .04), whereas no significant correlation was present between change in tear osmolarity and OSDI ( R = 0.09; P = .59) or between tear osmolarity and corneal fluorescein staining ( R = −0.11; P = .44).
When we analyzed the subset of patients in whom there was at least a “minimal clinically important difference” in symptoms of DED (≥4.5 OSDI units), the positive correlation between changes in corneal fluorescein staining and OSDI was higher than that observed in the total cohort ( R = 0.46; P < .001). In contrast, there was still no statistically significant correlation between change in tear osmolarity and either OSDI ( R = −0.12; P = .47) or corneal fluorescein staining ( R = −0.09; P = .58) ( Table 2 ). The positive and statistically significant correlation between OSDI and corneal fluorescein staining changes was maintained for other “minimal clinically important difference” cutoffs of OSDI, that is, >7 (for all DED severity categories) and >9.9 (for severe DED). Tear osmolarity changes did not show a significant correlation at any of those cutoffs (data not shown). Interestingly, in patients with ≥9.9 OSDI change (minimal clinically important difference reported by Miller for patients with severe DED), osmolarity scores did not change between the 2 visits ( P = .97). In the same population corneal fluorescein staining scores reflected a statistically significant difference, decreasing by a mean of 0.5 Oxford units or 26% ( P < .001).
When divided into treatment subgroups ( Table 3 ), the correlations between changes in corneal fluorescein staining and OSDI maintained statistical significance (except for topical autologous serum): topical cyclosporine A (n = 42, R = 0.51); topical corticosteroids (n = 34, R = 0.40); topical anakinra (n = 43, R = 0.40); oral tetracycline (n = 31, R = 0.48); topical autologous serum (n = 11, R = 0.17, P = .63). None of the treatment subgroups resulted in a statistically significant correlation between changes in tear osmolarity and corneal fluorescein staining; the highest degree of correlation was achieved in the cyclosporine A–treated group (n = 42, R = 0.11, P = .59). Similarly, the highest degree of correlation between changes in tear osmolarity and OSDI was only .02 ( P = .94), as noted in the cyclosporine A–treated subgroup (n = 42). There was a statistically significant ( P = .04) correlation between changes in tear osmolarity and OSDI in the autologous serum–treated subgroup (n = 11), but the correlation was negative ( R = −0.78), suggesting that as OSDI decreased tear osmolarity paradoxically increased .



