Purpose
To compare visual acuity, accommodation, and contrast sensitivity of the AkkoLens Lumina accommodative intraocular lens (AkkoLens Clinical b.v., Breda, The Netherlands) with a standard monofocal intraocular lens (IOL).
Design
Randomized clinical trial.
Methods
The study enrolled 86 eyes with cataract that all required cataract surgery and IOL implantation. The study group included 61 eyes that were implanted with the Lumina. The control group included 25 eyes that were implanted with an Acrysof SA60AT (Alcon, Fort Worth, TX, USA) monofocal IOL. The distance and near visual acuities, contrast sensitivity, and accommodation were measured over a 1-year follow-up period. Accommodation was measured subjectively, using defocus curves, and objectively, with an open-field autorefractor.
Results
Uncorrected (UDVA) and corrected (CDVA) distance visual acuities did not differ significantly between the groups ( P ≥ .21) over the 12 months. However, the uncorrected near visual acuity (UNVA) was 0.07 ± 0.08 logRAD for the Lumina group and 0.37 ± 0.19 logRAD for the control group ( P < .01) and the corrected distance near visual acuity (CDNVA) was 0.11 ± 0.12 LogRAD for the Lumina group and 0.41 ± 0.15 LogRAD for the control group ( P < .01). Defocus curves showed a statistically significant difference between groups for defocus ranging from −4.50 to −0.50 diopters (D) ( P < .01) with significantly higher visual acuities for the Lumina group. Subjective accommodation, as determined from defocus curves, was 3.05 ± 1.06, 3.87 ± 1.27, and 5.59 ± 1.02 D for the Lumina group and 1.46 ± 0.54, 2.00 ± 0.52, and 3.67 ± 0.75 D for the control group at visual acuities of 0.10, 0.20, and 0.4 logMAR for both groups, respectively. The objective accommodation, measured by an open-field autorefractor, was 0.63 ± 0.41, 0.69 ± 0.45, 0.91 ± 0.51, and 1.27 ± 0.76 D for the Lumina group and 0.10 ± 0.15, 0.12 ± 0.15, −0.06 ± 0.09 and 0.07 ± 0.10 D for the control group at accommodation stimuli of 2.0, 2.5, 3.0, and 4.0 D, respectively. Contrast sensitivity was the same for both groups ( P ≥ .26).
Conclusions
The Lumina accommodative IOL effectively restores the visual function, accommodation, and contrast sensitivity after cataract surgery with no influence on the postoperative contrast sensitivity.
Recently new types of intraocular lenses (IOLs), including multifocal lenses and truly accommodating lenses, have become available, all of which aim to offer spectacle independence to the patient by providing adequate uncorrected distance vision in combination with adequate uncorrected intermediate and near vision.
Truly accommodating lenses adjust focus for different distances. Different concepts for accommodating lenses were proposed. One concept employs axial shift of a fixed-power lens in the eye. Forward and backward movement of such lens along the optical axis, a movement caused by the ciliary muscle, was expected to change the eye’s dioptric power. However, several studies now show that such axial-shifting lenses provide only limited, or no, improvement in near vision. The other concept is based on curvature change, which changes the refractive power of the lens.
An intraocular lens can be positioned inside the capsular bag or, alternatively, can be positioned at the sulcus plane, in front of the bag. It was reported that capsular fibrosis in primate eyes can noticeably hamper lens movement with the lens in the capsular bag after only several months postoperatively. A sulcus placement separates the lens from the bag and thus reduces the risk of the adverse reactions owing to the postoperative capsular bag fibrosis, hardening, and shrinkage.
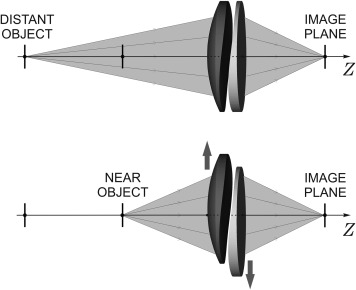
In this paper we report the results of a clinical study with the Lumina accommodative lens. The Lumina is designed for positioning at the sulcus plane, but the haptics on the flanges rest on top of the ciliary mass and do not significantly extend into the sulcus. The Lumina optical arrangement encompasses variable-focus optics according to Alvarez and additional free-form surfaces to reduce undesirable variable aberrations of the lens. The 2 optical elements of the Lumina slide in a plane perpendicular to the optical axis and produce a continuous variable-focus lens ( Figure 1 ). The lens is positioned in the sulcus plane in front and on top of the capsular bag and is driven directly, without interference of the capsular bag, by the ciliary muscle. This allows the eye to focus continuously from far to near.
Methods
Patients
This prospective, consecutive, comparative, and randomized clinical trial included 94 eyes of patients aged between 43 and 85 years implanted either with the Lumina lens or with a monofocal control IOL. Patients were informed about the purpose of the study and signed the informed consent form. The study complied with the tenets of the Declaration of Helsinki and it was approved by the Competent Authorities (CA) in Bulgaria, including the Ethics Committee for Multicenter Clinical Trials (ECMCT) of the Ministry of Health of Bulgaria (application No. KI-1097/07.11.2012, study protocol no. 1101-016 v04) and the Bulgarian Drug Agency (permission no. KI-MI-0007/26.11.2012 for the prospective clinical study in compliance with the same protocol) and, thus, registered with the European Medicines Agency (30 Churchill Place, Canary Wharf, London E14 5EU, UK). The study also complied with the HIPAA requirements and was completed within the required timeframe.
The following eyes were included in the study: eyes with clinical cataract (Lens Opacities Classification System III), causing a significant reduction of vision, plus eyes with no other ocular comorbidity, plus eyes with an expected positive effect of the surgery on the visual outcome, plus eyes with no previous ocular surgeries in the eye selected for the study. The other eye of the patient should present a normal condition, absence of any sight-threatening ocular comorbidity, no cataract, or a healthy pseudophakic eye. Only patients with less than 2 diopters (D) of corneal astigmatism were included in the study with no significant corneal irregularities as measured by corneal topography (Atlas 9000; Zeiss) and with axial lengths ranging from 21.5 to 25.0 mm (IOLMaster; Zeiss).
The randomization procedure was as follows: (1) a randomized table of study lenses and control lenses was prepared at the start of the study, and (2) once a patient signed the informed consent this patient was automatically assigned the next slot in the randomized table. All eyes underwent largely standard cataract surgery. In total, 94 eyes were screened, resulting in 86 randomized eyes because of randomization failures and withdrawal of informed consent. During the study, 3 control eyes and 2 Lumina eyes had early discontinuation because the patients passed away. In 1 case a posterior capsular tear occurred in an eye to be implanted with the Lumina during the surgery. This eye was therefore implanted with a 3-piece MA60 Acrysof Alcon lens (Alcon, Fort Worth, TX, USA) at the sulcus plane.
By the design of the clinical trial, 61 eyes were assigned to a study group, implanted with the Lumina accommodative intraocular lens (AIOL; AkkoLens international b.v., Breda, The Netherlands), and 25 eyes were assigned to a control group implanted with a standard monofocal IOL (Acrysof SA60AT; Alcon, Fort Worth, TX, USA).
The unequal sample sizes resulted from the 2-phase design of the clinical investigation according to the recommendations of the ANSI Z80 committee (Z80.29, Rev 016, Annex B, November 2011). In compliance with the recommendations, a performance endpoint of 1 D of objective accommodation should be demonstrated in phase I, with ∼40 study eyes at 4–6 months postoperatively, prior to phase II, when the majority of controls are implanted for the following comparative analysis.
All eyes in the present clinical trial were evaluated at 5 regular postoperative visits during a follow-up period of 12 months.
Intraocular Lenses
Figure 2 shows the Lumina study lens, which is designed and produced by AkkoLens Clinical, The Netherlands. The lens consists of 2 optical elements, each having an elastic, Ω-shaped loop with a spring function, and nonelastic connections to the main body of the lens. The lens is positioned at the sulcus plane and the ciliary muscle contacts the body of the lens and drives accommodation directly. The optics of the Lumina provides a fixed optical power to correct the refractive error of the aphakic eye. The fixed power is distributed over the 2 aspheric optical elements, with the anterior element providing 5.0 D and the posterior element 10–25 D, which power depends on the required correction of the eye. A variable optical power results from 2 mutually shifted internal aspheric surfaces whose power increases linearly with the shift.

In the unaccommodated eye, with the eye focused at far, the ciliary muscle has the largest diameter. The Ω-shaped springs of the Lumina are relaxed, the optical elements overlap, the lens shows its lowest optical power, and its optical diameter is ∼5.7 mm. When the eye accommodates, the ciliary muscle contracts and compresses the Lumina, resulting in mutual shift of the optical elements. The optical power increases linearly with the shift and the eye focuses at closer distances.
The size of the Lumina is customized for each patient eye based on the measured sulcus-to-sulcus diameter, by OCT (Visante OCT; Zeiss), at the 12 o’clock meridian (90-degree positions). This method allows an accurate estimation of the distance between the uttermost peripheral edges of the iris pigmentary epithelium, in that particular meridian, from the color-coded OCT images of the eye.
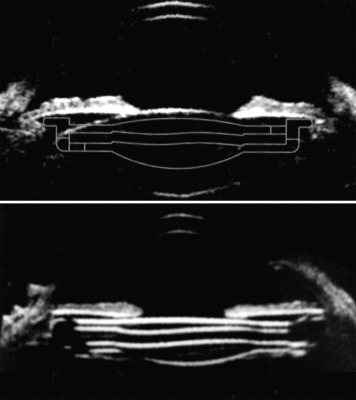
Figure 3 shows positioning of the Lumina at the sulcus plane by ultrasound biomicroscopy images. The fixed optical power of the lens (ie, the power required for an emmetropic eye) was calculated from optical biometry (IOL Master; Zeiss) by using proprietary C++ software integrated with Zemax ray-tracing software (Zemax, USA). The Lumina lenses were manufactured by CNC diamond lathing from Contaflex CI26 material (Contamac, UK), a widely used biocompatible hydrophilic acrylic polymer containing a UV-blocking agent. The Acrysof SA60AT control lens is a monofocal spheric acrylic foldable single-piece lens with an optic diameter of 6.0 mm and an overall diameter of 13.0 mm.
This investigation was preceded by a pilot feasibility study, also performed at Vissum Alicante, with 8 Lumina lenses. These eyes were implanted by the same surgeon (J.L.A.) in partially sighted eyes. These eyes are now followed up for more than 5 years and have shown no significant adverse events to date.
Surgical Technique
All surgeries were performed by J.L.A. at the Resbiomed-Vissum Eye Clinic in Sofia, Bulgaria. Procedures included phacoemulsification, retrobulbar anesthesia, and, in all cases, mild sedation with midazolam. Adequate dilation was with intracameral mydriasis of 0.1 mL of a vial containing cyclopentolate 1%, phenylephrine 10%, lignocaine 2%, and a balanced saline solution (BSS, 10 mL). The incision was at the ∼12 o’clock, 90 degree position in all eyes. The Lumina was implanted through a 2.8- to 3.0-mm incision in the sulcus plane of the eye by using a standard disposable injector system with an adapted, proprietary, AkkoLens butterfly cartridge. The Acrysof SA60AT control lenses were implanted in the capsular bag by a suitable injector. The IOL spherical power was calculated by a standard method for the control lenses and by ray-tracing (as explained above) for the Lumina lenses, respectively, to reach emmetropia in both groups. Postoperative topical therapy included a combination of topical antibiotics (ofloxacin 0.3% [Exocin]) and a steroid (dexamethasone 0.1%, Maxidex; Alcon, Spain). A video clip (Supplemental Material available at AJO.com ) shows the implantation of the Lumina AIOL.
Ocular Examinations
All eyes underwent preoperative evaluation including measurements of refraction, keratometry, distance and near visual acuities, slit-lamp examination, tonometry, and examination of the fundus. Distance visual acuity was measured under photopic conditions at 4 meters with standard Snellen charts and by a computerized LCD chart system (CC-100XP; Topcon). Additional measurements included corneal topography (Atlas 9000; Zeiss), anterior segment optical coherence tomography (Visante OCT; Zeiss), and posterior segment optical coherence tomography (Cirrus OCT; Zeiss). The procedures and test conditions complied with the requirements of the EN ISO 11979-7 standard (Ophthalmic implants – Intraocular lenses – Part 7: Clinical investigations) and the recommendations of the ANSI Z80 committee.
Patients were masked for the investigator carrying out the evaluations, meaning that the investigator was not informed as to whether the patient was implanted with the study lens or the control lens. The patients were masked at the beginning of the study, but some patients found out that they had received an accommodative IOL once they started to accommodate. Eyes were assessed at 1 day, 1 month, 3 months, 6 months, and at 12 months postoperatively. In addition to the measurements listed in the preoperative protocol, the postoperative protocol included additional assessments of near visual acuity using Radner reading charts (logarithm of reading acuity determination, logRAD) and contrast sensitivity.
Monocular defocus curves were obtained with distance refractive correction under low ambient light to keep an open pupil to minimize the undesirable effect of extended depth of focus owing to a small pupil, which can mask true accommodation. Negative and positive spherical lenses were added in 0.50-D steps, producing a stimulus for the eye to accommodate in the range from −5.00 to +2.00 D. The means of the measured visual acuities are summarized in mean defocus curves in which the measurements at 0.00 D and −2.50 D correspond to corrected distance visual acuity (CDVA) and corrected distance near visual acuity (CDNVA), respectively, and intermediate visual acuity corresponds to the measurement at −1.50 D. Depth of focus was evaluated from defocus curves at the levels of visual acuity of 0.10, 0.20, and 0.40 logMAR. The depth of focus for a particular eye was defined as the width of the defocus curve, in diopters, at a given level of visual acuity.
Objective accommodation was measured by measuring the refractive power of the eye with the open-field autorefractor Grand Seiko WAM-5500 (Grand Seiko, Japan). The WAM allows continuous measurement of the eye refraction (at about 5 times per second) while the patient looks at an approaching target through an open-field semi-transparent screen. During the measurement the patient was provided, if necessary, with standard spectacles to correct for distance vision. An approaching target, a reading chart, was mounted on a sliding stage and provided accommodative stimuli of −2.00, −2.50, −3.00, and −4.00 D. The pupil size was continuously measured by the WAM.
Contrast sensitivity of distance corrected eyes was measured with the Topcon LCD display at the spatial frequencies of 1.50, 3.0, 6.0, 12.0, and 24.0 cycles/degree (cpd).
Statistical Analysis
The statistical analysis was performed by the SPSS statistics software (SPSS for Windows, version 15.0; SPSS, Chicago, Illinois, USA). The normality of all data samples was evaluated by the Kolmogorov-Smirnov test. The Student t test was used for the parametric analysis of both paired data, for example comparison of preoperative and postoperative data of the same eye, and for unpaired data, for example comparison between groups. When the parametric analysis was not possible, the Wilcoxon rank-sum test was applied to assess the significance of differences between preoperative and postoperative data, whereas the Mann-Whitney test was used to compare the parameters between groups. A level of significance of P < .05 was applied to all tests. Main outcomes measures were distance and near visual acuities, defocus curve, objective accommodation in diopters, depth of focus, and contrast sensitivity function.
Results
All eyes were evaluated over 1 year postoperatively. No relevant complications occurred during this study except in the 1 eye, mentioned earlier, that received the control lens instead of the Lumina.
Intraocular inflammation other that the one normally observed postoperatively following cataract surgery did not occur in this study or in the preceding safety trial with a >4-year follow-up period. During the study, 10 Lumina eyes had to be treated after 3–12 months for posterior capsule opacification (PCO) by a standard yttrium-aluminum-garnet (YAG) capsulotomy. After the YAG treatment visual acuity and accommodation were restored almost immediately. No cases led to a chronic increase in intraocular pressure. Pigment particles adhering to the top of the iris were observed in some patients; these particles had no medical consequences.
Table 1 shows the preoperative conditions of patients included in this study. There were no statistically significant differences in age, mean keratometry, or axial length ( P ≥ .08) between the control and the study groups.
| Parameter | Lumina Intraocular Lens | Monofocal Control Lens | P Value |
|---|---|---|---|
| Age (y) | 69.44 (8.67) 46–85 | 67.52 (9.58) 43–80 | .44 |
| Sphere (D) | −1.40 (2.66) −11.00 to +2.25 | −0.81 (2.87) −5.00 to +3.00 | .64 |
| Cylinder (D) | −1.02 (0.43) −2.00 to −0.50 | −0.90 (0.42) −1.50 to 0.50 | .65 |
| Mean keratometry (D) | 43.60 (1.32) 40.91–46.78 | 43.80 (1.94) 40.62–48.26 | .53 |
| Axial length | 23.68 (0.65) 22.34–25.00 | 23.34 (0.49) 22.50–24.27 | .08 |
Visual and Refractive Outcomes
Tables 2, 3 , and 4 show the visual and refractive outcomes at 1 month, 6 months, and 12 months after surgery, respectively. There are no statistically significant differences in uncorrected distance visual acuity (UDVA) and CDVA between groups during postoperative evaluation ( P ≥ .21). At 12 months, the cumulative CDVA shows that 100% of eyes in both groups reached a CDVA of 0.10 logMAR. The values of UDVA were 0.04 ± 0.11 logMAR and 0.06 ± 0.18 logMAR in the Lumina and control groups, respectively, after 12 months postoperatively. However, the results show statistically significant differences in uncorrected near visual acuity (UNVA) and in CDNVA with significantly higher near visual acuities for the Lumina group ( P < .01) compared to the control group. Figure 4 represents the cumulative CDNVA at 12 months after surgery; 90.32% of eyes in the Lumina group and 0% of eyes in the control group showed a CDNVA of 0.10 logMAR. The UNVA was 0.07 ± 0.08 logMAR and 0.37 ± 0.19 logMAR for Lumina and control groups, respectively, 12 months after surgery. The CDNVA were 0.11 ± 0.12 logMAR and 0.41 ± 0.15 logMAR in the Lumina and control groups, respectively, 12 months after surgery.
| Parameter | Lumina Intraocular Lens | Monofocal Control Lens | P Value |
|---|---|---|---|
| LogMAR UDVA | 0.24 (0.36) −0.08 to 1.40 | 0.06 (0.11) −0.08 to 0.30 | .21 |
| Sphere (D) | −0.27 (1.10) −4.75 to +2.00 | +0.52 (0.81) −1.25 to +1.50 | <.01 |
| Cylinder (D) | −1.39 (0.79) −4.25 to −0.25 | −1.02 (0.60) −2.00 to 0.00 | .17 |
| LogMAR CDVA | 0.05 (0.26) −0.08 to 1.40 | 0.00 (0.06) −0.08 to 0.10 | .73 |
| LogRAD UNVA | 0.13 (0.14) 0.00–0.52 | 0.35 (0.16) 0.00–0.52 | <.01 |
| LogRAD CDNVA | 0.12 (0.20) −0.08 to 1.00 | 0.37 (0.18) 0.10–0.52 | <.01 |
| LogRAD CNVA | 0.02 (0.08) −0.08 to 0.30 | 0.06 (0.13) −0.08 to 0.40 | .51 |
| Parameter | Lumina Intraocular Lens | Monofocal Control Lens | P Value |
|---|---|---|---|
| LogMAR UDVA | 0.11 (0.22) −0.08 to 0.70 | 0.02 (0.06) −0.08 to 0.10 | .74 |
| Sphere (D) | 0.20 (0.99) −1.50 to +2.50 | +0.88 (0.94) −0.25 to +2.25 | .11 |
| Cylinder (D) | −1.30 (0.79) −3.75 to 0.00 | −1.06 (0.37) −1.50 to −0.50 | .47 |
| LogMAR CDVA | −0.02 (0.07) −0.18 to 0.22 | −0.01 (0.08) −0.08 to 0.22 | .65 |
| LogRAD UNVA | 0.08 (0.12) −0.20 to 0.30 | 0.40 (0.12) 0.22–0.52 | <.01 |
| LogRAD CDNVA | 0.11 (0.12) −0.08 to 0.40 | 0.39 (0.16) 0.00–0.52 | <.01 |
| LogRAD CNVA | −0.01 (0.06) −0.20 to 0.15 | 0.03 (0.12) −0.08 to 0.40 | .40 |
| Parameter | Lumina Intraocular Lens | Monofocal Control Lens | P Value |
|---|---|---|---|
| LogMAR UDVA | 0.04 (0.11) −0.18 to 0.30 | 0.06 (0.18) −0.18 to 0.52 | .93 |
| Sphere (D) | +0.35 (1.01) −1.75 to +2.50 | +1.03 (1.05) −0.75 to +3.25 | .06 |
| Cylinder (D) | −1.20 (0.67) −2.50 to 0.00 | −1.01 (0.57) −2.00 to 0.00 | .45 |
| LogMAR CDVA | −0.04 (0.05) −0.18 to 0.00 | −0.04 (0.07) −0.18 to 0.05 | .70 |
| LogRAD UNVA | 0.07 (0.08) 0.00–0.22 | 0.37 (0.19) 0.00–0.70 | <.01 |
| LogRAD CDNVA | 0.11 (0.12) 0.00–0.52 | 0.41 (0.15) 0.00–0.52 | <.01 |
| LogRAD CNVA | −0.02 (0.04) −0.10 to 0.10 | 0.01 (0.04) −0.08 to 0.10 | .23 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


