27
Vestibular Disorders
Mitchell K. Schwaber
Dizziness is the term used to describe a myriad of patient perceptions, including lightheadedness, syncope, disequilibrium, panic attacks, motion intolerance, visual disturbances, and true vertigo. It is the clinician’s responsibility to determine which of these perceptions the patient is experiencing by obtaining a careful history, often using descriptions in laymen’s terms. True vertigo is characterized by the perception that the external world is spinning, whirling, or swaying, and it is usually indicative of a vestibular disorder. Furthermore, true vertigo is usually accompanied by nystagmus. On the other hand, patients with nonvestibular dizziness more often perceive a sensation of movement or disorientation within their head, rather than the perception of the external world spinning. Also, nonvestibular dizziness is rarely accompanied by nystagmus. Chapter 14 discusses other features that can be helpful in differentiating vestibular and nonvestibular dizziness.
Once the clinician has established that the patient is experiencing vertigo, it is important next to determine if hearing loss is present or not. Depending on this determination, the most likely diagnosis can soon be reached.
 Episodic Vertigo with Hearing Loss
Episodic Vertigo with Hearing Loss
The differential diagnosis of episodic vertigo with hearing loss includes Meniere’s syndrome and Meniere’s disease, perilymph fistula, temporal bone fracture, syphilitic labyrinthitis, vascular occlusion of the labyrinthine artery, labyrinthine fistulas, labyrinthitis, and acoustic tumor. To rapidly differentiate these diagnoses, temporal bone fracture follows obvious head trauma; labyrinthine fistulas and labyrinthitis are associated with ear infections, pain, and drainage, or ear surgery; vascular occlusion is associated with a sudden single episode of vertigo with nearly total loss of hearing; Meniere’s syndrome is associated with ear fullness, fluctuating hearing loss, and episodic vertigo without antecedent factors; perilymph fistula is associated with sensorineural loss, vertigo, and disequilibrium that occur after an event associated with straining, lifting, or a surgical procedure; syphilitic labyrinthitis can be indistinguishable from Meniere’s syndrome and should be considered in any case that does not have a ready explanation, particularly when bilateral; acoustic tumor is usually associated with progressive sensorineural hearing loss, tinnitus, and usually only disequilibrium; ototoxicity is associated with the development of hearing loss, loss of balance, and ataxia during or immediately following a course of intravenous antibiotic therapy. Further information concerning each follows.
Meniere’s Syndrome and Meniere’s Disease
Meniere’s syndrome is defined as the clinical disorder associated with the histopathologic finding of endolymphatic hydrops. Clinically, Meniere’s syndrome includes the following features: recurrent, spontaneous episodic vertigo; hearing loss; aural fullness; and tinnitus. Under the most recent guidelines of the American Academy of Otolaryngology—Head and Neck Surgery,1 either tinnitus or fullness or both must be present on the affected side to establish the diagnosis. Recognized causes of Meniere’s syndrome include (1) idiopathic, also known as Meniere’s disease; (2) posttraumatic, following head injury or ear surgery; (3) postinfectious or delayed-onset Meniere’s syndrome following a viral infection, usually mumps or measles; (4) late-stage syphilis; (5) classic Cogan’s syndrome with episodic vertigo, hearing loss, interstitial keratitis, without syphilis; (6) variant Cogan’s syndrome with episodic vertigo, hearing loss, uveitis, or other ocular inflammation and without syphilis and other immune-mediated inner ear diseases.
Although Meniere’s disease is by far the most common cause of Meniere’s syndrome and the terms are often used interchangeably, it should be remembered that a patient has an idiopathic etiology only when the known causes have been excluded.
Clinical Presentation
Meniere’s disease is characterized by a history of increasing ear fullness with roaring tinnitus, followed by a sensation of blocked hearing. If the symptoms further worsen, a definitive vertigo spell may occur within a few minutes. Alternatively, patients may note the sudden onset of vertigo with little or no warning. Friberg et al2 found that the disorder begins with hearing loss alone in 42%, vertigo alone in 11%, vertigo with hearing loss in 44%, and tinnitus alone in 3%.
The definitive vertigo spell is spontaneous rotational vertigo lasting 20 minutes or longer, with accompanying nausea, vomiting, and other autonomic symptoms. Most vertigo episodes last from 2 to 4 hours, although some can last for more than 6 hours. During the episode of vertigo, horizontal or horizontalrotary nystagmus is always present. Following the vertigo episode, the patient may note that the hearing in the involved ear is markedly diminished. Disequilibrium may follow the definitive episode and may last for several days. Although the clinician might strongly suspect that the patient has Meniere’s disease, definitive diagnosis depends on the occurrence of two or more definitive episodes lasting 20 minutes or longer.
The hearing loss that occurs with Meniere’s disease typically begins with acoustic distortion that can be best described as a tinny quality to the signal. Another description is that only the higher-intensity peaks of certain frequencies are heard, whereas the rest of the frequencies are somewhat clipped or muffled. Loudness recruitment or loudness intolerance is also noted early in the illness, and is usually described by the patient as ordinary sounds being painfully loud in the affected ear. Early in the illness, many patients experience fluctuating hearing, with recovery beginning within a few hours of the episode. Hearing recovery may occur several days or months after a severe episode of vertigo. Either a shift of 10 dB or more in the average threshold of 0.5, 1, 2, and 3 kHz or a shift in speech discrimination of 15% is considered a significant change.
The hearing loss that accompanies Meniere’s disease typically follows one of three forms1: (1) a low-frequency sensorineural loss that is greatest at 250 Hz, 500 Hz, and at 1 kHz, with a normal threshold at 2 kHz, and a sensorineural loss above 2 kHz (Fig. 27–1); (2) a flat, moderately severe sensorineural loss at 500 Hz, 1 kHz, 2 kHz, and 3 kHz; (3) in patients with bilateral hearing loss, an asymmetry of greater than 25 dB in one ear. Most patients with Meniere’s disease, however, do experience a loss of hearing in the affected ear that slowly worsens over time, although it is extremely rare for a patient to lose all of the hearing in the ear. For most patients with Meniere’s disease, the hearing threshold usually stabilizes at 50 to 60 dB levels, with a flat configuration (“burned out ear”).
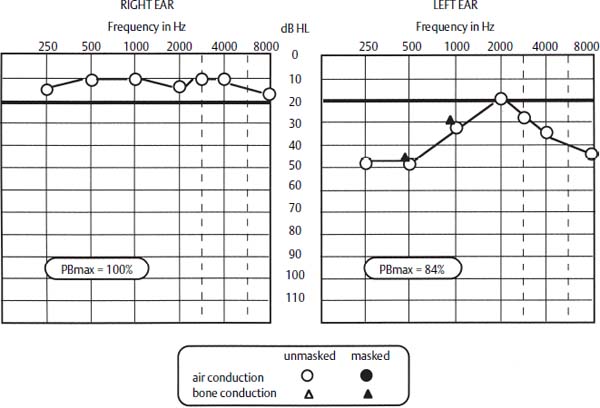
Figure 27–1 Audiogram of patient with Meniere’s disease. The left ear shows the typical audiometric pattern of early Meniere’s disease. PBmax, maximum-phonetic balance, or word-recognition, score.
Aural pressure, positional vertigo, and roaring tinnitus are extremely common between definitive episodes of severe vertigo, as is instability with fast movements as well as a constant rocking sensation. Following the onset, there may be a period of remission that can confuse the clinical picture. Over time, however, Meniere’s disease progresses from an early stage, through a middle stage, to a late or burn-out stage.
Meniere’s disease also presents in several atypical clinical forms, including Lermoyez’s variant, the otolithic crisis of Tumarkin or “drop attacks,” cochlear Meniere’s disease, vestibular Meniere’s disease, and delayed-onset Meniere’s syndrome. Lermoyez’s variant is associated with hearing improvement before, during, or after the vertigo episode. The otolithic crisis or drop attack is characterized by a sudden falling that is often accompanied by sudden firing of the extensor muscles of the extremities. The otolithic crisis is thought to represent firing of primitive muscle reflexes in response to sudden decompression of the saccule or utricle.
Cochlear and vestibular Meniere’s disease may represent early or possible Meniere’s disease, although the Committee on Hearing and Equilibrium1 has recommended that the use of these terms be discontinued. Cochlear Meniere’s disease is characterized by the auditory symptoms of Meniere’s disease, but none of the vestibular symptoms. Cochlear Meniere’s disease is thought to represent endolymphatic hydrops (ELH) confined to the cochlear duct. Vestibular Meniere’s disease is characterized by the vestibular symptoms of Meniere’s disease without the auditory symptoms. Although many of these patients progress to the classic form, other clinical entities include migraine, recurrent vestibular neuritis, and cochleovestibular nerve compression syndrome.3–5
Delayed-onset Meniere’s disease syndrome or postinfectious Meniere’s syndrome represents a distinct clinical entity, in which a patient with a long-standing unilateral sensorineural hearing loss begins to experience episodic vertigo indistinguishable from classic Meniere’s disease. There are two forms of the illness, ipsilateral and contralateral, indicating which ear is thought to be causing the problem. In the ipsilateral form, the deafened ear is usually found to have a caloric weakness on electronystagmography (ENG) testing. In the contralateral form, the better hearing ear begins to show fluctuating sensorineural levels, and on caloric testing shows a significant weakness. Schuknecht et al6 identified features in the temporal bones of these patients that suggested that these findings are due to a subclinical viral infection in childhood, but many clinicians feel delayed contralateral endolymphatic hydrops is immune or autoimmune.
The incidence of Meniere’s disease varies considerably in the various populations in which it has been studied.7 In the United States the incidence has been reported as between 15 and 40 per 100,000 per year, whereas in the United Kingdom the incidence is reported as 157 per 100,000. The incidence in Sweden is reported as 50 per 100,000. The symptoms of Meniere’s disease typically start at age 35 to 45, although later onset certainly occurs. There does seem to be a slight female-to-male preponderance, although the true incidence in males is probably underreported.
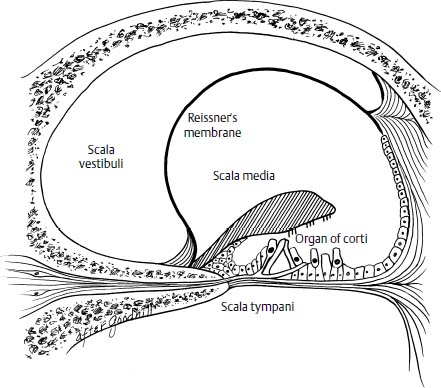
Figure 27–2 Histopathology of Meniere’s disease, demonstrating displacement of Reissner’s membrane and disruption of the organ of Corti by endolymphatic hydrops.
One third to one half of all patients have bilateral Meniere’s disease, and the vast majority becomes symptomatic in the other ear within the first 3 years of onset. Several reports suggest that Meniere’s disease, in particular bilateral Meniere’s disease, might be an immunologically related disorder.8,9 Hereditary, viral, noise, and allergic factors have also been identified, although their exact significance remains to be determined.10–12
Histopathology
Hallpike and Cairns13 and Yamakawa14 first reported the histopathologic finding of ELH in the temporal bones of patients with symptoms suggestive of Meniere’s syndrome. Most of the distention is seen in the cochlear and saccular ducts (Fig. 27–2), although occasionally the walls of the utricle and the ampullae are distorted. In some cases, the Reissner’s membrane is so distended that the space of the scala vestibulae is completely taken up by the scala media. Ruptures of the membranous labyrinth, fistulas between the endolymph and perilymph, collapse of the membranous labyrinth, and vestibular fibrosis further characterize the histopathologic picture in ELH.15
Altmann and Kornfield16 noted that only minimal histopathologic changes are seen in the sensory epithelia of these cases. However, other investigators have identified a variety of ultrastructural abnormalities including loss of inner and outer hair cells, and spiral ligament fibrocytes,17 as well as decreased strial vascularity.18
Endolymphatic hydrops has been documented at autopsy in a wide variety of disorders,19 including acoustic trauma, autoimmune inner ear disease, chronic otitis media, Cogan’s syndrome, congenital deafness, fenestration of the otic capsule, leukemia, Mondini dysplasia, otosclerosis, serous labyrinthitis, syphilis, temporal bone trauma, and viral labyrinthitis.
The exact mechanism by which idiopathic ELH occurs remains unproven, although it has become a central theory that ELH causes the symptoms of Meniere’s syndrome. Specifically, this theory states that it is the increased pressure that causes the ear fullness, the hyperacusis and distorted hearing, and the unsteadiness and disequilibrium. Furthermore, the theory holds that, if the membranous labyrinth breaks, the patient then suffers a severe episode of vertigo, with a decline in hearing and resultant tinnitus.
On the other hand, Rauch et al,20 in reviewing the temporal bone collection of the Massachusetts Eye and Ear Infirmary, have suggested that ELH does not cause the symptoms of Meniere’s syndrome, but rather is an epiphenomenon. These investigators theorize that ELH is perhaps an indicator of inner ear membrane dysfunction or failure, and that some other factor or factors usually affect fluid management within the inner ear. Altered fluid management then produces both symptoms and ELH. Similar conclusions were reached by Swart and Schuknecht,21 who reported the results of long-term destruction of the endolymphatic sac in monkeys.
Although the central role of ELH in Meniere’s syndrome remains controversial, several pathophysiologic mechanisms have been proposed to explain its development. The most prevalent theory of pathophysiology of ELH is that the distention of the endolymphatic system occurs because of excessive accumulation of endolymph, primarily due to altered resorption by the endolymphatic duct and sac.22 Altered resorption could be due to perisaccular and vestibular epithelial fibrosis,23–26 altered glycoprotein metabolism,27,28 viral infection of the inner ear,6,29,30 or immune-mediated injury.31,32 In addition, anatomic abnormalities in the bony structures surrounding the endolymphatic duct might also influence the development of ELH.26,33 A bony abnormality might cause only a marginal obstruction of the duct, but when added to these other factors would result in a much more severe obstruction to endolymph flow.
As a corollary to the theory of excessive accumulation of endolymph, increased endolymphatic pressure is thought by many to cause the symptoms in these cases.34,35 Furthermore, Zenner et al36 have demonstrated experimentally that the hearing loss and tinnitus of ELH could be caused by ruptures of the membranes lining the endolymphatic space, so that potassium-rich endolymph intoxicates the sensory and neural structures. As a result of this potassium influx into the perilymph, the outer hair cells are depolarized, with shortening and loss of motility. Both fluctuating and chronic hearing loss in Meniere’s syndrome can be explained by this model.
Other data tend to contradict the pressure hypothesis in EHL. For one thing, although various hearing abnormalities are commonly demonstrated35 in experimental hydrops, vestibular dysfunction is rarely observed in these models. Also, Long and Morizono37 measured the pressure gradients in experimental hydrops using microelectrode recording techniques, and found differences between endolymph and perilymph pressure that approximated 0.5 mm Hg. These investigators suggested that as pressure gradients increased, various corrective mechanisms such as an ion balance, endolymph secretion, and absorption are modulated to return the gradient to 0 mm Hg.
Kitahara et al,38 in a series of elegant pressure studies using artificial endolymph, demonstrated that the auditory and vestibular abnormalities found in ELH most likely arise from biochemical rather than pressure alterations. Juhn et al39 essentially concluded the same in their review of the subject. Following obliteration of the endolymphatic duct of guinea pigs, experimental findings, in addition to ELH, include a decrease in the endocochlear potential,40 an increase in intracochlear calcium,41 alterations in potassium permeability and in the inhibition of the electrogenic transport processes,42 and increased endolymphatic fluid protein content.43 Furthermore, Juhn et al found that infusion of epinephrine into the bloodstream results in increased osmolality of the serum and the perilymph, and postulated that this contributes to the development of episodic attacks in ELH.
In summary, evidence exists that the symptoms of Meniere’s disease may be due to membranous ruptures or to some alteration in the biochemical gradients within the endolymphatic space. Undoubtedly, our theories of the pathophysiology of Meniere’s disease will seem rather primitive and naive in the future. At this writing, however, most investigators believe that idiopathic ELH or Meniere’s disease is a multifactorial illness, and that individuals might have more than one factor simultaneously contributing to the development of this problem. The proposed factors include autoimmune reactions, allergic responses, blocked venous drainage, excess endolymph production or decreased resorption, autonomic imbalances, viral infections, vascular irregularities, bony labyrinthine or mastoid air cell maldevelopment, migraine, noise, otosclerosis, and hereditary degeneration. In addition, it is well know among clinicians that patients with Meniere’s disease often have relatives with similar symptoms and many also have a childhood history of car sickness or motion intolerance. Whether any of these factors are proven in the future remains to be seen.
Evaluation
The single most important step in the evaluation of the patient with recurrent vertigo and hearing loss is the medical history. An experienced clinician can often formulate the most likely diagnosis through history alone, and if Meniere’s syndrome is suspected, the history should then focus on the exclusion of other conditions that can mimic this disorder. Specifically, the diagnoses of perilymph fistula, ototoxicity, chronic labyrinthitis, syphilis, autoimmune inner ear syndrome, and acoustic tumor should be excluded.
Perilymph fistula is usually associated with sudden, severe sensorineural loss with disequilibrium, and only occasional vertigo episodes. However, fluctuating hearing loss and ear fullness can also be seen with this disorder. Most cases of perilymph fistula are associated with straining, barometric pressure changes, or trauma, although definitive exclusion can only be accomplished through surgical exploration in some cases. Ototoxicity should be suspected if the patient has received intravenous therapy for an infection, more specifically receiving an aminoglycoside in the majority of cases. Ototoxicity should be further suspected if the patient complains of unsteadiness, staggering gait, movement of the visual field when walking, and a loss of balance rather than vertigo.
Chronic labyrinthitis should be suspected if the patient gives a history of ear drainage, ear pain, or prior ear surgery. Late-stage syphilitic labyrinthitis should be suspected in any patient with prior treatment for syphilis and with slowly progressive sensorineural hearing loss and progressive disequilibrium. Late-stage syphilitic labyrinthitis can also be clinically indistinguishable from Meniere’s disease, with the only differentiating point being positive serology tests or interstitial keratitis on slit-lamp examination. Autoimmune inner ear syndrome should be suspected in patients with rapidly progressive or bilateral sensorineural hearing loss with episodic vertigo. These patients may have other autoimmune disorders such as systemic lupus erythematosa, rheumatoid arthritis, or vasculitis. Also there may have been a positive response to steroid therapy in the past in these cases. Acoustic tumor cases are characterized by progressive unilateral sensorineural hearing loss, decreased speech discrimination, tinnitus, and disequilibrium rather than episodic vertigo.
On completing the initial history, the details of the illness including onset, associated symptoms, and duration and frequency of vertigo episodes, and the provoking factors should be determined. The presence of roaring tinnitus, ear fullness, and fluctuating hearing loss, and their relationship to the episodic vertigo should also be noted. The past medical history, the response to any prior treatments, and the family history of illness should then be recorded. Any related medical illnesses, such as adult-onset diabetes mellitus, migraine, or vascular insufficiency, in addition to their treatments, are then noted.
The next step in the evaluation of the patient with suspected Meniere’s syndrome is to perform the otolaryngologic and head and neck examination. Any external or middle ear disease is noted, as are any related findings. Attention is then turned to the neurotologic examination. Spontaneous nystagmus, usually with the fast component beating away from the affected ear, is often visible with Frenzel glasses. Head-shake nystagmus is then elicited by having the patient rapidly shake the head back and forth 15 to 20 times, and then observing spontaneous nystagmus beating away from the affected ear. The Romberg, Quix, and rapid turning tests often show a drift toward the affected side, particularly if the patient has recently experienced vertigo. Hennebert’s sign should be sought or a fistula test should be performed with either a pneumatic otoscope or by compressing the tragus into the canal. A deviation of the eyes away from the ear being tested is considered to be an objective positive test and is noted in some Meniere’s disease patients. A sensation of sway may be reported by the patient and is considered to be a subjectively positive symptom of dysfunction.
The audiometric evaluation of these patients includes a pure-tone audiogram with speech discrimination testing, tympanometry and imittance testing, and acoustic reflex decay testing. The pattern of results seen with Meniere’s disease was described earlier (see Clinical Presentation). Acoustic reflex decay is an indicator of retrocochlear abnormality, and suggests the need for additional studies to exclude the possibility of a tumor in the cerebellopontine angle. If the initial evaluation suggests the possibility of Meniere’s disease, additional studies to confirm the diagnosis may be obtained, including dehydration testing, electrocochleography (ECochG), and otoacoustic emissions.
Dehydration tests are performed using either furosemide 20 to 40 mg p.o., urea, or glycerol 75 to 100 cc p.o. These agents cause a rapid diuresis, and as a result improved hearing thresholds can be noted in patients with early Meniere’s disease.44 Although this information proves useful in some circumstances, most clinicians do not routinely use this study because of the unpleasant side effects of the test as well as the availability of other diagnostic studies. Alternatively, many clinicians utilize ECochG in the evaluation of these patients.45
ECochG45 uses computerized signal-averaging techniques to record the electrical signals from the cochlea and the auditory nerve in response to an auditory signal. Electrodes used for this study include transtympanic needles placed on the promontory of the cochlea, tympanic membrane electrodes, and ear canal electrodes. The major advantage of all three is an improved signal-to-noise ratio, so that the action potential or N – 1 is accentuated. With a variety of stimuli, including clicks and tone bursts with alternating polarity, the summating potential (SP) and action potential (AP) are further accentuated. Meniere’s disease is characterized by an enhanced SP, and, relative to the amplitude of the AP, an increased SP/AP ratio (Fig. 27–3). An SP/AP ratio above 0.40 is thought by many clinicians to indicate ELH,45 although the test-retest reliability and specificity of this value remain in question.46 This information is helpful in cases where the diagnosis is not certain, such as differentiating nonhydropic sensorineural loss from early Meniere’s disease, and in the evaluation of the opposite ear in suspected bilateral Meniere’s disease.47 Otoacoustic emissions may provide information concerning the status of the outer hair cells in ELH, but the clinical value of this test is not certain at present.
Vestibular testing provides limited though useful information in the evaluation of Meniere’s disease patients. Vestibular studies commonly employed include ENG, rotational testing, and computerized dynamic platform posturography (CDP). Vestibular function in Meniere’s disease patients fluctuates and is extremely variable, and as a result test data may be completely normal even in cases with active episodic vertigo. ENG enables separate evaluation of each labyrinth, and abnormalities including decreased caloric response and positional nystagmus are noted in 50% of Meniere’s disease patients.48 ENG can be helpful to confirm the presence of a vestibular disorder, either by documentation of a deficit or by reproducing the symptoms for the patient.
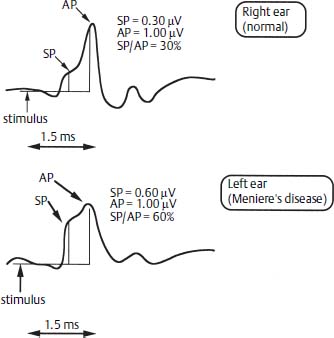
Figure 27–3 Electrocochleography demonstrates enhancing SP and SP/AP ratio in the left ear. SP, summating potential; AP, action potential.
Standard rotary chair testing has limited usefulness in the evaluation of patients with unilateral Meniere’s disease, because is evaluates both labyrinths simultaneously. During an acute vertigo episode, rotational testing can show an altered vestibulo-ocular reflex (VOR), specifically phase shift, decreased gain, and an asymmetric response. However, between episodes, rotary chair testing is usually normal. However, O’Leary and Davis49 have reported that with active head rotation or so-called vestibular autorotation in the vertical plane, Meniere’s disease patients typically demonstrate markedly increased gain. Rotational testing can be extremely helpful in the evaluation of compensation following vestibular surgery, bilateral vestibular disorders, and gentamicin treatment. When compensation has occurred, the VOR symmetry and gain are usually within the normal range.
The vestibular evoked myogenic potential (VEMP) can be used to evaluate the status of the saccule and the inferior vestibular nerve. The VEMP actually measures the contractions of the sternocleidomastoid muscle in response to acoustic stimulation of the saccule, that is, the sacculocolic reflex. VEMPs are records using a 750-Hz tone burst, at 95 dB HL. The stimulus rate is 4.3/second, with masking in the contralateral ear. The response is recorded using electrodes placed on the sternocleidomastoid muscle, which is placed under stretch tension. Usually 200 responses are averaged, and the threshold response is sought by lowering the signal intensity in 10 dB steps.
The VEMP response is a biphasic wave recorded between 13 and 23 ms after the stimulus, and its peaks are labeled P13 and P23, respectively. Alternatively, these two waves are labeled P1 and N1, as noted in Figure 27–4. The amplitude varies between 10 and 300 μV. The asymmetric ratio between sides utilizes a formula similar to that used for ENG. The peak-to-peak amplitude (A) is measured for both the left and right ears, AL and AR, respectively (Fig. 27–4):
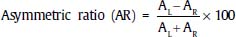
A percent difference greater than 35% is considered significant.
In approximately half of patients with active Meniere’s disease,50 the VEMP is asymmetric on the side of the affected ear; this is thought to indicate saccular hydrops,51 but no definitive data have yet proved this point. However, an asymmetric VEMP does indicate abnormal vestibular function, and therefore may confirm a history of Meniere’s syndrome or early ELH.
The CDP is useful in determining if a patient in fact has vestibular dysfunction,48 in measuring compensation after vestibular surgery, and in providing objective data to confirm the presence of a vestibular handicap. It is also useful in determining the proper physical therapy regimen for patients with stabilized vestibular deficits. However, it does not lateralize the side in Meniere’s disease nor does it provide pathognomonic findings to confirm Meniere’s disease.
Laboratory evaluation of patients with Meniere’s syndrome is aimed at excluding several readily identifiable disorders that can affect the patient with Meniere’s syndrome. These laboratory studies include fluorescent tregonemal antibody absorption (FTA-abs) or microhemagglutination-Treponema pallidum (MHA-TP) to exclude syphilis, fasting blood glucose to exclude adult-onset diabetes mellitus, cholesterol and triglyceride studies, and thyroid function tests. In addition, a large number of clinicians routinely obtain laboratory studies to exclude immune-related illnesses, particularly in bilateral Meniere’s syndrome. These studies include the sedimentation rate, antinuclear antibody, rheumatoid factor, serum immunoglobulins, and, in some cases of atopy, the radioallergosorbent test (RAST).19 If there is a strong suspicion of autoimmune inner ear syndrome, Cogan’s syndrome, or variant Cogan’s syndrome, specific testing with humerol heat shock protein-70 (HSP-70) or cellular COCH-protein testing may be obtained. Many clinicians use a positive response to a corticosteroid trial to denote a positive test for these disorders.
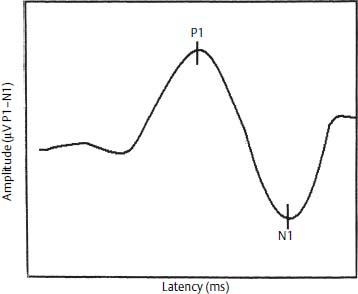
Figure 27–4 Vestibular evoked myogenic potential. P1 and N1 denote the peaks of biphasic waveforms.
Finally, on rare occasions acoustic tumors can present with episodic vertigo. If during the course of evaluating patients with suspected Meniere’s syndrome, the clinical features or audiometric or vestibular findings suggest the possibility of an acoustic tumor or another mass lesion, additional imaging studies should be performed. These studies should also be performed prior to any definitive surgical procedure. Diagnostic imaging usually includes magnetic resonance imaging (MRI) scans—either a focused internal auditory canal study (fast spin echo) or a contrasted study with gadolinium agents such as Magnevist. In some cases, auditory brainstem responses or high-resolution computed tomography (CT) scans of the temporal bone might provide additional information.
Classfication and Staging
The Committee on Hearing and Equilibrium1 of the American Academy of Otolaryngology-Head and Neck Surgery has recently updated the diagnosis and classification of Meniere’s disease, as noted in Table 27–1. The diagnosis takes into account the certainty of the clinical diagnosis and includes possible, probable, definite, and certain Meniere’s disease. A single definitive episode of vertigo with documented hearing loss is considered probable Meniere’s disease, whereas multiple episodes with hearing loss are definite Meniere’s disease. In staging Meniere’s disease, the Committee recommends that the stage be related to the hearing level only, as noted in Table 27–2. This staging system is in contrast to prior staging systems that were based on the frequency of vertigo episodes. The hearing level is based on a four-tone average of 0.5, 1, 2, and 3 kHz, obtained from the worst audiogram in the prior 6 months. The Committee noted that not all patients would progress through these stages in sequence. To quantify the episodic vertigo, the Committee recommended that the results be reported by comparing the number of episodes per month between 18 and 24 months postsurgery to the 6 months prior to the treatment. A ratio of the number of episodes between these two time intervals is multiplied by 100, to create a numerical value and related lettered classification, as noted in Table 27–3.
Medical Management
The medical management of Meniere’s disease is directed at decreasing the fluid volume of the endolymph, increasing the circulation of the inner ear, or altering the immune reactivity or blockage of the endolymphatic duct.19 None of these proposed regimens has ever been shown in a double-blind controlled study to be effective.52 Despite that fact, most clinicians utilize one or more of these management strategies in an effort to alter the natural history of the disorder.
| Certain Meniere’s disease |
| Definite Meniere’s disease, plus histopathologic confirmation |
| Definite Meniere’s disease |
| Two or more definitive spontaneous episodes of vertigo 20 minutes or longer |
| Audiometrically documented hearing loss on at least one occasion |
| Tinnitus or aural fullness in the treated ear |
| Other causes excluded |
| Probable Meniere’s disease |
| One definitive episode of vertigo |
| Audiometrically documented hearing loss on at least one occasion |
| Tinnitus or aural fullness in the treated ear |
| Other causes excluded |
| Possible Meniere’s disease |
| Episodic vertigo of the Meniere type without documented hearing loss, or |
| Sensorineural hearing loss, fluctuating or fixed, with disequilibrium but without definitive episodes |
| Other causes excluded |
| Stage | Four-Tone Average (dB) | |
|---|---|---|
| 1 | ≤25 | |
| 2 | 26–40 | |
| 4 | 41–70 | |
| 4 | >70 | |
*Staging is based on the four-tone average (arithmetic mean rounded to the nearest whole number) of the pure-tone thresholds at 0.5, 1, 2, and 3 kHz of the worst audiogram during the interval 6 months before treatment. This is the same audiogram that is used as the baseline evaluation to determine hearing outcome from treatment. Staging should be applied only to cases of definite or certain Meniere’s disease.
| Numerical Value* | Class | |
|---|---|---|
| 0 | A (complete control of definitive spells) | |
| 1 to 40 | B | |



